Abstract
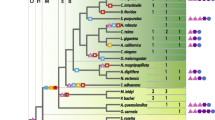
Adenosine deaminase (ADA) is a well-characterized enzyme involved in the depletion of adenosine levels. A group of proteins with similarity to ADA, the adenosine deaminase-related growth factors (ADGF; known as CECR1 in vertebrates), has been described recently in various organisms. We have determined the phylogenetic relationships of various gene products with significant amino acid similarity to ADA using parsimony and Bayesian methods, and discovered a novel paralogue, termed ADA-like (ADAL). The ADGF proteins share a novel amino acid motif, “MPKG,” within which the proline and lysine residues are also conserved in the ADAL and ADA subfamilies. The significance of this new domain is unknown, but it is located just upstream of two ADA catalytic residues, of which all eight are conserved among the ADGF and ADAL proteins. This conservation suggests that ADGF and ADAL may share the same catalytic function as ADA, which has been proven for some ADGF members. These analyses also revealed that some genes previously thought to be classic ADAs are instead ADAL or ADGFs. We here define the ADGF, ADAL, ADA, adenine deaminase (ADE), and AMP deaminase (AMPD) groups as subfamilies of the adenyl-deaminase family. The availability of genomic data for the members of this family allowed us to reconstruct the intron evolution within the phylogeny and strengthen the introns-late hypothesis of the synthetic introns theory. This study shows that ADA activity is clearly more complex than once thought, perhaps involving a delicately balanced pattern of temporal and spatial expression of a number of paralogous proteins.



Similar content being viewed by others
References
Akalal DB, Nagle GT (2001) Mollusk-derived growth factor: cloning and developmental expression in the central nervous system and reproductive tract of Aplysia. Brain Res Mol Brain Res 91:163–168
Akalal DB, Bottenstein JE, Lee SH, Han JH, Chang DJ, Kaang BK, Nagle GT (2003) Aplysia mollusk-derived growth factor is a mitogen with adenosine deaminase activity and is expressed in the developing central nervous system. Brain Res Mol Brain Res 117:228–236
Akalal DB, Schein CH, Nagle GT (2004) Mollusk-derived growth factor and the new subfamily of adenosine deaminase–related growth factors. Curr Pharm Des 10:3893–3900
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Becerra A, Lazcano A (1998) The role of gene duplication in the evolution of purine nucleotide salvage pathways. Orig Life Evol Biosph 28:539–553
Bendtsen JD, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340:783–795
Benson DA, Karsch–Mizrachi I, Lipman DJ, Ostell J, Wheeler DL (2004) GenBank: update. Nucleic Acids Res 32 (database issue):D23–D26
Blackburn MR, Datta SK, Kellems RE (1998) Adenosine deaminase-deficient mice generated using a two-stage genetic engineering strategy exhibit a combined immunodeficiency. J Biol Chem 273:5093–5100
Chang ZY, Nygaard P, Chinault AC, Kellems RE (1991) Deduced amino acid sequence of Escherichia coli adenosine deaminase reveals evolutionarily conserved amino acid residues: implications for catalytic function. Biochemistry 30:2273–2280
Charlab R, Rowton ED, Ribeiro JM (2000) The salivary adenosine deaminase from the sand fly Lutzomyia longipalpis. Exp Parasitol 95:45–53
Charlab R, Valenzuela JG, Andersen J, Ribeiro JM (2001) The invertebrate growth factor/CECR1 subfamily of adenosine deaminase proteins. Gene 267:13–22
Cordero OJ, Salgado FJ, Fernandez-Alonso CM, Herrera C, Lluis C, Franco R, Nogueira M (2001) Cytokines regulate membrane adenosine deaminase on human activated lymphocytes. J Leukoc Biol 70:920–930
de Souza SJ (2003) The emergence of a synthetic theory of intron evolution. Genetica 118:117–121
de Souza SJ, Long M, Klein RJ, Roy S, Lin S, Gilbert W (1998) Toward a resolution of the introns early/late debate: only phase zero introns are correlated with the structure of ancient proteins. Proc Natl Acad Sci USA 95:5094–5099
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Emanuelsson O, Nielsen H, Brunak S, von Heijne G (2000) Predicting subcellular localization of proteins based on their N-terminal amino acid sequence. J Mol Biol 300:1005–1016
Fedorov A, Cao X, Saxonov S, de Souza SJ, Roy SW, Gilbert W (2001) Intron distribution difference for 276 ancient and 131 modern genes suggests the existence of ancient introns. Proc Natl Acad Sci USA 98:13177–13182
