Abstract
Purpose
Multi-targeted tyrosine kinase inhibitors (MTKIs) are the standard in the treatment of metastatic renal cell carcinoma (mRCC). In spite their clinical activity, interaction with physiological functions has been shown. Here, we report on alterations of the bone mineral metabolism in patients with mRCC treated with MTKIs.
Methods
Fifty-nine patients with mRCC treated during April 2005 and September 2009 at our center were evaluated. Demographics, chemistry, parathyroid and renal function, bone metastasis and clinics were assessed, retrospectively. Parathyroid hormone (PTH), calcium and phosphate were either determined prior to, during or after cessation of MTKI therapy.
Results
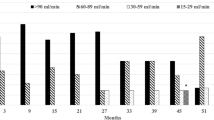
From evaluable patients, 90% (N = 53) received at least one MTKI treatment, 10% (N = 8) had evaluations without MTKI exposure. The mean PTH value prior to MTKI treatment was 49.4 (range (r):2.5–115), increased during the therapy to 121.2 (r:5–302) (P = 0.003) and returned to its basic values after MTKI cessation. In parallel, mean phosphate significantly decreased during the treatment from 1.10 (r = 0.66–1.59) to 0.87 (r = 0.48–1.45) (P < 0.001) and calcium showed a slight decrease (P = 0.039). PTH alterations were associated with clinical signs in some patients but not with bone metastasis or renal function. Univariate logistic regression analysis of pathologically elevated PTH levels revealed an association with MTKI treatment duration.
Conclusion
Even though the mechanism of bone mineral alteration remains elusive, the MTKI treatment is associated with a dysregulated parathyroid axis, which may have clinical implications in a number of patients. Furthermore, prospective trials are mandatory, and PTH monitoring should be considered in selected patients during MTKI treatment.


Similar content being viewed by others
Abbreviations
- AE(s):
-
Adverse event(s)
- CI 95%:
-
Confidence interval
- c-Kit:
-
Stem cell factor receptor
- CML:
-
Chronic myeloid leukemia
- eGFR:
-
Estimated glomerular filtration rate according to the Cockroft–Gault Formulation
- IFN:
-
Interferon
- mRCC:
-
Metastatic renal cell carcinoma
- MTKI(s):
-
Multi-target tyrosine kinase inhibitor(s)
- NSAID:
-
Nonsteroidal anti-inflammatory drugs
- r:
-
Range
- sHPT:
-
Secondary hyperparathyroidism
- PDGF-R:
-
Platelet-derived growth factor receptor
- TK(s):
-
Tyrosine kinase(s)
- VEGF-R:
-
Vascular endothelial growth factor receptor
References
Escudier B, Eisen T, Stadler WM et al (2007) Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 356:125–134
Motzer RJ, Hutson TE, Tomczak P et al (2007) Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 356:115–124
Cohen HT, McGovern FJ (2005) Renal-cell carcinoma. N Engl J Med 353:2477–2490
Lam JS, Leppert JT, Belldegrun AS, Figlin RA (2005) Novel approaches in the therapy of metastatic renal cell carcinoma. World J Urol 23(3):202–212
Brugarolas J (2007) Renal-cell carcinoma–molecular pathways and therapies. N Engl J Med 356:185–187
Stadler WM (2005) Targeted agents for the treatment of advanced renal cell carcinoma. Cancer 104(11):2323–2333
Ivanyi P, Winkler T, Ganser A et al (2008) Novel therapies in advanced renal cell carcinoma: management of adverse events from sorafenib and sunitinib. Dtsch Arztebl Int 105:232–237
Grunwald V, Soltau J, Ivanyi P et al (2009) Molecular targeted therapies for solid tumors: management of side effects. Onkologie 32:129–138
Telli ML, Witteles RM, Fisher GA, Srinivas S (2008) Cardiotoxicity associated with the cancer therapeutic agent sunitinib malate. Ann Oncol 19:1613–1618
Schmidinger M, Zielinski CC, Vogl UM et al (2008) Cardiac toxicity of sunitinib and sorafenib in patients with metastatic renal cell carcinoma. J Clin Oncol 26:5204–5212
Cohen MH, Johnson JR, Pazdur R (2005) U.S. Food and Drug Administration Drug Approval Summary: conversion of imatinib mesylate (STI571; Gleevec) tablets from accelerated approval to full approval. Clin Cancer Res 11:12–19
Kerkela R, Grazette L, Yacobi R et al (2006) Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nat Med 12:908–916
Isshiki I, Yamaguchi K, Okamoto S (2004) Interstitial pneumonitis during imatinib therapy. Br J Haematol 125:420
Berman E, Nicolaides M, Maki RG et al (2006) Altered bone and mineral metabolism in patients receiving imatinib mesylate. N Engl J Med 354:2006–2013
Grey A, O’Sullivan S, Reid IR, Browett P (2006) Imatinib mesylate, increased bone formation, and secondary hyperparathyroidism. N Engl J Med 355:2494–2495
Owen S, Hatfield A, Letvak L (2006) Imatinib and altered bone and mineral metabolism. N Engl J Med 355:627
Joensuu H, Reichardt P (2006) Imatinib and altered bone and mineral metabolism. N Engl J Med 355:628
O’Sullivan S, Horne A, Wattie D et al (2009) Decreased bone turnover despite persistent secondary hyperparathyroidism during prolonged treatment with imatinib. J Clin Endocrinol Metab 94:1131–1136
O’Sullivan S, Naot D, Callon K et al (2007) Imatinib promotes osteoblast differentiation by inhibiting PDGFR signaling and inhibits osteoclastogenesis by both direct and stromal cell-dependent mechanisms. J Bone Miner Res 22:1679–1689
Cockcroft DW, Gault MH (1976) Prediction of creatinine clearance from serum creatinine. Nephron 16:31–41
Fraser WD (2009) Hyperparathyroidism. Lancet 374:145–158
Seibel MJ (2006) Clinical application of biochemical markers of bone turnover. Arq Bras Endocinol Metab 40:603–619
Acknowledgments
We would like to thank our colleagues who were involved in the treatment and care of our patients: Martin Fenner and Steve Ehrlich.
Conflict of interest statement
PI, TW, AGr, CR, ASM, AG, declare no finical conflict. VG received honoria and travel grants from Novartis, Pfizer, Bayer Health Care and Roche Pharma.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ivanyi, P., Winkler, T., Großhennig, A. et al. Treatment with tyrosine kinase inhibitors in patients with metastatic renal cell carcinoma is associated with drug-induced hyperparathyroidism: a single center experience in 59 patients. World J Urol 28, 311–317 (2010). https://doi.org/10.1007/s00345-010-0558-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-010-0558-y