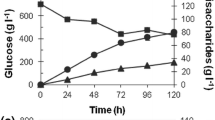
Abstract
The α-glucosidase inhibitor acarbose, O-{4,6-dideoxy-4[1s-(1,4,6/5)-4,5,6-trihydroxy-3-hydroxymethyl-2-cyclohexen-1-yl]-amino-α-d-glucopyranosyl}-(1→4)-O-α-d-glucopyranosyl-(1→4)-d-glucopyranose, is produced in large-scale fermentation by the use of strains derived from Actinoplanes sp. SE50. It has been used since 1990 in many countries in the therapy of diabetes type II, in order to enable patients to better control blood sugar contents while living with starch-containing diets. Thus, it is one of the latest successful products of bacterial secondary metabolism to be introduced into the pharmaceutical world market. Cultures of Actinoplanes sp. also produce various other acarbose-like components, of which component C is hard to separate during downstream processing, which is one of the most modern work-up processes developed to date. The physiology, genetics and enzymology of acarbose biosynthesis and metabolism in the producer have been studied to some extent, leading to the proposal of a new pathway and metabolic cycle, the “carbophore”. These data could give clues for further biotechnological developments, such as the suppression of side-products, enzymological or biocombinatorial production of new metabolites and the engineering of production rates via genetic regulation in future.






Similar content being viewed by others
References
Ahlert J, Distler J, Mansouri K, Piepersberg W (1997) Identification of the gene, stsC, encoding the l-glutamine:scyllo-inosose aminotransferase from streptomycin-producing streptomycetes. Arch Microbiol 168:102–113
Ahr HJ, Boberg M, Krause HP, Maul W, Mueller FO, Ploschke HJ, Weber H, Wuensche C (1989) Pharmacokinetics of acarbose. Part I: absorption, concentration in plasma, metabolism and excretion after single administration of [14C]acarbose to rats, dogs and man. Arzneimittelforschung 39:1254–1260
Apeler H, Wehlmann H, Piepersberg W, Diaz-Guardamino P-M, Jarling M, Thomas H, Wehmeier U (2001) Neue Enzyme in der Acarbose-Synthese und deren Verwendung. German patent DE-OS 10021667
Asano N, Kameda Y, Matsui K, Horii S, Fukase H(1990) Validamycin H, a new pseudo-tetrasaccharide antibiotic. J Antibiot (Tokyo) 43:1039–1041
Bentley SD, Chater KF, Cerdeno-Tarraga AM, Challis GL, Thomson NR, James KD, Harris DE, Quail MA, Kieser H, Harper D, Bateman A, Brown S, Chandra G, Chen CW, Collins M, Cronin A, Fraser A, Goble A, Hidalgo J, Hornsby T, Howarth S, Huang CH, Kieser T, Larke L, Murphy L, Oliver K, O’Neil S, Rabbinowitsch E, Rajandream MA, Rutherford K, Rutter S, Seeger K, Saunders D, Sharp S, Squares R, Squares S, Taylor K, Warren T, Wietzorrek A, Woodward J, Barrell BG, Parkhill J, Hopwood DA (2002) Complete genome sequence of the model actinomycete Streptomyces coelicolor A3(2). Nature 417:141–147
Beunink J, Schedel; M, Steiner U (1997) Osmotically controlled fermentation process for the preparation of acarbose. German patent DE 19637591 (US patent 6,130,072)
Bischoff H, Ahr HJ, Schmidt D, Stoltefuss J (1994) Acarbose—ein neues Wirkprinzip in der Diabetestherapie. Nachr Chem Tech Lab 42:1119–1128
Boos W, Lucht JM (1996) Periplasmic binding protein-dependent ABC transporters. In: Neidhardt FC (ed) Escherichia coli and Salmonella, 2nd edn. ASM Press, Washington D.C., pp 1175–1209
Brayer GD, Sidhu G, Maurus R, Rydberg EH, Braun C,| Wang Y, Nguyen NT, Overall CM, Withers SG (2000) Subsite mapping of the human pancreatic r-amylase active site through structural, kinetic, and mutagenesis techniques. Biochemistry 39:4778–4791
