Abstract
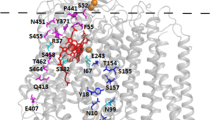
Cytochrome c oxidase accepts electrons from cytochrome c and transfers them to oxygen to form water. Electrons enter the complex through the CuA site, formed by two copper atoms bound to mitochondrially encoded subunit II. The effect of amino-acid alterations in one of the CuA ligands and in an amino acid adjacent to another of the ligands in the yeast enzyme is examined. Substitution of tyrosine for the CuA ligand, cysteine 221, completely abolishes enzyme activity. In addition, 19 independent revertants of this mutant yeast strain recover function by restoring the cysteine codon. Replacement of a non-conserved glycine at position 228 by valine, adjacent to the CuA-ligand histidine 229, virtually blocks enzyme activity. Activity is restored by inserting alanine or phenylalanine at position 228 or by amino-acid substitutions at nearby codons. Our results demonstrate that the CuA ligand appears to be essential for enzyme function while other residues in the copper-binding region are less functionally constrained.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 8 December 1998 / 22 May 1999
Rights and permissions
About this article
Cite this article
Mazourek, M., Torello, A. & Cameron, V. Analysis of strains of Saccharomyces cerevisiae with amino acid substitutions in the CuA-binding region of subunit II of cytochrome c oxidase. Curr Genet 36, 249–255 (1999). https://doi.org/10.1007/s002940050497
Issue Date:
DOI: https://doi.org/10.1007/s002940050497