Fedorova L, Fedorov A (2003) Introns in gene evolution. Genetica 118:123–131
Felsenstein J (2000) PHYLIP (Phylogeny Inference Package) version 3.6 alpha. Computer programs and documentation. Department of Genetics, University of Washington, Seattle
Franco R, Casado V, Ciruela F, Saura C, Mallol J, Canela EI, Lluis C (1997) Cell surface adenosine deaminase: much more than an ectoenzyme. Prog Neurobiol 52:283–294
Franco R, Mallol J, Casado V, Lluis C, Canela EI, Saura C, Blanco J, Ciruela F (1998) Ecto-denosine deaminase: an ecto-enzyme and a costimulatory protein acting on a variety of cell surface receptors. Drug Dev Res 45:261–268
Gasteiger E, Gattiker A, Hoogland C, Ivanyi I, Appel RD, Bairoch A (2003) ExPASy: The proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res 31:3784–3788
Gilbert W, Marchionni M, McKnight G (1986) On the antiquity of introns. Cell 46:151–153
Gross M (1994) Molecular biology of AMP deaminase deficiency. Pharm World Sci 16:55–61
Gu X (2001) Maximum-likelihood approach for gene family evolution under functional divergence. Mol Biol Evol 18:453–464
Hershfield MS (2003) Genotype is an important determinant of phenotype in adenosine deaminase deficiency. Curr Opin Immunol 15:571–577
Hirschhorn R, Ratech H (1980) Isozymes of adenosine deaminase. Isozymes Curr Top Biol Med Res 4:131–157
Holder M, Lewis PO (2003) Phylogeny estimation: traditional and Bayesian approaches. Nat Rev Genet 4:275–284
Homma K, Matsushita T, Natori S (1996) Purification, characterization, and cDNA cloning of a novel growth factor from the conditioned medium of NIH-Sape-4, an embryonic cell line of Sarcophaga peregrina (flesh fly). J Biol Chem 271:13770–13775
Homma KJ, Tanaka Y, Matsushita T, Yokoyama K, Matsui H, Natori S (2001) Adenosine deaminase activity of insect-derived growth factor is essential for its growth factor activity. J Biol Chem 276:43761–43766
Huelsenbeck JP, Ronquist F, Nielsen R, Bollback JP (2001) Bayesian inference of phylogeny and its impact on evolutionary biology. Science 294:2310–2314
Iwaki-Egawa S, Namiki C, Watanabe Y (2004) Adenosine deaminase 2 from chicken liver: purification, characterization, and N-terminal amino acid sequence. Comp Biochem Physiol B Biochem Mol Biol 137:247–254
Jones DT, Taylor WR, Thornton JM (1992) The rapid generation of mutation data matrices from protein sequences. Comput Appl Biosci 8:275–282
Li S, Aksoy S (2000) A family of genes with growth factor and adenosine deaminase similarity are preferentially expressed in the salivary glands of Glossina m. morsitans. Gene 252:83–93
Maddison WP, Maddison DR (1989) Interactive analysis of phylogeny and character evolution using the computer program MacClade. Folia Primatol 53:190–202
Maier SA, Podemski L, Graham SW, McDermid HE, Locke J (2001) Characterization of the adenosine deaminase–related growth factor (ADGF) gene family in Drosophila. Gene 280:27–36
Matsushita T, Fujii-Taira I, Tanaka Y, Homma KJ, Natori S (2000) Male-specific IDGF, a novel gene encoding a membrane-bound extracellular signaling molecule expressed exclusively in testis of Drosophila melanogaster. J Biol Chem 275:36934–36941
McDermid HE, Duncan AM, Brasch KR, Holden JJ, Magenis E, Sheehy R, Burn J, Kardon N, Noel B, Schinzel A (1986) Characterization of the supernumerary chromosome in cat eye syndrome. Science 232:646–648
Mohamedali KA, Kurz LC, Rudolph FB (1996) Site-directed mutagenesis of active site glutamate-217 in mouse adenosine deaminase. Biochemistry 35:1672–1680
Niedzwicki JG, Abernethy DR (1991) Structure–activity relationship of ligands of human plasma adenosine deaminase2. Biochem Pharmacol 41:1615–1624
Niedzwicki JG, Liou C, Abernethy DR, Lima JE, Hoyt A, Lieberman M, Bethlenfalvay NC (1995) Adenosine deaminase isoenzymes of the opossum Didelphis virginiana: initial chromatographic and kinetic studies. Comp Biochem Physiol B Biochem Mol Biol 111:291–298
Papadakis MN, Patrinos GP (1999) Contribution of gene conversion in the evolution of the human beta-like globin gene family. Hum Genet 104:117–125
Riazi MA, Brinkman-Mills P, Nguyen T, Pan H, Phan S, Ying F, Roe BA, Tochigi J, Shimizu Y, Minoshima S, Shimizu N, Buchwald M, McDermid HE (2000) The human homolog of insect-derived growth factor, CECR1, is a candidate gene for features of cat eye syndrome. Genomics 64:277–285