Brunkhorst C, Andersen C, Schneider E (1999) Acarbose, a pseudooligosaccharide, is transported but not metabolized by the maltose-maltodextrin system of Escherichia coli. J Bacteriol 18:2612–2619
Crueger A, Piepersberg W, Distler J, Stratmann A (1996) Acarbose biosynthesis genes from Actinoplanes sp., process for the isolation thereof and the use thereof. German patent DE 19507214.6 (US patent 5,753,501)
Crueger A, Dellweg H-G, Lenz J, Schroder, Pape H, Goeke K, Schaper B, Hemker M, Piepersberg W, Distler J, Stratmann A (1997) Processes for preparing acarviosyl transferase and for using it in the conversion of acarbose homologues into acarbose, for the preparation of acarbose homologues. German patents DE 19611252, 19625269 (US patent 5,989,882)
Decker H (1997) Isolation of the biosynthesis genes for pseudo-oligosaccharides from Streptomyces glaucescens GLA.O, and their use. German patent DE 19622783 (US patent 6,306,627)
Decker H, Gaisser S, Pelzer S, Schneider P, Westrich L, Wohlleben W, Bechthold A (1996) A general approach for cloning and characterizing dNDP-glucose dehydratase genes from actinomycetes. FEMS Microbiol Lett 141:195–201
Degwert U, Hulst R van, Pape H, Herrold RE, Beale JM, Keller PJ, Lee JP, Floss HG (1987) Studies on the biosynthesis of the alpha-glucosidase inhibitor acarbose: valienamine, am-C7N unit not derived from the shikimate pathway. J Antibiot (Tokyo) 40:855–861
Demain AL, Somkuti GA, Hunter-Creva JC, Rossmoore HW (1989) Novel microbial products for medicine and agriculture. Elsevier Science, Amsterdam
Díaz-Guardamino Uribe PM (2000) Untersuchungen zum Einbau des Stickstoffes in der Acarviose-Einheit der Acarbose bei Actinoplanes sp. 50/110: die Aminotransferase AcbV. PhD thesis, Bergische Universität Gesamthochschule, Wuppertal
Dong H, Mahmud T, Tornus I, Lee S, Floss HG (2001 ) Biosynthesis of the validamycins: identification of intermediates in the biosynthesis of validamycin A by Streptomyces hygroscopicus var. limoneus. J Am Chem Soc 123:2733–2742
Drepper A, Pape H (1996) Acarbose 7-phosphotransferase from Actinoplanes sp.: purification, properties, and possible physiological function. J Antibiot (Tokyo) 49:664–668
Drepper A, Peitzmann R, Pape H (1996) Maltokinase (ATP:maltose 1-phosphotransferase) from Actinoplanes sp.: demonstration of enzyme activity and characterization of the reaction product. FEBS Lett 388:177–179
Frommer W, Puls W, Schaefer D, Schmidt D (1975) Glycoside-hydrolase enzyme inhibitors. German patent DE 2064092 (US patent 3,876,766)
Frommer W, Junge B, Keup U, Mueller L, Schmidt D (1977a) Amino sugar derivatives. German patent DE 2347782 (US patent 4,062,950)
Frommer W, Puls W, Schmidt D (1977b) Process for the production of a saccharase inhibitor. German patent DE 2209834 (US patent 4,019,960)
Frommer W, Junge B, Mueller L, Schmidt D, Truscheit E (1979) Neue Enzyminhibitoren aus Mikroorganismen. Planta Med 35:195–217
Gilles C, Astier J-P, Marchis-Mouren G, Cambillau C, Payan F (1996) Crystal structure of pig pancreatic α-amylase isoenzyme II, in complex with the carbohydrate inhibitor acarbose. Eur J Biochem 238:561–569
Goeke K, Drepper A, Pape H (1996) Formation of acarbose phosphate by a cell-free extract from the acarbose producer Actinoplanes sp. J Antibiot (Tokyo) 49:661–663
He XM, Liu HW (2002) Formation of unusual sugars: mechanistic studies and biosynthetic applications. Annu Rev Biochem 71:701–754
Hemker M (1997) Pseudooligosaccharide und Staerkestoffwechsel bei Actinoplanes sp. PhD thesis, Westfaelische Wilhelms-Universitaet, Muenster
