Summary
Background
Solid organ recipients have a substantial risk of developing bladder cancer, with high-risk non-muscle-invasive bladder cancer (NMIBC) being the most frequent diagnosis. Theoretically, adjuvant bacillus Calmette–Guérin (BCG) therapy is contraindicated, but limited data indicate its feasibility. The objective of this study was to evaluate the safety and efficacy of BCG following solid organ transplantation.
Materials and methods
We reviewed the data of four solid organ recipients who received adjuvant BCG for high-risk NMIBC at our institution. Additionally, individual data of 12 patients were extracted from case series and case reports, which were identified through a systematic review of the literature. A meta-analysis was performed.
Results
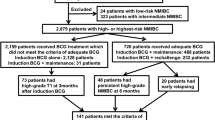
Fourteen patients (88 %) had received a kidney, one a heart, and one a liver transplant. The median time from transplantation to bladder cancer was 60.5 months. The regimen of immunosuppression was not modified in 12 patients (75 %). Forty-two percent of patients did not receive prophylactic antibiotics, and 70 % had no side effects. Ten patients (63 %) experienced recurrence after a median of 14 months. Progression to muscle-invasive or metastatic disease was observed in two patients (13 %). Four patients (25 %) underwent radical cystectomy, and two patients died of the disease.
Conclusions
BCG therapy is a safe option for patients with high-risk NMIBC following solid organ transplantation. However, there is a substantial risk of recurrence and progression. Urologists and patients considering BCG therapy should be aware of this and may consider early cystectomy. There is no evidence to support the need for prophylactic antibiotics.
Zusammenfassung
Hintergrund
Organtransplantierte Patienten haben ein signifikantes Risiko, ein Urothelkarzinom der Harnblase (HCA) zu entwickeln. Dabei sind oberflächliche HCA mit hohem Rezidiv- und Progressionsrisiko der häufigste Befund. Die adjuvante Instillationstherapie mit Bacillus Calmette-Guérin (BCG) ist kontraindiziert, obwohl einige Autoren diese Therapie beschrieben haben. Ziel dieser Arbeit ist die Evaluation der Sicherheit und Effektivität von BCG bei organtransplantierten Patienten.
Material und Methoden
Es wurden die unizentrischen Daten von vier transplantierten Patienten erhoben, welche sich aufgrund eines HCA einer adjuvanten BCG-Instillationstherapie unterzogen. Eine systematische Literaturrecherche mit Meta-Analyse von insgesamt 16 Patienten wurde durchgeführt.
Ergebnisse
Bei 14 Patienten (88 %) bestand ein Zustand nach Nierentransplantation und bei jeweils einem nach Herz- bzw. Lebertransplantation. Das mediane Intervall vom Zeitpunkt der Transplantation bis zum HCA betrug 60,5 Monate. Die Immunsuppression wurde bei 12/16 Patienten (75 %) nicht modifiziert. Insgesamt erhielten 42 % keine begleitende Antibiotikatherapie und 70 % hatten keine therapie-assoziierten Nebenwirkungen. Zehn Patienten (63 %) entwickelten ein Tumorrezidiv nach einem medianen Zeitraum von 14 Monaten. Eine HCA-Progression in ein muskelinvasives oder metastasiertes Stadium wurde bei zwei Patienten (13 %) beobachtet. Vier Patienten (25 %) unterzogen sich letztlich einer radikalen Zystektomie und zwei Patienten verstarben am HCA.
Schlussfolgerungen
BCG-Instillationen sind eine sichere Therapieoption bei organtransplantierten Patienten mit HCA, wobei ein signifikantes Risiko für Tumorrezidiv und Progress vorliegt. Eine frühe Zystektomie sollte daher in Erwägung gezogen werden. Für eine begleitende Antibiotikatherapie gibt es keine Evidenz.

Similar content being viewed by others
Abbreviations
- BCG:
-
Bacillus Calmette–Guérin
- NMIBC:
-
Non-muscle-invasive bladder cancer
- UBC:
-
Urothelial bladder cancer
- TURBT:
-
Transurethral resection of bladder tumor
References
Engels EA, Pfeiffer RM, Fraumeni JF Jr, Kasiske BL, Israni AK, Snyder JJ, et al. Spectrum of cancer risk among US solid organ transplant recipients. JAMA. 2011;306(17):1891–901.
Adami J, Gabel H, Lindelof B, Ekstrom K, Rydh B, Glimelius B, et al. Cancer risk following organ transplantation: a nationwide cohort study in Sweden. Br J Cancer. 2003;89(7):1221–7.
Wallerand H, Ravaud A, Ferriere JM. Bladder cancer in patients after organ transplantation. Curr Opin Urol. 2010;20(5):432–6.
Babjuk M, Oosterlinck W, Sylvester R, Kaasinen E, Böhle A, Palou-Redorta J, et al. EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder, the 2011 update. Eur Urol. 2011;59(6):997–1008.
Tomaszewski JJ, Larson JA, Smaldone MC, Hayn MH, Jackman SV. Management of bladder cancer following solid organ transplantation. Adv Urol. 2011;2011:256985.
