Abstract
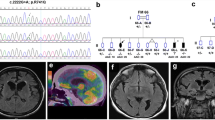
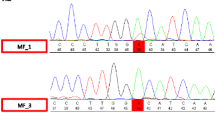
Homozygous mutations in the PINK1 gene have been shown to cause early-onset parkinsonism. Here, we describe a novel homozygous mutation (Q126P), identified in two affected German sisters with a clinical phenotype typical for PINK1-associated parkinsonism.We analysed lactate, pyruvate, carnitine and acylcarnitine blood levels, lactate levels under exercise and in the cerebrospinal fluid, activity of respiratory chain complexes I–IV in muscle biopsies and proteasomal activity in immortalized lymphoblasts, but found no evidence for mitochondrial or proteasomal dysfunction. MR spectroscopy revealed raised myoinositol levels in the basal ganglia of both patients, reflecting possible astroglial proliferation.
Similar content being viewed by others
References
Valente EM, Abou-Sleiman PM, Caputo V, et al. (2004) Hereditary early-onset Parkinson’s Disease caused by mutations in PINK1. Science 304:1158–1160
Hatano Y, Li Y, Sato K, et al. (2004) Novel PINK1 mutations in early-onset parkinsonism. Ann Neurol 56:424–427
Rogaeva E, Johnson J, Lang AE, et al. (2004) Analysis of the PINK1 gene in a large cohort of cases with Parkinson Disease. Arch Neurol 61:1898–1904
Ibáñez P, Lesage S, Lohmann E, et al. (2006) Mutational analysis of the PINK1 gene in early-onset parkinsonism in Europe and North Africa. Brain 129:686–694
Hedrich K, Hagenah J, Djarmati A, et al. (2006) Clinical spectrum of homozygous and heterozygous PINK1 mutations in a large German family with Parkinson disease: role of a single hit? Arch Neurol 63:833–838
Leutenegger AL, Salih MA, Ibáñez P, et al. (2006) Juvenile-onset parkinsonism as a result of the first mutation in the adenosine triphosphate orientation domain of PINK1. Arch Neurol 63:1257–1261
Valente EM, Salvi S, Ialongo T, et al. (2004) PINK1 mutations are associated with sporadic early-onset parkinsonism. Ann Neurol 56:336–341
Bonifati V, Rohé CF, Breedveld GJ, et al. (2005) Early-onset parkinsonism associated with PINK1 mutations. Neurology 65:87–95
Abou-Sleiman PM, Muqit MM, Mc- Donald NQ, et al. (2006) A heterozygous effect for PINK1 mutations in Parkinson’s disease? Ann Neurol 60:414–419
Silvestri L, Caputo V, Bellacchio E, Atorino L, Dallapiccola B, Valente EM, Casari G (2005) Mitochondrial import and enzymatic activity of PINK1 mutants associated to recessive parkinsonism. Hum Mol Genet 14:3477–3492
Deng H, Jankovic J, Guo Y, Xie W, Le W (2005) Small interfering RNA targeting the PINK1 induces apoptosis in dopaminergic cells SH-SY5Y. Biochem Biophys Res Commun 337:1133–1138
Petit A, Kawarai T, Paitel E, et al. (2005) Wild-type PINK1 prevents basal and induced neuronal apoptosis, a protective effect abrogated by Parkinson disease- related mutations. J Biol Chem 280:34025–34032
Park J, Lee SB, Lee S, et al. (2006) Mitochondrial dysfunction in Drosophila PINK1 mutants is complemented by parkin. Nature 441:1157–1161
Clark IE, Dodson MW, Jiang C, et al. (2006) Drosophila pink1 is required for mitochondrial function and interacts genetically with parkin. Nature 441:1162–1166
Yang Y, Gehrke S, Imai Y, et al. (2006) Mitochondrial pathology and muscle and dopaminergic neuron degeneration caused by inactivation of Drosophila Pink1 is rescued by Parkin. Proc Natl Acad Sci USA 103:10793–10798
Wang D, Qian L, Xiong H, et al. (2006) Antioxidants protect PINK1-dependent dopaminergic neurons in Drosophila. Proc Natl Acad Sci USA 103:13269–13270
Valente EM, Brancati F, Ferraris A, et al. (2002) PARK6-linked parkinsonism occurs in several European families. Ann Neurol 51:14–18
Fischer JC, Ruitenbeek W, Gabreels FJ, et al. (1986) A mitochondrial encephalomyopathy: the first case with an established defect at the level of coenzyme Q Eur J Pediatr 144:441–444
Barrientos A (2002) In vivo and in organello assessment of OXPHOS activities. Methods 26:307–316
Heinemeyer W, Fischer M, Krimmer T, Stachon U, Wolf DH (1997) The active sites of the eukaryotic 20 S proteasome and their involvement in subunit precursor processing. J Biol Chem 272:25200–25209
Seeger U, Klose U, Mader I, Grodd W, Nagele T (2003) Parameterized evaluation of macromolecules and lipids in proton MR spectroscopy of brain diseases. Magn Reson Med 49:19–28
Bentivoglio AR, Cortelli P, Valente EM, et al. (2001) Phenotypic characterisation of autosomal recessive PARK6- linked parkinsonism in three unrelated Italian families. Mov Disord 16:999–1006
Kantarci K, Petersen RC, Boeve BF, et al. (2004) 1H MR spectroscopy in common dementias. Neurology 63:1393–1398
O'Neill J, Schuff N, Marks WJ Jr, Feiwell R, Aminoff MJ, Weiner MW (2002) Quantitative 1H magnetic resonance spectroscopy and MRI of Parkinson’s disease. Mov Disord 17:917–927
Rango M, Arighi A, Biondetti P, et al. (2007) Magnetic resonance spectroscopy in Parkinson’s disease and parkinsonian syndromes. Funct Neurol 22:75–79
Firbank MJ, Harrison RM and O’Brien JT (2002) A comprehensive review of proton magnetic resonance spectroscopy studies in dementia and Parkinson’s disease. Dement Geriatr Cogn Disord 14:64–76
Shen J and Cookson MR (2004) Mitochondria and dopamine: new insights into recessive parkinsonism. Neuron 43:301–304
Tang B, Xiong H, Sun P, et al. (2006) Association of PINK1 and DJ-1 confers digenic inheritance of early onset Parkinson’s disease. Hum Mol Genet 15:1816–1825
Albanese A, Valente EM, Romito LM, Bellacchio E, Elia AE and Dallapiccola B (2005) The PINK1 phenotype can be indistinguishable from idiopathic Parkinson disease. Neurology 64:1958–1960
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prestel, J., Gempel, K., Hauser, T.K. et al. Clinical and molecular characterisation of a Parkinson family with a novel PINK1 mutation. J Neurol 255, 643–648 (2008). https://doi.org/10.1007/s00415-008-0763-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-008-0763-4