Summary
Background
Rooibos tea (Aspalathus linearis) is commonly used for hyperactive gastrointestinal, respiratory and cardiovascular disorders.
Aim of study
The aqueous extract of Rooibos tea (RT) was studied for the possible bronchodilator, antispasmodic and blood pressure lowering activities in an attempt to rationalize some of its medicinal uses.
Methods
Isolated tissue preparations, such as rabbit jejunum, aorta and guinea-pig trachea and atria were set up in appropriate physiological salt solutions and aerated with carbogen. For in vivo studies rats were anesthetized with pentothal sodium and blood pressure was measured through carotid artery cannulation.
Results
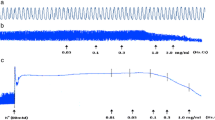
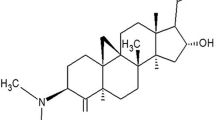
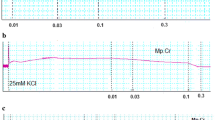
In jejunum, RT caused a concentration-dependent relaxation of low K+ (25 mM)-induced contractions, with mild effect on the contractions induced by high K+ (80 mM). In presence of glibenclamide, the relaxation of low K+-induced contractions was prevented. Similarly, cromakalim caused glibenclamide-sensitive inhibition of low K+, but not of high K+, while verapamil did not differentiate in its inhibitory effect on contractions produced by the two concentrations of K+. Like in jejunum, RT caused glibenclamide-sensitive relaxation of low K+-induced contractions in trachea and aorta, but with a 20 times higher potency in trachea. In atria, RT was least potent with weak inhibitory effect on atrial force and rate of contractions. RT caused a dose-dependent fall in arterial blood pressure in rats under anesthesia. Among the tested pure compounds of Rooibos, chrysoeriol showed selective bronchodilator effect. Chrysoeriol (luteolin 3′-methyl ether) is a bioactive flavonoid known for antioxidant, antiinflammatory, antitumor, antimicrobial, antiviral, and free radical scavenging activities.
Conclusion
These results indicate that the bronchodilator, antispasmodic and blood pressure lowering effects of Rooibos tea are mediated predominantly through KATP channel activation with the selective bronchodilatory effect. This study provides a sound mechanistic basis for the wide medicinal use of Rooibos tea, with the therapeutic potential to be developed for congestive respiratory ailments.





Similar content being viewed by others
References
Wyk BV, Oudtshoorn BV, Gericke N (2002) Medicinal plants of South Africa. Briza Publications, Pretoria, pp 48–49
Wesgro background report (2000) The Rooibos industry in the Western Cape. WESGRO, Western Cape investment and trade promotion agency, Cape Town, http://www.wesgro.org.za
Joubert E, Winterton P, Britz TJ, Ferreira D (2004) Superoxide anion and α,α-diphenyl-β-picrylhydrazyl radical scavenging capacity of Rooibos (Aspalathus linearis) aqueous extracts, crude phenolic fractions, tannin and flavonoids. Food Res Int 37:133–138
Duke JA, Bogenschutz-Godwin MJ, Du celliar J, Duke PAK (2002) Hand book of medicinal herbs, 2nd edn. CRC Press, Boca Raton, pp 612–613
Brown D (1995) Encyclopaedia of herbs and their uses. Dorling Kindersley, London, p 244
Nakano M (1997) Rooibos tea as an anti-aging beverage. Rooibos Limited, Clanwilliam, 8135
Inanami O, Asanuma T, Inukai N, Jin T, Shimokawa S, Kasai N, Nakano M, Sato F, Kuwabara M (1995) The supressión of age-related accumulation of lipid peroxides in rat brain by administration of Rooibos tea (Aspalathus linearis). Neurosci Lett 196:85–88
Marnewick J, Joubert E, Joseph S, Swanevelder S, Swart P, Gelderbolm W (2005) Inhibition of tumour promoton in mouse skin by extracts of rooibos (Aspalathus linearis) and honeybush (Cyclopia intermedia), unique Southern African herbal teas. Cancer Lett 224:193–202