Hemker M, Stratmann A, Goeke K, Schroder W, Lenz J, Piepersberg W, Pape H (2001). Identification, cloning, expression, and characterization of the extracellular acarbose-modifying glycosyltransferase, AcbD, from Actinoplanes sp. strain SE50. J Bacteriol 183:4484–4492
Hurtubise Y, Shareck F, Kluepfel D, Morosoli R (1995) A cellulase/xylanase-negative mutant of Streptomyces lividans 1326 defective in cellobiose and xylobiose uptake is mutated in a gene encoding a protein homologous to ATP-binding proteins. Mol Microbiol 17:367–377
Ikeda H, Ishikawa J, Hanamoto A, Shinose M, Kikuchi H, Shiba T, Sakaki Y, Hattori M, Omura S (2003) Complete genome sequence and comparative analysis of the industrial microorganism Streptomyces avermitilis. Nat Biotechnol 21:526–531
Itoh J, Omoto S, Shomura T, Ogino H, Iwamatsu K, Inouye S, Hidaka H (1981) Oligostatins, new antibiotics with amylase inhibitory activity. I. Production, isolation and characterization. J Antibiot (Tokyo) 34:1424–1428
Iwasa T, Yamamoto H, Shibata M (1970) Studies on validamycins, new antibiotics. I. Streptomyces hygroscopicus var. limoneus nov. var., validamycin-producing organism. J Antibiot (Tokyo) 23:595–602
Kim YH, Kwon TK, Park S, Seo HS, Cheong JJ, Kim CH, Kim JK, Lee JS, Choi YD (2000) Trehalose synthesis by sequential reactions of recombinant maltooligosyltrehalose synthase and maltooligosyltrehalose trehalohydrolase from Brevibacterium helvolum. Appl Environ Microbiol. 66:4620–4624
Lange PM, Rauenbusch E (1986) Polymers for the purification of acarbose. German patent DE 3439008 (US patent 4,767,850)
Lee S, Egelkrout E (1998) Biosynthetic studies on the alpha-glucosidase inhibitor acarbose in Actinoplanes sp.: glutamate is the primary source of the nitrogen in acarbose. J Antibiot (Tokyo) 51:225–227
Lee S, Sauerbrei B, Niggemann J, Egelkrout E (1997) Biosynthetic studies on the alpha-glucosidase inhibitor acarbose in Actinoplanes sp.: source of the maltose unit. J Antibiot (Tokyo) 50:954–960
Liu H-W, Thorson JS (1994) Pathways and mechanisms in the biogenesis of novel deoxysugars by bacteria. Annu Rev Microbiol 48:223–256
Machius M, Vértesy L, Huber R, Wiegand G (1996) Carbohydrate and protein-based inhibitors of porcine pancreatic α-amylase: structure analysis and comparison of their binding characteristics. J Mol Biol 260:409–421
Mahmud T (2003) The C7N aminocyclitol family of natural products. Nat Prod Rep 20:137–166
Mahmud T, Tornus I, Engelkrout E, Wolf E, Uy C, Floss HG, Lee S (1999) Biosynthetic studies on the α-glucosidase inhibitor acarbose in Actinoplanes sp.: 2-epi-5-epi-valiolone is the direct precursor of the valienamine moiety. J Am Chem Soc 121:6973–6983
Mahmud T, Xu J, Choi YU (2001) Synthesis of 5-epi-[6-(2)H(2)]valiolone and stereospecifically monodeuterated 5-epi-valiolones: exploring the steric course of 5-epi-valiolone dehydratase in validamycin A biosynthesis. J Org Chem 66:5066–5073
Mueller L (1989) Chemistry, biochemistry and therapeutic potential of microbial a-glucosidase inhibitors. In: Demain AL, Somkuti GA, Hunter-Creva JC, Rossmoore HW (eds) Novel microbial products for medicine and agriculture. Elsevier Science, Amsterdam, pp 109–116
Murao S (1975) Microbial amylase inhibitor and preparation thereof with the use of Streptomyces diasticus var. amylostaticus. Japanese patent 49-29646 (US patent 4,010,258)
Murao S, Ohyama K (1975) New amylase inhibitor (S-1) from Streptomyces diasticus var. amylostaticus no. 2476. Agric Biol Chem 39:2271–2273
Namiki S, Kangouri K, Nagate T, Hara H, Sugita K, Omura S (1982) Studies on the alpha-glucoside hydrolase inhibitor, adiposin. I. Isolation and physicochemical properties. J Antibiot (Tokyo) 35:1234–1236
