Abstract
Purpose
To determine the clinical and hisotological efficacy and toxicities of induction chemotherapy with docetaxel (DOC) and nedaplatin (CDGP) for oral squamous cell carcinoma (OSCC) in the preoperative setting.
Methods
A total of 30 patients with locally advanced but operable OSCC were enrolled. Combination induction chemotherapy consisted of DOC 60 mg/m2 followed by CDGP 100 mg/m2.
Results
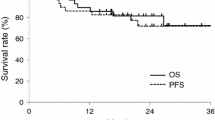
All patients received one cycle of chemotherapy. In the clinical assessment, ten patients achieved partial response for an overall response rate of 33.3% (95% CI, 16.4–50.2%). Histological assessment of surgical specimens showed an overall response rate of 56.6% (95% CI, 38.9–74.3%). Although severe neutropenia was observed in 90% of patients, only one patient (3.3%) experienced severe infection. Toxicities associated with this regimen did not interfere with planned radical surgery.
Conclusions
A single cycle of preoperative combination chemotherapy with DOC and CDGP showed moderate histological activity with an acceptable safety profile for the planned radical surgery. Further studies testing more cycles before surgery might be more appropriate.
Similar content being viewed by others
References
Verwij J, de Jong PC, de Mulder MPH, van der Broek P, Alexieva-Figusch J, van Putten WL et al (1989) Induction chemotherapy with cisplatin and continuous infusion 5-fluorouracil in locally far-advanced head and neck cancer. Am J Clin Oncol 12:420–424
Argiris A, Jayaram P, Pichardo D (2005) Revisiting induction chemotherapy for head and neck cancer. Oncology (Williston Park) 19:759–770
Pignon JP, Bourhis J, Domenge C, Designè L, on behalf of the MACH-NC Collaborative Group (2000) Chemotherapy added to locoregional treatment for head and neck squamous-cell carcinoma: three meta-analyses of updated individual data. Lancet 355:949–955
Al-Kourainy K, Kish J, Ensley J, Tapazoglou E, Jacobs J, Weaver A et al (1987) Achievement of superior survival for histologically negative versus histologically positive clinically complete responders to cisplatin combination in patients with locally advanced head and neck cancer. Cancer 59:233–238
Adjuvant chemotherapy for advanced head and neck squamous carcinoma (1987) Final report of the Head and Neck Contracts Program. Cancer 60:301–311
Ensley J, Crissman J, Kish J, Jacobs J, Weaver A, Kinzie J et al (1986) The impact of conventional morphologic analysis on response rates and survival in patients with advanced head and neck cancers treated initially with cisplatin-containing combination chemotherapy. Cancer 57:711–717
Hitt R, López-Pousa A, Martínez-Trufero J, Escrig V, Carles J, Rizo A et al (2005) Phase III study comparing cisplatin plus fluorouracil to paclitaxel, cisplatin, and fluorouracil induction chemotherapy followed by chemoradiotherapy in locally advanced head and neck cancer. J Clin Oncol 23:8636–8645
Posner MR, Hershock DM, Blajman CR, Mickiewicz E, Winquist E, Gorbounova V et al (2007) Cisplatin and fluorouracil alone or with docetaxel in head and neck cancer. N Engl J Med 357:1705–1715
Vermorken JB, Remenar E, van Herpen C, Gorlia T, Mesia R, Degardin M et al (2007) Cisplatin, fluorouracil, and docetaxel in unresectable head and neck cancer. N Engl J Med 357:1695–1704
Posner M, Vermorken JB (2008) Induction chemotherapy in the modern era of combined-modality therapy for locally advanced head and neck cancer. Semin Oncol 35:221–228
Kurita H, Yamamoto E, Nozaki S, Wada S, Furuta I, Kurashina K (2004) Multicenter phase I trial of induction chemotherapy with docetaxel and nedaplatin for oral squamous cell carcinoma. Oral Oncol 40:1000–1006
Sobin LH, Wittenkind CH (eds) (2002) TNM classification of malignant tumors, 6th edn. Wiley and Sons, New York
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L et al (2000) New guidelines to evaluate the response of treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States. National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Shimosato Y, Oboshi S, Baba K (1971) Histological evaluation of effects of radiotherapy and chemotherapy for carcinomas. Jpn J Clin Oncol 1:19–35
Dreyfuss AI, Clark JR, Norris CM, Rossi RM, Lucarini JW, Busse PM et al (1996) Docetaxel: an active drug for squamous cell carcinoma of the head and neck. J Clin Oncol 14:1672–1678
Catimel G, Verweij J, Mattijssen V, Hanauske A, Piccart M, Wanders J et al (1994) Docetaxal (Taxotere): an active drug for treatment of patients with advanced squamous cell carcinoma of the head and neck. Ann Oncol 5:533–537
Ebihara S, Fujii H, Sasaki Y et al (1997) A late phase II study of docetaxel (Taxotere) in patients with head and neck cancer (HNC). In: Proc Am Soc Clin Oncol 399a (Abstr 1425)