Yossepowitch O, Eggener SE, Bochner BH, Donat SM, Herr HW, Dalbagni G. Safety and efficacy of intravesical bacillus Calmette-Guerin instillations in steroid treated and immunocompromised patients. J Urol. 2006;176(2):482–5.
Wang HB, Hsieh HH, Chen YT, Chiang CY, Cheng YT. The outcome of post-transplant transitional cell carcinoma in 10 renal transplant recipients. Clin Transplant. 2002;16(6):410–3.
Palou J, Angerri O, Segarra J, Caparros J, Guirado L, Diaz JM, et al. Intravesical bacillus Calmette-Guerin for the treatment of superficial bladder cancer in renal transplant patients. Transplantation. 2003;76(10):1514–6.
Elkentaoui H, Robert G, Pasticier G, Bernhard JC, Couzi L, Merville P, et al. Therapeutic management of de novo urological malignancy in renal transplant recipients: the experience of the French Department of Urology and Kidney Transplantation from Bordeaux. Urology. 2010;75(1):126–32.
Kamal MM, Soliman SM, Shokeir AA, Abol-Enein H, Ghoneim MA. Bladder carcinoma among live-donor renal transplant recipients: a single-centre experience and a review of the literature. BJU Int. 2008;101(1):30–5.
Shang PF, Kwong J, Wang ZP, Tian J, Jiang L, Yang K, et al. Intravesical Bacillus Calmette-Guerin versus epirubicin for Ta and T1 bladder cancer. Cochrane Database Syst Rev. 2011(5):CD006885.
Neuzillet Y, Davin JL, Lechevallier E. Utilisation des instillations intra-vésicales de bacille de Calmette et Guérin (BCG) pour le traitement des tumeurs superficielles de vessie chez les patients transplantés rénaux. Prog Urol. 2006;16(1):32–5.
Neuzillet Y, Lebret T. Les vraies contre-indications du BCG dans le traitement des tumeurs de vessie. Prog Urol. 2010;20 Suppl 1:S41–5.
Sylvester RJ, Brausi MA, Kirkels WJ, Hoeltl W, Calais Da Silva F, Powell PH, et al. Long-term efficacy results of EORTC genito-urinary group randomized phase 3 study 30911 comparing intravesical instillations of epirubicin, bacillus Calmette-Guerin, and bacillus Calmette-Guerin plus isoniazid in patients with intermediate- and high-risk stage Ta T1 urothelial carcinoma of the bladder. Eur Urol. 2010;57(5):766–73.
Sylvester RJ, van der Meijden AP, Lamm DL. Intravesical bacillus Calmette-Guerin reduces the risk of progression in patients with superficial bladder cancer: a meta-analysis of the published results of randomized clinical trials. J Urol. 2002;168(5):1964–70.
Yates DR, Roupret M. Contemporary management of patients with high-risk non-muscle-invasive bladder cancer who fail intravesical BCG therapy. World J Urol. 2011;29(4):415–22.
Chade DC, Shariat SF, Dalbagni G. Intravesical therapy for urothelial carcinoma of the urinary bladder: a critical review. Int Braz J Urol. 2009;35(6):640–50; discussion 51.
Fonseca FP, Bachega W Jr, Zequi SC, Sarkis AS, Guimaraes G, Priante AV, et al. Treatment of patients with superficial bladder cancer stratified by risk groups treated with lyophilized Moreau-Rio de Janeiro BCG strain. Int Braz J Urol. 2002;28(5):426–35; discussion 35–6.
Tillou X, Raynal G, Limani K, Saint F, Petit J. Carcinome in situ vésical et urétral chez un patient transplanté rénal: échec de la BCG thérapie. Prog Urol. 2008;18(13):1097–9.
Vegt PD, van der Meijden AP, Sylvester R, Brausi M, Holtl W, de Balincourt C. Does isoniazid reduce side effects of intravesical bacillus Calmette-Guerin therapy in superficial bladder cancer? Interim results of European Organization for Research and Treatment of Cancer Protocol 30911. J Urol. 1997;157(4):1246–9.
Rischmann P, Desgrandchamps F, Malavaud B, Chopin DK. BCG intravesical instillations: recommendations for side-effects management. Eur Urol. 2000;37 Suppl 1:33–6.
Durek C, Rüsch-Gerdes S, Jocham D, Böhle A. Interference of modern antibacterials with bacillus Calmette-Guerin viability. J Urol. 1999;162(6):1959–62.
Kasiske BL, Zeier MG, Craig JC, Ekberg H, Garvey CA, Green MD, et al. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant. 2009;9 Suppl 3:S1–155.
Conflict of interest
The authors declare that there are no actual or potential conflicts of interest in relation to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Swietek, N., Waldert, M., Susani, M. et al. Intravesical bacillus Calmette-Guérin instillation therapy for non-muscle-invasive bladder cancer following solid organ transplantation. Wien Klin Wochenschr 125, 189–195 (2013). https://doi.org/10.1007/s00508-013-0343-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-013-0343-1