Nguyen J, Francou F, Virolle MJ, Guérineau M (1997) Amylase and chitinase genes in Streptomyces lividans are regulated by reg1, a pleiotropic regulatory gene. J Bacteriol 179:6383–6390
Ota Y, Tamegai H, Kudo F, Kuriki H, Koike-Takeshita A, Eguchi T, Kakinuma K (2000) Butirosin-biosynthetic gene cluster from Bacillus circulans. J Antibiot 53:1158–1167
Piepersberg W (1994) Pathway engineering in secondary metabolite-producing actinomycetes. Crit Rev Biotechnol 14:251–285
Piepersberg W (1997) Molecular biology, biochemistry, and fermentation of aminoglycoside antibiotics. In: Strohl WR, (ed) Biotechnology of industrial antibiotics, 2nd edn. Marcel–Dekker, New York, pp 81–163
Piepersberg W (2001) Endogenous antimicrobial molecules: an ecological perspective. In: Sussman M (ed) Molecular medical microbiology. Academic Press, London, pp 561–583
Piepersberg W, Distler J (1997) Aminoglycosides and sugar components in other secondary metabolites. In: Rehm H-J, Reed G, Pühler A, Stadler P (eds) Biotechnology. (Products of secondary metabolism, vol 7) VCH, Weinheim, pp 397–488
Piepersberg W, Diaz-Guardamino PM, Stratmann A, Thomas H, Wehmeier UF, Zhang CS (2002) Recent developments in the biosynthesis and regulation of aminoglycosides. In: Fierro F, Martín JF (eds) Microbial secondary metabolites: biosynthesis, genetics and regulation. Research Signpost, Kerala, pp 1–26
Puls W, Keup U (1973) Influence of an α-amylase inhibitor (BAY d7791) on blood glucose, serum insulin and NEFA in starch loading test in rats, dogs and man. Diabetologia 9:97
Puls W, Keup U, Krause HP, Thomas G, Holtmeier F (1977) Glucosidase inhibition: a new approach to the treatment of diabetes, obesity and hyperlipoproteinaemia. Naturwissenschaften 64:536–537
Qian M, Haser R, Buisson G, Duée E, Payan F (1994) The active center of a mammalian α-amylase. Structure of the complex of a pancreatic α-amylase with a carbohydrate inhibitor refined to 2.2-Å resolution. Biochemistry 33:6284–6294
Rauenbusch E (1987) Highly pure acarbose. German patent DE 3543999 (US patent 4,904,769)
Rauenbusch E, Schmidt D (1978) Verfahren zur Isolierung von (O{4,6-Dideoxy-4[[1s-(1,4,6/5)-4,5,6-trihydroxy-3-hydroxymethyl-2-cyclohexen-1-yl]-amino]-α-d-glucopyranosyl}-(1→4)-O-α-d-glucopyranosyl-(1→4)-d-glucopyranose) aus Kulturbrühen. German patent DE 2719912 (Process for isolating glucopyranose compound from culture broths; US patent 4,174,439)
Rehm H-J, Reed G, Pühler A, Stadler P (1997) Products of secondary metabolism, 2nd edn. (Biotechnology, vol 7) VCH, Weinheim
Rinehart KL Jr, Snyder WC, Staley AL, Lau RCM (1992) Biosynthetic studies on antibiotics. In: Petroski RJ, McCormick SP (eds) Secondary-metabolite biosynthesis and metabolism. Plenum Press, New York, pp 41–60
Russell R.B, Aduse-Opuko J, Sutcliffe IC, Tao L, Ferretti JJ (1992) A binding protein-dependent transport system in Streptococcus mutans responsible for multiple sugar metabolism. J Biol Chem 267:4631–4637
Stockmann M, Piepersberg W (1992) Gene probes for the detection of 6-deoxyhexose metabolism in secondary metabolite-producing streptomycetes. FEMS Microbiol Lett 90:185–190
Stolpe T (2001) Zur Bildung von Pseudooligosaccharidyl-Trehalosen durch Actinoplanes sp. PhD thesis, Westfaelische Wilhelms-Universitaet, Muenster
Stratmann A (1997) Identifizierung eines Acarbose-Biosynthesegenclusters in Actinoplanes sp. und Charakterisierung ausgewählter Enzyme des Acarbose-Stoffwechsels. PhD thesis, Bergische Universitaet, Wuppertal
