Abstract
Purpose
Sentinel lymph node biopsy (SLNB) is a widely accepted procedure to accurately stage melanomas with a thickness ≥1 mm. The value of SLNB in thin melanomas is still controversial, especially because long-term observations of these patients are rare. The purpose of the current study was to identify the positive sentinel lymph node (SLN) ratio in low-risk patients with cutaneous melanoma (CM) of thickness less than 1 mm and its possible prognostic value, focusing on long-term follow-up data.
Methods
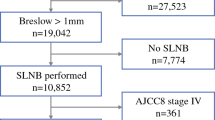
In a retrospective single-centre study performed at the Department of Dermatology and Allergy, University of Bonn, 121 patients who had received SLNB were identified out of 621 patients with a diagnosis of CM of <1.00 mm thickness presenting between September 2000 and February 2009 (mean follow-up time, 50.9 months).
Results
Of the 121 patients, 5 (4.1%) had a positive SLN. All positive SLNs were found in patients with a tumour thickness between 0.90 mm and 1.00 mm. There were no significant differences in the presence of positive SLNs according to Clark level and ulceration status (Clark levels II and III and no ulceration vs. Clark levels IV and V or ulceration), regression, gender or age. Disease-free survival was 100% in the SLN-positive patients. On the other hand, five SLN-negative patients (4.1%) developed disease progression. One of these five progressive patients showed recurrence in the former negative SLN basin (16.7% false-negative rate).
Conclusion
A positive SLN in thin melanomas is uncommon with a prevalence of 4.1% in our study population. We could not identify reliable clinicopathological risk factors which could predict results of SLNB in thin melanomas. Based on our results, SLNB may be considered in patients with a melanoma of thickness in the range 0.90–0.99 mm, because all SLN-positive patients belonged to this subgroup.

Similar content being viewed by others
References
Morton DL, Wen DR, Wong JH, et al. Technical details of intraoperative lymphatic mapping of early stage melanoma. Arch Surg. 1992;127:392–9.
Balch CM, Gershenwald JE, Soong S, Thompson JF, Atkins MB, Byrd DR, et al. Final version of 2009 AJCC Melanoma Staging and Classification. J Clin Oncol. 2009;27:6199–206.
Cascinelli N, Morabito A, Santinami M, MacKie RM, Belli F. Immediate or delayed dissection of regional nodes in patients with melanoma of the trunk: a randomised trial. WHO Melanoma Programme. Lancet. 1998;351:793–6.
Morton DL, Thompson JF, Cochran AJ, Mozzillo N, Elashoff R, Essner R, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med. 2006;355:1307–17.
Ross MI, Gershenwald JE. How should we view the results of the Multicenter Selective Lymphadenectomy Trial-1 (MSLT-1)? Ann Surg Oncol. 2008;15:670–3.
Gershenwald JE, Thompson W, Mansfield PF, Lee JE, Colome MI, Tseng CH, et al. Multi-institutional melanoma lymphatic mapping experience: the prognostic value of sentinel lymph node status in 612 stage I or II melanoma patients. J Clin Oncol. 1999;17:976–83.
Vuylsteke RJ, van Leeuwen PA, Statius Muller MG, Gietema HA, Kragt DR, Meijer S. Clinical outcome of stage I/II melanoma patients after selective sentinel lymph node dissection: long term follow-up results. J Clin Oncol. 2003;21:1057–65.
Agnese DM, Abdessalam SF, Burak WE, Magro CM, Pozderac RV, Walker MJ. Cost-effectiveness of sentinel lymph node biopsy in thin melanomas. Surgery. 2003;134:542–7.
Statius Muller MG, van Leeuwen PA, van Diest PJ, Vuylsteke RJ, Pijpers R, Meijer S. No indication for performing sentinel lymph node biopsy in melanoma patients with a Breslow thickness of less than 0.9 mm. Melanoma Res. 2001;11:303–7.