Ribard C, Rochet M, Labedan B, Daignan-Fornier B, Alzari P, Scazzocchio C, Oestreicher N (2003) Sub-families of alpha/beta barrel enzymes: a new adenine deaminase family. J Mol Biol 334:1117–1131
Ribeiro JM, Charlab R, Valenzuela JG (2001) The salivary adenosine deaminase activity of the mosquitoes Culex quinquefasciatus and Aedes aegypti. J Exp Biol 204:2001–2010
Riveros-Rosas H, Julian-Sanchez A, Villalobos-Molina R, Pardo JP, Pina E (2003) Diversity, taxonomy and evolution of medium-chain dehydrogenase/reductase superfamily. Eur J Biochem 270:3309–3334
Rogozin IB, Lyons-Weiler J, Koonin EV (2000) Intron sliding in conserved gene families. Trends Genet 16:430–432
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Schinzel A, Schmid W, Fraccaro M, Tiepolo L, Zuffardi O, Opitz JM, Lindsten J, Zetterqvist P, Enell H, Baccichetti C, Tenconi R, Pagon RA (1981) The “cat eye syndrome”: dicentric small marker chromosome probably derived from a no.22 (tetrasomy 22pter to q11) associated with a characteristic phenotype. Report of 11 patients and delineation of the clinical picture. Hum Genet 57:148–158
Schmitt DM, Brower DL (2001) Intron dynamics and the evolution of integrin beta-subunit genes: maintenance of an ancestral gene structure in the coral, Acropora millepora. J Mol Evol 53:703–710
Sideraki V, Wilson DK, Kurz LC, Quiocho FA, Rudolph FB (1996) Site-directed mutagenesis of histidine 238 in mouse adenosine deaminase: substitution of histidine 238 does not impede hydroxylate formation. Biochemistry 35:15019–15028
Stoltzfus A, Logsdon JMJ, Palmer JD, Doolittle WF (1997) Intron “sliding” and the diversity of intron positions. Proc Natl Acad Sci USA 94:10739–10744
Swofford DL (2001) PAUP*: Phylogenetic Analysis Using Parsimony (*and other methods) version 4.0b8. Sinauer Associates, Sunderland, MA
Taylor JS, Braasch I, Frickey T, Meyer A, Van de Peer Y (2003) Genome duplication, a trait shared by 22000 species of ray-finned fish. Genome Res 13:382–390
Tyshenko MG, Walker VK (1997) Towards a reconciliation of the introns early or late views: triosephosphate isomerase genes from insects. Biochim Biophys Acta 1353:131–136
Ungerer JP, Oosthuizen HM, Bissbort SH, Vermaak WJ (1992) Serum adenosine deaminase: isoenzymes and diagnostic application. Clin Chem 38:1322–1326
Valenzuela JG, Pham VM, Garfield MK, Francischetti IM, Ribeiro JM (2002) Toward a description of the sialome of the adult female mosquito Aedes aegypti. Insect Biochem Mol Biol 32:1101–1122
Van der Weyden MB, Kelley WN (1976) Human adenosine deaminase. Distribution and properties. J Biol Chem 251:5448–5456
Venkatesh B, Ning Y, Brenner S (1999) Late changes in spliceosomal introns define clades in vertebrate evolution. Proc Natl Acad Sci USA 96:10267–10271
Wang Z, Quiocho FA (1998) Complexes of adenosine deaminase with two potent inhibitors: X-ray structures in four independent molecules at pH of maximum activity. Biochemistry 37:8314–8324
Weijer CJ (2004) Dictyostelium morphogenesis. Curr Opin Genet Dev 14:392–398
Wilson DK, Rudolph FB, Quiocho FA (1991) Atomic structure of adenosine deaminase complexed with a transition-state analog: understanding catalysis and immunodeficiency mutations. Science 252:1278–1284
Zurovec M, Dolezal T, Gazi M, Pavlova E, Bryant PJ (2002) Adenosine deaminase-related growth factors stimulate cell proliferation in Drosophila by depleting extracellular adenosine. Proc Natl Acad Sci USA 99:4403–4408
Acknowledgments
We would like to thank Fang Yang for sequencing the chicken CECR1 clone. Rezika Zurch and Twila Yobb sequenced the mouse ADAL clone. N. Ueno and H. Lillehoj provided the Xenopus and chicken cDNA clones, respectfully. We thank Isabelle Delisle for critical reading of the manuscript. Special thanks go to Warren Gallin for guidance and helpful discussions throughout the phylogenetic analysis. This study was supported by the Canadian Institutes of Health Research (H.E.M.). S.A.M. held graduate studentships from the Canadian Institutes of Health Research and the Alberta Heritage Foundation for Medical Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
[Reviewing Editor: Dr. Martin Kreitman]
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Maier, S.A., Galellis, J.R. & McDermid, H.E. Phylogenetic Analysis Reveals a Novel Protein Family Closely Related to Adenosine Deaminase. J Mol Evol 61, 776–794 (2005). https://doi.org/10.1007/s00239-005-0046-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-005-0046-y