Stratmann A, Mahmud T, Lee S, Distler J, Floss HG, Piepersberg W (1999) The AcbC protein from Actinoplanes species is a c7-cyclitol synthase related to 3-dehydroquinate synthases and is involved in the biosynthesis of the α-glucosidase inhibitor acarbose. J Biol Chem 274:10889–10896
Strokopytov B, Penninga D, Rozeboom HJ, Kalk KH, Dijkhuizen L, Dijkstra BW (1995) X-ray structure of cyclodextrin glycosyltransferase complexed with acarbose. Implications for the catalytic mechanism of glycosidases. Biochemistry 34:2234–2240
Takeda H, Nakagawa Y, Kiuchi A (1983) Novel amino oligosaccharide derivative. Japanese patent JP 58-172400
Tamegai H, Nango E, Kuwahara M, Yamamoto H, Ota Y, Kuriki H, Eguchi T, Kakinuma K (2002) Identification of l-glutamine: 2-deoxy-scyllo-inosose aminotransferase required for the biosynthesis of butirosin in Bacillus circulans. J Antibiot 55:707–714
Tatusov RL, Natale DA, Garkavtsev IV, Tatusova TA, Shankavaram UT, Rao BS, Kiryutin B, Galperin MY, Fedorova ND, Koonin EV (2001) The COG database: new developments in phylogenetic classification of proteins from complete genomes. Nucleic Acids Res 29:22–28
Taylor RH, Barker HM (1983) Alpha-amylase inhibitors. Lancet 1:1228–1229
Thomas H (2001) Acarbose Metabolismus in Actinoplanes sp. SE50/110. PhD, Bergische Universitaet, Wuppertal
Truscheit E, Frommer W, Junge B, Müller L, Schmidt D, Wingeder W (1981) Chemistry and biochemistry of bacterial alpha-glucosidase inhibitors. Angew Chem Int Ed Eng 20:744–761
Truscheit E, Junge B, Mueller L, Puls W, Schmidt D (1988) Microbial alpha-glucosidase inhibitors: chemistry, biochemistry and therapeutic potential. Prog Clin Biochem Med 7:17
Virolle MJ, Gagnat J (1994) Sequences involved in growth-phase-dependent expression and glucose repression of a Streptomyces α-amylase gene. Microbiology 140:1059–1067
Wiegand G, Epp O, Huber R (1995) The crystal structure of porcine pancreatic alpha-amylase in complex with the microbial inhibitor tendamistat. J Mol Biol 247:99–110
Yokose K, Ogawa K, Sano T, Watanabe K, Maruyama HB, Suhara Y (1983) New alpha-amylase inhibitor, trestatins. I. Isolation, characterization and biological activities of trestatins A, B and C. J Antibiot 36:1157–1165
Zhang CS, Stratmann A, Block O, Brueckner R, Podeschwa M, Altenbach HJ, Wehmeier UF, Piepersberg W (2002) Biosynthesis of the C(7)-cyclitol moiety of acarbose in Actinoplanes species SE50/110. 7-O-phosphorylation of the initial cyclitol precursor leads to proposal of a new biosynthetic pathway. J Biol Chem 277:22853–22862
Zhang CS, Podeschwa M, Altenbach HJ, Piepersberg W, Wehmeier UF (2003a) The acarbose-biosynthetic enzyme AcbO from Actinoplanes sp. SE 50/110 is a 2-epi-5-epi-valiolone-7-phosphate 2-epimerase. FEBS Lett 540:47–52
Zhang CS, Podeschwa M, Block O, Altenbach HJ, Piepersberg W, Wehmeier UF (2003b) Identification of a 1-epi-valienol 7-kinase activity in the producer of acarbose, Actinoplanes sp. SE50/110. FEBS Lett 540:53–57
Acknowledgements
We acknowledge excellent cooperation with the groups of A. Crueger and H. Wehlmann (Bayer AG), H. Pape (University of Muenster), H.G. Floss (University of Seattle) and H.J. Altenbach (BU Wuppertal) in this project. The work in the laboratories of the authors was supported by grants from Bayer AG (Leverkusen, Germany) and the German Ministry of Education and Research (BMBF grant BEO22-0310815).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wehmeier, U.F., Piepersberg, W. Biotechnology and molecular biology of the α-glucosidase inhibitor acarbose. Appl Microbiol Biotechnol 63, 613–625 (2004). https://doi.org/10.1007/s00253-003-1477-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-003-1477-2