Balch CM, Murad TM, Soong SJ, Ingalls AL, Halpern NB, Maddox WA. A multifactorial analysis of melanoma: prognostic histopathologic factors comparing Clark’s and Breslow’s staging methods. Ann Surg. 1978;188:732–42.
Breslow A, Macht SD. Evaluation of prognosis in stage I cutaneous melanoma. Plast Reconstr Surg. 1978;61:342–6.
Gimotty PA, Guerry D, Ming ME, Elenitsas R, Xu X, Czerniecki B, et al. Thin primary cutaneous malignant melanoma: a prognostic tree for 10-year metastasis is more accurate than American Joint Committee on Cancer staging. J Clin Oncol. 2004;22:3668–76.
Gimotty PA, Botbyl J, Soong SJ, Guerry D. A population-based validation of the American Joint Committee on Cancer melanoma staging system. J Clin Oncol. 2005;23:8065–75.
Garbe C, Paul A, Kohler-Späth H, Ellwanger U, Stroebel W, Schwarz M, et al. Prospective evaluation of a follow-up schedule in cutaneous melanoma patients: recommendations for an effective follow-up strategy. J Clin Oncol. 2003;21:520–9.
Balch CM. Cutaneous melanoma: prognosis and treatment results worldwide. Semin Surg Oncol. 1992;8:400–14.
Carlson GW, Murray DR, Lyles RH, Staley CA, Hestley A, Cohen C. The amount of metastatic melanoma in a sentinel lymph node: does it have prognostic significance? Ann Surg Oncol. 2003;10:575–81.
Spanknebel K, Coit DG, Bieligk SC, Gonen M, Rosai J, Klimstra DS. Characterization of micrometastatic disease in melanoma sentinel lymph nodes by enhanced pathology: Recommendations for standardizing pathologic analysis. Am J Surg Pathol. 2005;29:305–17.
McKinnon JG, Yu XQ, McCarthy WH, Thompson JF. Prognosis for patients with thin cutaneous melanoma: long-term survival data from New South Wales Central Cancer Registry and the Sydney Melanoma Unit. Cancer. 2003;98:1223–31.
Schmid-Wendtner MH, Baumert J, Schmidt M, Konz B, Hölzel D, Plewig G, et al. Late metastases of cutaneous melanoma: an analysis of 31 patients. J Am Acad Dermatol. 2000;43:605–9.
Stitzenberg KB, Groben PA, Stern SL, Thomas NE, Hensing TA, Sansbury LB, et al. Indications for lymphatic mapping and sentinel lymphadenectomy in patients with thin melanoma (Breslow thickness < or = 1.0 mm). Ann Surg Oncol. 2004;11:900–6.
Oliveira Filho RS, Ferreira LM, Biasi LJ, Enokihara MM, Paiva GR, Wagner J. Vertical growth phase and positive sentinel node in thin melanoma. Braz J Med Biol Res. 2003;36:347–50.
Rousseau Jr DL, Ross MI, Johnson MM, Prieto VG, Lee JE, Mansfield PF, et al. Revised American Joint Committee on Cancer staging criteria accurately predict sentinel lymph node positivity in clinically node-negative melanoma patients. Ann Surg Oncol. 2003;10:569–74.
Puleo CA, Messina JL, Riker AI, Glass LF, Nelson C, Cruse CW, et al. Sentinel node biopsy for thin melanomas: which patient should be considered? Cancer Control. 2005;12:230–5.
Lens MB, Dawes M, Newton-Bishop JA, Goodacre T. Tumour thickness as a predictor of occult lymph node metastases in patients with stage I and II melanoma undergoing sentinel lymph node biopsy. Br J Surg. 2002;89:1223–7.
Wong SL, Brady MS, Coit DG, et al. Results of sentinel lymph node biopsy in thin melanoma. Presented at the 4th Biennial International Sentinel Node Congress, Los Angeles, CA, 3–6 December 2004.