Ulicna O, Vancova O, Bozek P, Carsky J, Sebekova K, Boor P, Nakano M, Greksak M (2006) Rooibos tea partially prevents oxidative stress in streptozotocin-induced diabetic rats. Physiol Res 55:157–164
Na HK, Mossanda KS, Lee JY, Surh YJ (2004). Inhibition of phorbol ester-induced COX-2 expression by some edible African plants. Biofactors 21:149–153
Gilani AH, Khan A, Ghayur MN, Ali SF, Herzig JW (2006) Antispasmodic effect of Rooibos tea (Aspalathus linearis) is mediated predominantly through K+ channel activation. Basic Clin Pharm Toxicol 99:365–373
National Research Council (1996) Guide for the care and use of laboratory animals. National Academy Press, Washington, pp 1–7
Evans WC (1996) Trease and Evan’s pharmacognosy, 14th edn. WB Sounders, London, pp 161–408
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and oxidants by means of Folin-Ciocalteu Reagent. Methods in Enzymol 299:152–178
Huang DJ, Lin CD, Chen HJ, Lin YH (2004) Antioxidant and antiproliferative activaties of sweet potato (Ipomoea batatas [L.] Lam ‘Tainong 57’) constituents. Bot Bull Acad Sin 45:179–186
Gilani AH, Shah AJ, Ghayur MN, Majeed K (2005a) Pharmacological basis for the use of turmeric in gastrointestinal and respiratory disorders. Life Sci 76:3089–3105
Frank H, Puschmann A, Schusdziarra V, Allescher HD (1994) Functional evidence for a glibenclamide-sensitive K+ channels in rat ileal smooth muscle. Eur J Pharmacol 271:379–386
Quest U (1992) Potassium channel openers: Pharmacological and clinical aspects. Fund Clin Pharmacol 6:279–293
Pelaia G, Gallell L, Vatrella A, Grembiale RD, Maselli R, De Sarro GB, Marsico SA (2002) Potential role of potassium channel openers in the treatment of asthma and chronic obstructive pulmonary disease. Life Sci 70:977–990
Hamilton TC, Weir SW, Weston TH (1986) Comparison of the effects of BRL34915 and verapamil on electrical and mechanical activity in rat portal vein. Br J Pharmacol 88:103–111
Aguilar-Bryan L, Clement JP, Gonzalez G, Kunjilwar K, Babenko A, Bryan J (1998) Towards understanding the assembly and structure of KATP channels. Physiol Rev 78:227–245
Weston AH, Edwards G (1992) Recent progress in potassium channel opener pharmacology. Biochem Pharmacol 43:47–54
Revuelta MP, Cantabrana B, Hidalgo A (1997) Depolarization-dependent effect of flavonoids in rat uterine smooth muscle contraction elicited by CaCl2. Gen Pharmacol 29:847–857
Sanchez de Rojas, Somoza VR, Ortega BT, Villar AM (1996) Isolation of vasodilatory active flavonoids from the traditional remedy Satureja obovata. Planta Med 62:272–274
Ripoll C, Lederer WJ, Nichols CG (1990) Modulation of ATP-sensitive K+-channel activity and contractile behavior in mammalian ventricles by the potassium channel openers: cromakalim and RP49356. J Pharmacol Exp Ther 255:429–435
Osterrieder W (1988) Modefication of K+ conductance of heart cell membrane by BRL 34915. Naunyn Schmiedeberg’s Arch Pharmacol 33:93–97
McPherson GA, Angus JA (1990) Characterization of responses to cromakalim and pinacidil in smooth and cardiac muscle by use of selective antagonists. Br J Pharmacol 100:201–206
Tong X, Porter LM, Liu G, Chowdhury PD, Srivastava S, Pountney DJ, Yoshida H, Artman M, Fishman GI, Yu C, Iyer R, Morley GE, Gutstein DE, Coetzee WA (2006) Consequences of cardiac myocyte-specific ablation of KATP channels in transgenic mice expressing dominant negative Kir6 subunits. Am J Physiol Heart Circ Physiol 291:H543–H551
Moreau C, Jacques H, Prosa AL, Dhahan M, Vivaudou M (2000) The molecular basis of the specificity of action of KATP channel openers. EMBO J 19:6644–6651
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khan, Au., Gilani, A.H. Selective bronchodilatory effect of Rooibos tea (Aspalathus linearis) and its flavonoid, chrysoeriol. Eur J Nutr 45, 463–469 (2006). https://doi.org/10.1007/s00394-006-0620-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-006-0620-0