Couteau C, Leyvraz S, Outlid-Aissa D et al (1996) A phase II study of docetaxol in squamous cell carcinoma (SCC) of the head and neck. Proc Am Soc Clin Oncol 15:315 (Abst 889)
Sasaki Y, Shinkai T, Eguchi K, Tamura T, Ohe Y, Ohmori T et al (1991) Prediction of the antitumor activity of new platinum analogs based on their ex vivo pharmacodynamics as determined by bioassay. Cancer Chemother Pharmacol 27:263–270
Alberts DS, Fanta PT, Ruuing KL, Adair LP Jr, Garcia DJ, Liu-Stevens R et al (1997) In vitro phase II comparison of the cytotoxicity of a novel platinum analog, nedaplatin (254-S), with that of cisplatin and carboplatin against fresh, human ovarian cancers. Cancer Chemother Pharmacol 39:493–497
Tsukuda M, Yoshida T, Kubota A, Yanoma S, Watanabe S, Takahashi K et al (1988) Experimental study on the anti-tumor effects of CDDP derivatives against the head and neck squamous cell carcinoma cell lines. Gan To Kagaku Ryoho 15:2811–2814
Inuyama Y, Miyake H, Horiuchi M, Hayasaki K, Komiyama S, Ota K (1992) A late phase II clinical study of cis-diamine glycolato platinum, 254-S, for head and neck cancers. Gan To KagakuRyoho 19:871–877
Yamada H, Uchida N, Maekawa R, Yoshioka T (2001) Sequence-dependent antitumor efficacy of combination chemotherapy with nedaplatin, a newly developed platinum, and paclitaxel. Cancer Lett 172:17–25
Miyazaki A, Kobayashi J, Yamamoto T, Kido Y, Takemura K, Abe M et al (2008) A single-institute phase I/II trial combining nedaplatin dose escalation with a fixed dose of docetaxel for induction chemotherapy of oral squamous cell carcinoma. Oral Oncol 44:471–476
Schöffski P, Catimel G, Planting QST, Droz JP, Verweij J, Schrijvers D et al (1999) Docetaxel and cisplatin: an active regimen in patients with locally advanced, recurrent or metastatic squamous cell carcinoma of the head and neck. Ann Oncol 10:119–122
Specht L, Larsen SK, Hansen HS (2000) Phase II study of docetaxel and cisplatin in patients with recurrent or disseminated squamous-cell carcinoma of the head and neck. Ann Oncol 11:845–849
Caponigro F, Massa E, Manzione L, Rosati G, Rosati G, Biglietto M, De Lucia L et al (2001) Docetaxel and cisplatin in locally advanced or metastatic squamous-cell carcinoma of the head and neck: a phase II study of the southern Italy cooperating oncology group (SICOG). Ann Oncol 12:199–202
Glisson BS, Murphy BA, Frenette G, Khuri FR, Forastiere AA (2002) Phase II trial of docetaxel and cisplatin combination chemotherapy in patients with squamous cell carcinoma of the head and neck. J Clin Oncol 20:1593–1599
Paccagnella A, Orlando A, Marciori C, Zorat PL, Cavaniglia G, Sileni VC et al (1994) Phase III trial of initial chemotherapy in stage III or IV head and neck cancers: a study by the Gruppe di Studio sui Tumori della Testa e del Collo. J Natl Cancer Inst 86:265–272
Veterans Affairs Laryngeal Cancer Study Group V (1991) Induction chemotherapy plus radiation compared with surgery plus radiation in patients with advanced laryngeal cancer. N Engl J Med 324:1685–1689
Kish J, Ensley J, Jacobs J, Weaver A, Cummings G, Al-Sarraf M (1985) A randomized trial of cisplatin (CACP) + 5-fluorouracil (5-FU) infusion and CACP + 5-FU bolus for recurrent and advanced squamous cell carcinoma of the head and neck. Cancer 56:2740–2744
Lefebvre J, Chevalier D, Luboinske B, Kirkpatrick A, Collette L, Sahmoud T (1996) Larynx preservation in pyriform sinus cancer: Preliminary results of a European Organization for Research and Treatment of Cancer phase III trial. J Natl Cancer Inst 13:890–898
Vokes E, Wechselbaum R, Mick R, McEvilly JM, Haraf DJ, Panje WR (1992) Favorable long-term survival following induction chemotherapy with cisplatin, fluorouracil and leucovorin and concomitant chemoradiotherapy for locally advanced head and neck cancer. J Natl Cancer Inst 84:877–882
Gibson MK, Li Y, Murphy B, Hussain MH, DeConti RC, Ensley J et al (2005) Randomized phase III evaluation of cisplatin plus fluorouracil versus cisplatin plus paclitaxel in advanced head and neck cancer (E1395): an Intergroup trial of the Eastern Cooperative Oncology Group. J Clin Oncol 23:3562–3567
Fujta Y, Hiramatsu M, Kawai M, Sumiyoshi K, Nishimura H, Tanigawa N (2008) Evaluation of combined docetaxel and nedaplatin chemotherapy for recurrent esophageal cancer compared with conventional chemotherapy using cisplatin and 5-fluorouracil: a pilot study. Dis Esophagus 21:496–501
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kurita, H., Yamamoto, E., Nozaki, S. et al. Multicenter phase 2 study of induction chemotherapy with docetaxel and nedaplatin for oral squamous cell carcinoma. Cancer Chemother Pharmacol 65, 503–508 (2010). https://doi.org/10.1007/s00280-009-1056-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-009-1056-z