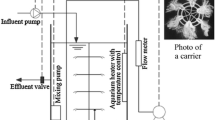
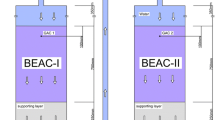
Abstract
Halophilic nitrifiers belonging to the genus Nitrosomonas and Nitrospira were enriched from seawater and marine sediment samples of the North Sea. The maximal ammonia oxidation rate (AOR) in batch enrichments with seawater was 15.1 mg N L−1 day−1. An intermediate nitrite accumulation was observed. Two fixed-bed reactors for continuous nitrification with either polyethylene/clay sinter lamellas (FBR A) or porous ceramic rings (FBR B) were run at two different ammonia concentrations, three different ammonia loading rates (ALRs), ± pH adjustment, and at an increased upflow velocity. A better overall nitrification without nitrite accumulation was observed in FBR B. However, FBR A revealed a higher AOR and nitrite oxidation rate of 6 and 7 mg N L−1 h−1, compared to FBR B with 5 and 5.9 mg N L−1 h−1, respectively. AORs in the FBRs were at least ten times higher than in suspended enrichment cultures. Whereas a shift within the ammonia-oxidizing population in the genus Nitrosomonas at the subspecies level occurred in FBR B with synthetic seawater at an increasing ALR and a decreasing pH, the nitrite oxidizing Nitrospira population apparently did not change.







Similar content being viewed by others
References
APHA (1995) Standard methods for the examination of water and wastewater. American Public Health Association, Washington
Antileo C, Aspe E, Urrutia H, Zaror C, Roeckel M (2002) Nitrifying biomass acclimation to high ammonia concentration. J Environ Eng 128:367–375
Ausubel FM, Brent R, Kingson RE, Moore DD, Seidman JG, Smith JA, Struhl K (1999) Short protocols in molecular biology. Wiley, New York
Campos JL, Mosquera-Collal A, Sānchez M, Méndez R, Lema JM (2002) Nitrification in saline wastewater with high ammonia concentration in an activated sludge unit. Water Res 36:2555–2560
Blackburne R, Vadivelu VM, Yuan Z, Keller J (2007) Kinetic characterization of an enriched Nitrospira culture with comparison to Nitrobacter. Water Res 41(14):3033–3042
Catalan MAB, Wang PC, Matsumura M (1997) Nitrification performance of marine nitrifyers immobilized in polyester and macroporous cellulose carriers. J Ferment Bioeng 84:563–571
Daims H, Nielsen JL, Nielsen PH, Schleifer KH, Wagner M (2001) In situ characterization of Nitrospira-like nitrite-oxidizing bacteria active in wastewater treatment plants. Appl Environ Microbiol 67:5273–5284
DEV (1983) Deutsche Einheitsverfahren zur Wasser-, Abwasser- und Schlammuntersuchung. Chemie, Weinheim
Dincer AR, Kargi F (2001) Performance of rotating biological disc system treating saline wastewater. Process Biochem 36:901–906
Fitch MW, Pearson N, Richard G, Burken JG (1998) Biological fixed-film system. Water Environ Res 70:495–518
Fontenot Q, Bonvillain C, Kilgen M, Boopathy R (2007) Effects of temperature, salinity, and carbon: nitrogen ratio on sequencing batch reactor treating shrimp aquaculture wastewater. Bioresource Technol 98:1700–1703
Fox P, Suidan MT, Bandy JT (1990) A comparison of media types in acetate fed expanded-bed anaerobic reactor. Water Res 24:827–835
Francis CA, Roberts KJ, Beman JM, Santoro AE, Oakley BB (2005) Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. Proc Natl Acad Sci USA 102:14683–14688
Gharsallah N, Khannous L, Souissi N, Nasri M (2002) Biological treatment of saline wastewaters from marine-products processing factories by a fixed-bed reactor. J Chem Technol Biotechnol 77:865–870
Gjaltema A, van der Marel N, van Loosdrecht MCM, Heijnen JJ (1997) Adhesion and biofilm development on suspended carriers in airlift reactor: hydrodynamic conditions versus surface characteristics. Biotechnol Bioeng 55:880–889
Huilinir M, Cristina Marti M, Aspe E, Roecke M (2008) Organic and nitrogenous matter effects on the denitrification of saline protein-rich effluents. Environ Technol 29:881–890
Intrasungkha N, Keller J, Blackall LL (1999) Biological nutrient removal efficiency in treatment of saline wastewater. Water Sci Technol 39(6):183–190
Koops HP, Böttcher B, Möller UC, Pommerening-Röser A, Stehr G (1991) Classification of eight new species of ammonia-oxidizing bacteria: Nitrosomonas communis sp. nov., Nitrosomonas ureae sp. nov., Nitrosomonas aestuarii sp. nov., Nitrosomonas marina sp. nov., Nitrosomonas nitrosa sp. nov., Nitrosomonas eutropha sp. nov., Nitrosomonas oligotropha sp. nov., and Nitrosomonas halophila sp. nov. J Gen Microbiol 137:1689–1699
Krüner G, Rosenthal H (1983) Efficiency of nitrification in trickling filters using different substrates. Aquacult Eng 2:49–67
Liu S, Yang F, Gong Z, Su Z (2008) Assessment of the positive effect of salinity on the nitrogen removal performance and microbial composition during the start-up of CANON process. Appl Microbiol Biotechnol 80:339–348
