Abstract
Purpose
Most ovarian cancers recur after first-line treatment. We studied the pharmacology, tolerability, and therapeutic potential of intraperitoneal (IP) topotecan, alone and with IP cisplatin.
Methods
Patients received IP topotecan 1.5 mg (flat dose) daily on days 1–5 (level 0) via IP catheter. Subsequent cohorts received IP cisplatin 50 mg/m2 on day 1 added to topotecan 1.5 mg on days 1–3 (level I), topotecan 1.25 mg on days 1–3 (level II), or topotecan 1.25 mg on days 1–5 (level III). Plasma and IP concentrations of total and lactone (E-ring closed) topotecan were measured on days 1 and 2 in cycles 1 and 2.
Results
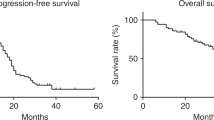
Sixteen patients (15 tubo-ovarian, 1 gastric cancers) were entered at levels 0 (3), I (4), II (4), or III (5). Dose-limiting neutropenias occurred in seven patients at dose levels I and III; grade 3 thrombocytopenia occurred in two at level III. Other toxicities included grade 1 hives in two, serum creatinine elevations in two, and Staphylococcus epidermidis and chemical peritonitis (one each). A median progression-free survival of 13 months was recorded among ovarian cancer patients who had minimal (6) or no residuum (3) after platinum-based induction; 5 are alive at 4 years. Topotecan’s AUC IP/AUC plasma ratios ranged from 13 to 119.
Conclusion
Topotecan IP for 3–5 days is tolerable; occasionally, myelosuppression is dose-limiting. Topotecan 1.25 mg (days 1–3) with IP cisplatin 50 mg/m2 (day 1) is a regimen suitable for consolidation in phase 3 trials.

Similar content being viewed by others
References
Dedrick RL, Myers CE, Bungay PM, DeVita VT Jr (1978) Pharmacokinetic rationale for peritoneal drug administration in the treatment of ovarian cancer. Cancer Treat Rep 62(1):1–11
Howell SB, Pfeifle CE, Wung We, Olshen RA (1983) Intraperitoneal cis-diamminedichloroplatinum with systemic thiosulfate protection. Cancer Res 43(3):1426–1431
Berek JS, Markman M, Stonebraker B et al (1999) Intraperitoneal interferon-alpha in residual ovarian carcinoma: a phase II gynecologic oncology group study. Gynecol Oncol 75(1):10–14
Morgan RJ Jr, Margolin K, Raschko J et al (1995) Phase I trial of carboplatin and infusional cyclosporin in advanced malignancy. J Clin Oncol 13(9):2238–2246
Morgan RJ Jr, Synold TW, Xi B et al (2007) Phase I trial of intraperitoneal gemcitabine in the treatment of advanced malignancies primarily confined to the peritoneal cavity. Clin Cancer Res 13:1232–1237
Morgan RJ Jr, Doroshow JH, Synold T et al (2003) Phase I trial of intraperitoneal docetaxel in the treatment of advanced malignancies primarily confined to the peritoneal cavity: dose-limiting toxicity and pharmacokinetics. Clin Cancer Res 9(16 Pt 1):5896–5901
Choi MK, Ahn BJ, Yim DS et al (2010) Phase I study of intraperitoneal irinotecan in patients with gastric adenocarcinoma with peritoneal seeding. Cancer Chemother Pharmacol Mar 6 [Epub ahead of print]
Ozols RF (1992) Intraperitoneal salvage chemotherapy in ovarian cancer: who is left to treat? Gynecol Oncol 45(1):1–2
Muggia FM, Alberts DS (1991) Intraperitoneal therapy in ovarian cancer: time’s not up (letter). J Clin Oncol 9(8):1510–1516
Muggia FM (1992) More on intraperitoneal therapy (letter). Ann Oncol 3:492
NCI Clinical Alert on Intraperitoneal Therapy of Ovarian Cancer (2006) Jan 4
Alberts DS, Liu PY, Hannigan EV et al (1996) Intraperitoneal cisplatin plus intravenous cyclophosphamide versus intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. N Engl J Med 335(26):1950–1955
Armstrong DK, Bundy B, Wenzel L et al (2006) Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med 354(1):34–43
Markman M, Bundy BN, Alberts DS et al (2001) Phase III trial of standard dose intravenous cisplatin plus paclitaxel versus moderately high-dose carboplatin followed by intravenous paclitaxel and intraperitoneal cisplatin in small volume stage III ovarian carcinoma: an intergroup study of the Gynecologic Oncology Group. J Clin Oncol 19:1001–1007
Jaaback K, Johnson N (2006) Intraperitoneal chemotherapy for the initial management of primary epithelial ovarian cancer. Cochrane Database Syst Rev (1):CD005340
Elit L, Oliver TK, Covens A, Kwon J, Fung MF, Hirte HW, Oza AM (2007) Intraperitoneal chemotherapy in the first-line treatment of women with stage III epithelial ovarian cancer: a systematic review with metaanalyses. Cancer 109(4):692–702
United States National Library of Medicine, National Institutes of Health (2005) NCI Issues Clinical Announcement for preferred method of treatment for Advanced Ovarian Cancer. http://www.nlm.nih.gov/databases/alerts/ovarian_ip_chemo.html accessed Jan 2010
