Abstract
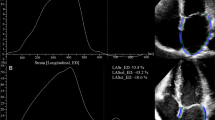
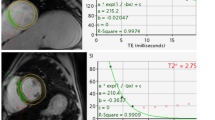
Cardiovascular involvement is a leading cause of mortality and morbidity in patients with inherited hemoglobinopathies, but it has not been adequately assessed in sickle β-thalassemia. We evaluated 115 sickle β-thalassemia patients, aged 34 ± 14 years, along with 50 healthy controls, by resting echocardiography. Patients with systolic left ventricular (LV) dysfunction or severe pulmonary hypertension (PHT) also underwent left and right cardiac catheterization and cardiac magnetic resonance imaging (CMR). Left and right chamber dimensions, LV mass, and cardiac index were significantly higher in patients compared to controls (p < 0.001 in most cases). Three patients (2.9%) had reduced LV ejection fraction (<55%); mean LV ejection fraction was significantly lower in patients (p < 0.001). Left and right ventricular systolic tissue Doppler indices and LV diastolic tissue Doppler indices were also impaired in patients. All three patients with systolic LV dysfunction had normal coronary arteries and mild myocardial iron load (CMR T2* values, 18–25 ms). Systolic pulmonary artery pressure was significantly higher in patients compared to controls (p = 0.002); PHT was present in 28 patients (27%), while severe PHT in three (2.9%). In three patients with severe PHT, only one had impaired LV ejection fraction and increased pulmonary wedge pressure. Overall, three patients (2.9%) had a history of heart failure, two with systolic LV dysfunction, and one with severe PHT. Cardiac involvement in sickle β-thalassemia concerns biventricular dilatation and dysfunction along with PHT, leading to congestive heart failure.

Similar content being viewed by others
References
Lane P (1996) Sickle cell disease. Ped Clin N Am 43:639–664. doi:10.1016/S0031-3955(05)70426-0
Loukopoulos D (1996) Current status of thalassemia and the sickle cell syndromes in Greece. Semin Hematol 33:76–86
Tsironi M, Aessopos A (2005) The heart in sickle cell disease. Acta Cardiol 60:589–598. doi:10.2143/AC.60.6.2004929
Aessopos A, Farmakis D, Deftereos S, Tsironi M, Tassiopoulos S, Moyssakis I et al (2005) Thalassemia heart disease: a comparative evaluation of thalassemia major and thalassemia intermedia. Chest 127:1523–1530. doi:10.1378/chest.127.5.1523
Moyssakis I, Tzanetea R, Tsaftaridis P, Rombos I, Papadopoulos DP, Kalotychou V et al (2005) Systolic and diastolic function in middle aged patients with sickle beta thalassaemia. An echocardiographic study. Postgrad Med J 81:711–714. doi:10.1136/pgmj.2004.031096
Aessopos A, Tsironi M, Vassiliadis I, Farmakis D, Fountos A, Voskaridou E et al (2001) Exercise-induced myocardial perfusion abnormalities in sickle beta-thalassemia: Tc-99m tetrofosmin gated SPECT imaging study. Am J Med 111:355–360. doi:10.1016/S0002-9343(01)00835-X
Aessopos A, Farmakis D, Tsironi M, Diamanti-Kandarakis E, Matzourani M, Fragodimiri C et al (2006) Endothelial function and arterial stiffness in sickle-thalassemia patients. Atherosclerosis 191:427–432. doi:10.1016/j.atherosclerosis.2006.04.015
Aessopos A, Farmakis D, Hatziliami A, Fragodimitri C, Karabatsos F, Joussef J et al (2004) Cardiac status in well-treated patients with thalassemia major. Eur J Haematol 73:359–366. doi:10.1111/j.1600-0609.2004.00304.x
Sahn DJ, De Maria A, Kisslo J, Weyman A (1978) The committee on M-mode standardization of the American Society of Echocardiography: results of a survey of echocardiographic measurements. Circulation 58:1072–1083
Schiller NB, Shah PM, Crawford M, DeMaria A, Devereux R, Feigenbaum H et al (1989) Recommendations for quantitation of the left ventricle by two-dimensional echocardiography: American Society of Echocardiography Committee on Standards Subcommittee. J Am Soc Echocardiogr 2:358–367