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acids techniques in bacterial systematics. Wiley, Chichester, pp 115–175
Mariangel L, Aspe E, Cristina Marti M, Roeckel M (2008) The effect of sodium chloride on the denitrification of saline fishery wastewater. Environ Technol 29:871–879
McCaig AE, Embley TM, Prosser JI (1994) Molecular analysis of enrichment cultures of marine ammonia oxidisers. FEMS Microbiol Lett 120(3):363–368
Mobarry BK, Wagner M, Urbain V, Rittmann BE, Stahl DA (1996) Phylogenetic probes for analyzing abundance and spatial organization of nitrifying bacteria. Appl Environ Microbiol 62:2156–2162
Moussa MS, Sumanasekera DU, Ibrahim SH, Lubberding HJ, Hooijmans CM, Gijzen HJ, van Loosdrecht MCM (2006) Long term effects of salt on the activity, population structure and floc characteristics in enriched bacterial cultures for nitrifiers. Water Res 40:1377–1388
Muyzer G, de Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Nijhof M, Bovendeur J (1990) Fixed film nitrification characteristic in sea water recirculation fish culture systems. Aquaculture 87:133–134
Nogueira R, Lazarova V, Manem J, Melo LF (1998) Influence of dissolved oxygen on the nitrification kinetics in a circulating bed biofilm reactor. Bioprocess Eng 19:441–449
Okabe S, Satoh H, Watanabe Y (1999) In situ analysis of nitrifying biofilms as determined by in situ hybridization and the use of microelectrodes. Appl Environ Microbiol 65:3182–3191
Omil F, Mendez R, Lema JM (1995) Anaerobic treatment of saline wastewaters under high sulphide and ammonia content. Bioresource Technol 54:269–278
Panswad T, Anan C (1999) Specific oxygen, ammonia, and nitrate uptake rates of a biological nutrient removal process treating elevated salinity wastewater. Bioresource Technol 70:237–243
Purkhold U, Wagner M, Timmermann G, Pommerening-Röser A, Koops HP (2003) 16S rRNA and amoA-based phylogeny of 12 novel betaproteo bacterial ammonia-oxidizing isolates: extension of the dataset and proposal of a new lineage within the nitrosomonads. Int J Syst Evol Microbiol 53:1485–1494
Rejish Kumar VJ, Achuthan C, Manju NJ, Philip R, Bright Singh IS (2009) Mass production of nitrifying bacterial consortia for the rapid establishment of nitrification in saline recirculating aquaculture systems. World J Microb Biot 25:407–414
Rosa MF, Furtado AAL, Albuquerque RT, Leite SGF, Medronho RA (1998) Biofilm development and ammonia removal in the nitrification of a saline wastewater. Bioresource Technol 65:135–138
Schenk H, Hegemann W (1995) Nitrification inhibition by high salinity concentrations in the aerobic biological treatment of tannery wastewater. Wasser/Abwasser 136:465–470
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Tsuneda S, Mikami M, Kimochi Y, Hirata A (2005) Effect of salinity on nitrous oxide emission in the biological nitrogen removal process for industrial wastewater. J Hazard Mater 119:93–98
Uygur A, Kargi F (2004) Salt inhibition on biological nutrient removal from saline wastewater in a sequencing batch reactor. Enzyme Microb Tech 34:313–318
Verran J, Lees G, Shakespeare AP (1991) The effect of surface roughness on the adhesion of Candida albicans to acrylic. Biofouling 3:183–192
Villaverde S, Garcia-Encina PA, Fdz-Polanco F (1997) Influence of pH over nitrifying biofilm activity in submerged biofilters. Water Res 31:1180–1186
Wagner M, Rath G, Koops HP, Flood J, Amann R (1996) In situ analysis of nitrifying bacteria in sewage treatment plants. WatSciTechn 34:237–244
Watson SW, Bock E, Valois FW, Waterbury JB, Schlosser U (1986) Nitrospira marina gen. nov. sp. nov.: a chemolithotrophic nitrite-oxidizing bacterium. Arch Microbiol 144:1–7
Windey K, Bo ID, Verstraete W (2005) Oxygen-limited autotrophic nitrification–denitrification (OLAND) in a rotating biological contactor treating high-salinity wastewater. Water Res 39:4512–4520
Wu G, Guan Y, Zhan X (2008) Effect of salinity on the activity, settling and microbial community of activated sludge in sequencing batch reactors treating synthetic saline wastewater. Water Sci Technol 58(2):351–358
Zhu S, Chen S (2001) Impacts of Reynolds number on nitrification biofilm kinetics. Aquacult Eng 24:213–229
Acknowledgment
This work was supported by a grant from the German Academic Exchange Service DAAD to Mr. Sudarno.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sudarno, U., Bathe, S., Winter, J. et al. Nitrification in fixed-bed reactors treating saline wastewater. Appl Microbiol Biotechnol 85, 2017–2030 (2010). https://doi.org/10.1007/s00253-009-2301-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2301-4