Olah J, Gyulai R, Korom I, Varga E, Dobozy A. Tumor regression predicts higher risk of sentinel node involvement in thin cutaneous melanomas. Br J Dermatol. 2003;149:662–3.
Sekula-Gibbs SA, Shearer MA. Sentinel node biopsy should be offered in thin melanoma with mitotic rate greater than one. Dermatol Surg. 2011;37:1080–8.
Garbe C, Eigentler TK, Bauer J, Blödorn-Schlicht N, Fend F, Hantschke M, et al. Histopathological diagnostics of malignant melanoma in accordance with the recent AJCC classification 2009: Review of the literature and recommendations for general practice. J Dtsch Dermatol Ges. 2011;9:690–700.
Bleicher RJ, Essner R, Foshag IJ, Wanek LA, Morton DL. Role of sentinel lymphadenectomy in thin invasive cutaneous melanomas. J Clin Oncol. 2003;21:1326–31.
Jacobs IA, Chang CK, DasGupta TK, Salti GI. Role of sentinel lymph node biopsy in patients with thin primary melanoma. Ann Surg Oncol. 2003;10:558–61.
Warycha MA, Zakrzewski J, Ni Q, Shapiro RL, Berman RS, Pavlick AC, et al. Meta-analysis of sentinel lymph node positivity in thin melanoma (< or =1 mm). Cancer. 2009;115:869–79.
Zapas JL, Coley HC, Beam SL, Brown SD, Jablonski KA, Elias EG. The risk of regional lymph node metastases in patients with melanoma less than 1.0 mm thick: recommendation for sentinel lymph node biopsy. J Am Coll Surg. 2003;197:403–7.
Miller MW, Vetto JT, Monroe MM, Weerasinghe R, Andersen PE, Gross ND. False-negative sentinel lymph node biopsy in head and neck melanoma. Otolaryngol Head Neck Surg. 2011;145:606–11.
Gadd MA, Cosimi AB, Yu J, Duncan LM, Yu L, Flotte TJ, et al. Outcome of patients with melanoma and histological negative sentinel lymph nodes. Arch Surg. 1999;134:381–7.
Panasiti V, Devirgiliis V, Curzio M, Roberti V, Gobbi S, Rossi M, et al. Predictive factors for false negative sentinel lymph node in melanoma patients. Dermatol Surg. 2010;36:1521–8.
Statius Muller MG, Borgstein PJ, Pijpers R, van Leeuwen PA, van Diest PJ, Gupta A, et al. Reliability of the sentinel node procedure in melanoma patients: analysis of failures after long-term follow up. Ann Surg Oncol. 2000;7:461–8.
Gershenwald JE, Colome MI, Lee JE, Mansfield PF, Tseng C, Lee JJ, et al. Patterns of recurrence following a negative sentinel lymph node biopsy in 243 patients with stage I or II melanoma. J Clin Oncol. 1998;16:2253–60.
Wrightson WR, Wong SL, Edwards MJ, Chao C, Reintgen DS, Ross MI, et al. Complications associated with sentinel lymph node biopsy for melanoma. Ann Surg Oncol. 2003;10:676–80.
Acknowledgments
The study was supported by a grant from the German Cancer Aid (Program for the Development of Interdisciplinary Oncology Centers of Excellence in Germany), Bonn, Germany.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Torsten Hinz and Hojjat Ahmadzadehfar contributed equally to this work.
Rights and permissions
About this article
Cite this article
Hinz, T., Ahmadzadehfar, H., Wierzbicki, A. et al. Prognostic value of sentinel lymph node biopsy in 121 low-risk melanomas (tumour thickness <1.00 mm) on the basis of a long-term follow-up. Eur J Nucl Med Mol Imaging 39, 581–588 (2012). https://doi.org/10.1007/s00259-011-2009-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-011-2009-4