Feigenbaum H (1994) Echocardiographic evaluation of cardiac chambers. In: Feigenbaum H (ed) Echocardiography. 5th edn. Lea & Febiger, Philadelphia, pp 134–173
Devereux RB, Alonso DR, Lutas EM, Gottlieb GJ, Campo E, Sachs I et al (1986) Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol 57:450–458. doi:10.1016/0002-9149(86)90771-X
Lubien E, DeMaria A, Krishnaswamy P, Clopton P, Koon J, Kazanegra R et al (2002) Utility of B-natriuretic peptide in detecting diastolic dysfunction: comparison with Doppler velocity recordings. Circulation 105:595–601. doi:10.1161/hc0502.103010
Chan KL, Currie PJ, Seward JB, Hagler DJ, Mair DD, Tajik AJ (1987) Comparison of three Doppler ultrasound methods in the prediction of pulmonary artery disease. J Am Coll Cardiol 9:549–554
Aessopos A, Farmakis D, Taktikou H, Loukopoulos D (2000) Doppler-determined peak systolic tricuspid pressure gradient in persons with normal pulmonary function and tricuspid regurgitation. J Am Soc Echocardiogr 13:645–649. doi:10.1067/mje.2000.104535
Feigenbaum H, Armstrong W (2004) Feigenbaum’s echocardiography, 6th edn. Lippincott Williams & Wilkins, Philadelphia
Raman SV, Simonetti OP, Cataland SR, Kraut EH (2006) Myocardial ischemia and right ventricular dysfunction in adult patients with sickle cell disease. Haematologica 91:1329–1335
Voskaridou E, Douskou M, Terpos E, Papassotiriou I, Stamoulakatou A, Ourailidis A et al (2004) Magnetic resonance imaging in the evaluation of iron overload in patients with beta thalassaemia and sickle cell disease. Br J Haematol 126:736–742. doi:10.1111/j.1365-2141.2004.05104.x
Sachdev V, Machado RF, Shizukuda Y, Rao YN, Sidenko S, Ernst I et al (2007) Diastolic dysfunction is an independent risk factor for death in patients with sickle cell disease. J Am Coll Cardiol 49:472–479. doi:10.1016/j.jacc.2006.09.038
Murray JF, Dawson AM, Sherlock S (1958) Circulatory changes in chronic liver diseases. Am J Med 24:358–367. doi:10.1016/0002-9343(58)90322-X
Aessopos A, Farmakis D, Loukopoulos D (2002) Elastic tissue abnormalities resembling pseudoxanthoma elasticum in beta thalassemia and the sickling syndromes. Blood 99:30–35. doi:10.1182/blood.V99.1.30
Aessopos A, Farmakis D, Loukopoulos D (2002) Elastic tissue abnormalities in inherited haemolytic syndromes. Eur J Clin Invest 32:640–642. doi:10.1046/j.1365-2362.2002.01033.x
Aessopos A, Farmakis D (2004) Sickle cell disease: introducing a distinct elastic tissue defect associated with inherited hemolytic syndromes. In: Plasmar RL (ed) Focus on sickle cell research. Nova Biomedical Books, New York, pp 157–179
Aessopos A, Voskaridou E, Kavouklis E, Vassilopoulos G, Rombos Y, Gavriel L et al (1994) Angioid streaks in sickle-thalassemia. Am J Ophthalmol 117:589–592
Tsomi K, Karagiorga-Lagana M, Karabatsos F, Fragodimitri C, van Vliet-Konstantinidou C, Premetis E et al (2001) Arterial elastorrhexis in beta-thalassaemia intermedia, sickle cell thalassaemia and hereditary spherocytosis. Eur J Haematol 67:135–141. doi:10.1034/j.1600-0609.2001.5790349.x
Aessopos A, Deftereos S, Farmakis D, Corovesis C, Tassiopoulos S, Tsironi M et al (2004) Cardiovascular adaptation to chronic anemia in the elderly: an echocardiographic study. Clin Invest Med 27:265–273
Cheung YF, Chan GC, Ha SY (2002) Arterial stiffness and endothelial function in patients with beta-thalassemia major. Circulation 106:2561–2566. doi:10.1161/01.CIR.0000037225.92759.A7
Kato GJ, Martyr S, Blackwelder WC, Nichols JS, Coles WA, Hunter LA et al (2005) Levels of soluble endothelium-derived adhesion molecules in patients with sickle cell disease are associated with pulmonary hypertension, organ dysfunction, and mortality. Br J Haematol 130:943–953. doi:10.1111/j.1365-2141.2005.05701.x
Wood JC, Tyszka JM, Carson S, Nelson MD, Coates TD (2004) Myocardial iron loading in transfusion-dependent thalassemia and sickle cell disease. Blood 103:1934–1936. doi:10.1182/blood-2003-06-1919