Piccart M, Floquet A, Scarfone G et al (2003) Intraperitoneal cisplatin versus no further treatment: 8-year results of EORTC 55875, a randomized Phase III study in ovarian cancer patients with a pathologically complete remission after platinum-based intravenous chemotherapy. Int J Gynecol Cancer 13(suppl 2):196–203
McGuire WP (2009) Maintenance therapy for ovarian cancer: of Helsinki and Hippocrates. J Clin Oncol 27:4633–4634
Muggia FM, Liu PY, Alberts DS et al (1996) Intraperitoneal mitoxantrone or floxuridine: effects on time-to-failure and survival in patients with minimal residual ovarian cancer after second-look laparotomy. Gynecol Oncol 61:395–402
Muggia FM, Groshen S, Russell C et al (1993) Intraperitoneal carboplatin and etoposide for persistent epithelial ovarian cancer: analysis of results by prior sensitivity to platinum-based regimens. Gynecol Oncol 50:232–238
Barakat RR, Sabbatini P, Bhaskaran D et al (2002) Intraperitoneal chemotherapy for ovarian carcinoma: results of long-term follow up. J Clin Oncol 20:694–698
Lu MJ, Sorich J, Hazarika M et al (2003) Intraperitoneal therapy as consolidation for patients with ovarian cancer and negative reassessment after platinum-based chemotherapy. Hematol Oncol Clin North Am 17(4):969–975
Muggia F, Jeffers S, Muderspach L et al (1997) Phase I/II study of intraperitoneal floxuridine and platinum (cisplatin and/or carboplatin). Gynecol Oncol 66(2):290–294
Menczer J, Ben-Baruch G, Rizel S, Brenner H (1992) Intraperitoneal cisplatin chemotherapy in ovarian carcinoma patients who are clinically in complete remission. Gynecol Oncol 46(2):222–225
De Gramont A, Demuynck B, Louvet C et al (1992) Survival with intraperitoneal cisplatin in advanced ovarian cancer after second look laparotomy. Am J Clin Oncol 15(1):7–11
Tarraza HM Jr, Boyce CR, Smith WG et al (1993) Consolidation intraperitoneal chemotherapy in epithelial ovarian cancer patients following negative second look laparotomy. Gynecol Oncol 50(3):287–290
Barakat RR, Almadrones L, Venkatraman ES et al (1998) A phase II trial of intraperitoneal cisplatin and etoposide as consolidation therapy in patients with stage II-IV epithelial ovarian cancer following negative surgical assessment. Gynecol Oncol 69(1):17–22
Rothenberg ML, Liu PY, Braly PS et al (2003) Combined intraperitoneal and intravenous chemotherapy for women with optimally debulked ovarian cancer: results from an intergroup phase II trial. J Clin Oncol 21(7):1313–1319
Muggia F, Liebes L, Potmesil M et al (2000) Intraperitoneal topoisomerase-I inhibitors: preliminary findings with 9-aminocamptothecin. Ann N Y Acad Sci 922:178–187
Rowinsky EK, Kaufman SH, Baker SD et al (1996) Sequences of topotecan and cisplatin: phase I pharmacologic and in vitro studies to examine sequence dependence. J Clin Oncol 14:3074–3084
Hochster H, Liebes L, Speyer J et al (1997) Effect of prolonged topotecan infusion on topoisomerase 1 levels: a phase I and pharmacodynamic study. Clin Can Res 3:1245–1252
Hofstra LS, Bos AM, de Vries EG et al (2001) A phase I and pharmacokinetic study of intraperitoneal topotecan. Br J Cancer 85(11):1627–1633
Plaxe SC, Christen RD, O’Quigley J et al (1998) Phase I and pharmacokinetic study of intraperitoneal topotecan. Invest New Drugs 16:147–153
Muntz HG, Malpass TW, McGonigle KG et al (2008) Phase II study of intraperitoneal topotecan as consolidation chemotherapy in ovarian and primary peritoneal cancer. Cancer 113:490–496
De Placido S, Scambia G, Di Vagno G et al (2004) Topotecan compared with no therapy after response to surgery and carboplatin/paclitaxel in patients with ovarian cancer: Multicenter Italian Trials in Ovarian Cancer (MITO-1) randomized study. J Clin Oncol 22:2635–2642
Pfisterer J, Weber B, Reuss A et al (2006) Randomized phase III trial of topotecan following carboplatin and paclitaxel in first-line treatment of advanced ovarian cancer: a gynecologic cancer intergroup trial of the AGO-OVAR and GINECO. J Natl Cancer Inst 98:1036–1045
Shah DK, Shin BS, Veith J, Tóth K, Bernacki RJ, Balthasar JP (2009) Use of an anti-vascular endothelial growth factor antibody in a pharmacokinetic strategy to increase the efficacy of intraperitoneal chemotherapy. J Pharmacol Exp Ther 329:580–591
Acknowledgments
Supported in part by grants from Glaxo Smith-Kline, The Chemotherapy Foundation, and the GCRC M01 RR00096.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andreopoulou, E., Chen, T., Liebes, L. et al. Phase 1/pharmacology study of intraperitoneal topotecan alone and with cisplatin: potential for consolidation in ovarian cancer. Cancer Chemother Pharmacol 68, 457–463 (2011). https://doi.org/10.1007/s00280-010-1510-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-010-1510-y