Tsironi M, Polonifi K, Deftereos S, Farmakis D, Andriopoulos P, Moyssakis I et al (2005) Transfusional hemosiderosis and combined chelation therapy in sickle thalassemia. Eur J Haematol 75:355–358. doi:10.1111/j.1600-0609.2005.00528.x
Aessopos A, Samarcos M, Hadjinikolaou P, Vaiopoulos G, Skoumas V, Kolainis I et al (1996) Myocardial infarction with normal coronary arteries in S/beta thalassaemia patients. Eur Heart J 17:317–318
Rubler S, Fleischer RA (1967) Sickle cell states and cardiomyopathy. Sudden death due to pulmonary thrombosis and infarction. Am J Cardiol 19:867–873. doi:10.1016/0002-9149(67)90510-3
Lippman SM, Abergel RP, Ginzton LE, Uitto J, Tanaka KR, Miyamoto EK et al (1985) Mitral valve prolapse in sickle cell disease: manifestation of a generalized connective tissue disorder. Am J Hematol 19:1–12. doi:10.1002/ajh.2830190102
Gladwin MT, Sachdev V, Jison ML, Shizukuda Y, Plehn JF, Minter K et al (2004) Pulmonary hypertension as a risk factor for death in patients with sickle cell disease. N Engl J Med 350:886–895. doi:10.1056/NEJMoa035477
Voskaridou E, Tsetsos G, Tsoutsias A, Spyropoulou E, Christoulas D, Terpos E (2007) Pulmonary hypertension in patients with sickle cell/beta thalassemia: incidence and correlation with serum N-terminal pro-brain natriuretic peptide concentrations. Haematologica 92:738–743. doi:10.3324/haematol.11136
Ataga KI, Sood N, De Gent G, Kelly E, Henderson AG, Jones S et al (2004) Pulmonary hypertension in sickle cell disease. Am J Med 117:665–669. doi:10.1016/j.amjmed.2004.03.034
Derchi G, Forni GL, Formisano F, Cappellini MD, Galanello R, D’Ascola G et al (2005) Efficacy and safety of sildenafil in the treatment of severe pulmonary hypertension in patients with hemoglobinopathies. Haematologica 90:452–458
Hsu LL, Champion HC, Campbell-Lee SA, Bivalacqua TJ, Manci EA, Diwan BA et al (2007) Hemolysis in sickle cell mice causes pulmonary hypertension due to global impairment in nitric oxide bioavailability. Blood 109:3088–3098. doi:10.1182/blood-2006-06-029173
Morris CR, Kato GJ, Poljakovic M, Wang X, Blackwelder WC, Sachdev V et al (2005) Dysregulated arginine metabolism, hemolysis-associated pulmonary hypertension, and mortality in sickle cell disease. JAMA 294:81–90. doi:10.1001/jama.294.1.81
Kato GJ, McGowan V, Machado RF, Little JA, Taylor J 6th, Morris CR, Nichols JS, Wang X, Poljakovic M, Morris SM Jr, Gladwin MT (2006) Lactate dehydrogenase as a biomarker of hemolysis-associated nitric oxide resistance, priapism, leg ulceration, pulmonary hypertension, and death in patients with sickle cell disease. Blood 107:2279–2285. doi:10.1182/blood-2005-06-2373
Machado RF, Kyle Mack A, Martyr S, Barnett C, Macarthur P, Sachdev V et al (2007) Severity of pulmonary hypertension during vaso-occlusive pain crisis and exercise in patients with sickle cell disease. Br J Haematol 136:319–325. doi:10.1111/j.1365-2141.2006.06417.x
Steinberg MH, Rosenstock W, Coleman MB, Adams JG, Platica O, Cedeno M et al (1984) Effects of thalassemia and microcytosis on the hematologic and vaso-occlusive severity of sickle cell anemia. Blood 63:1353–1360
Aessopos A, Farmakis D, Karagiorga M, Voskaridou E, Loutradi A, Hatziliami A et al (2001) Cardiac involvement in thalassemia intermedia: a multicenter study. Blood 97:3411–3416. doi:10.1182/blood.V97.11.3411
Kato GJ, Onyekwere OC, Gladwin MT (2007) Pulmonary hypertension in sickle cell disease: relevance to children. Pediatr Hematol Oncol 24:159–170. doi:10.1080/08880010601185892
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aessopos, A., Farmakis, D., Trompoukis, C. et al. Cardiac involvement in sickle β-thalassemia. Ann Hematol 88, 557–564 (2009). https://doi.org/10.1007/s00277-008-0661-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-008-0661-y