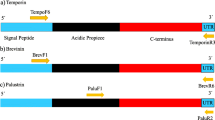
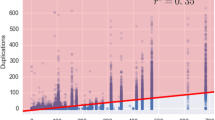
Abstract
Nonrandom patterns associated with adaptively evolving genes can shed light on how selection and mutation produce rapid changes in sequences. I examine such patterns in two independent families of antimicrobial peptide genes: those in frogs, which are known to have evolved under positive selection, and those in flatfishes, which I show have also evolved under positive selection. I address two recently proposed hypotheses about the molecular evolution of antimicrobial peptide genes. The first is that the mature peptide region is replicated by an error-prone polymerase that increases the mutation rate and the transversion/transition ratio compared to the signal sequence of the same genes. The second is that mature peptides evolve in a coordinated fashion with their propieces, such that a change in net charge in one molecular region prompts an opposite change in charge in the other region. I test these hypotheses using alternative methods that minimize alignment errors, correct for phylogenetic nonindependence, reduce sequence saturation, and account for differing selection pressures on different regions of the gene. In both gene families I show that divergence at both synonymous and nonsynonymous sites within the mature peptide region is enhanced. However, in neither gene family is there evidence of an increased mutational transversion/transition ratio or coordinated evolution. My observations are consistent with either an elevated mutation rate in an adaptively evolving gene region or widespread selection on “silent” sites. These hypotheses challenge the assumption that mutations are random and can be measured by the synonymous substitution rate.






Similar content being viewed by others
References
Alvarez-Valin F, Jabbari K, Bernardi G (1998) Synonymous and nonsynonymous substitutions in mammalian genes: intragenic correlations. J Mol Evol 46:37–44
Amiche M, Ducancel F, Mor A, Boulain JC, Menez A, Nicolas P (1994) Precursors of vertebrate peptide antibiotics dermaseptin b and adenoregulin have extensive sequence identities with precursors of opioid peptides dermorphin, dermenkephalin, and deltorphins. J Biol Chem 269:17847–17852
Amiche M, Seon AA, Wroblewski H, Nicolas P (2000) Isolation of dermatoxin from frog skin, an antibacterial peptide encoded by a novel member of the dermaseptin genes family. Eur J Biochem 267:4583–4592
Andreu D, Rivas L (1998) Animal antimicrobial peptides: an overview. Biopolymers 47:415–433
Anisimova M, Bielawski JP, Yang Z (2001) Accuracy and power of the likelihood ratio test in detecting adaptive molecular evolution. Mol Biol Evol 18:1585–1592
Anisimova M, Bielawski JP, Yang Z (2002) Accuracy and power of Bayes prediction of amino acid sites under positive selection. Mol Biol Evol 19:950–958
Bernardi G, Mouchiroud D, Gautier C (1993) Silent substitutions in mammalian genomes and their evolutionary implications. J Mol Evol 37:583–589
Bowman HG, (2003) Antibacterial peptides: basic facts and emerging concepts. J Intern Med 254:197–215
Chen T, Farragher S, Bjourson AJ, Orr DF, Rao P, Shaw C (2003a) Granular gland transcriptomes in stimulated amphibian skin secretions. Biochem J 371:125–130
Chen T, Tang L, Shaw C (2003b) Identification of three novel Phyllomedusa sauvagei dermaseptins (sVI–sVIII) by cloning from a skin secretion-derived cDNA library. Regul Pept 116:139–146
Cole AM, Weis P, Diamond G (1997) Isolation and characterization of pleurocidin, an antimicrobial peptide in the skin secretions of winter flounder. J Biol Chem 272:12008–12013
Conticello SG, Gilad Y, Avidan N, Ben-Asher E, Levy Z, Fainzilber M (2001) Mechansims for evolving hypervariability: the case of conopeptides. Mol Biol Evol 18:120–131
Douglas SE, Gallant JW, Gong Z, Hew C (2001) Cloning and developmental expression of a family of pleurocidin-like antimicrobial peptides from winter flounder, Pleuronectes americanus (Walbaum). Dev Comp Immunol 25:137–147
Douglas SE, Patrzykat A, Pytyck J, Gallant JW (2003) Identification, structure and differential expression of novel pleurocidins clustered on the genome of the winter flounder, Pseudopleuronectes americanus (Walbaum). Eur J Biochem 270:3720–3730
Duda TF Jr, Vanhoye D, Nicolas P (2002) Roles of diversifying selection and coordinated evolution in the evolution of amphibian antimicrobial peptides. Mol Biol Evol 19:858–864
DuMont VB, Fay JC, Calabrese PP, Aquadro CF (2004) DNA variability and divergence at the Notch locus in Drosophila melanogaster and D. simulans: a case of accelerated synonymous site divergence. Genetics 167:171–185
Felsenstein J, (1985) Phylogenies and the comparative method. Am Nat 125:1–15
Felsenstein J (2004) PHYLIP: (Phylogeny Inference Package), version 3.61. Department of Genetics, University of Washington, Seattle
Froy O, Gurevitz M (2003) Arthropod and mollusk defensins—evolution by exon-shuffling. Trends Genet 19:684–687
Hughes AL, Yeager M (1997) Coordinated amino acid changes in the evolution of mammalian defensins. J Mol Evol 44:675–682
Lercher MJ, Chamary J-V, Hurst LD (2004) Genomic regionality in rates of evolution is not explained by clustering of genes of comparable expression profile. Genome Res 14:1002–1013
Metz EC, Robles-Sikisaka R, Vacquier VD (1998) Nonsynonymous substitution in abalone sperm fertilization genes exceeds substitution in introns and mitochondrial DNA. Proc Natl Acad Sci USA 95:10676–10681
Michaelson D, Rayner J, Conto M, Ganz T (1992) Cationic defensins arise from charge-neutralized propeptides: a mechanism for avoiding leukocyte autotoxicity? J Leukoc Biol 51:632–639
Morrison GM, Semple CAM, Kilanowski FM, Hill RE, Dorin JR (2003) Signal sequence conservation and mature peptide divergence within subgroups of the murine β-defensin gene family. Mol Biol Evol 20:460–470
Nicolas P, Vanhoye D, Amiche M (2003) Molecular strategies in biological evolution of antimicrobial peptides. Peptides 24:1669–1680
Ogawa T, Ishii C, Kagawa D, Muramoto K, Kamiya H (1999) Accelerated evolution in the protein-coding region of galectin cDNAs, congerin I and congerin II, from skin mucus of conger eel (Conger myriaster). Biosci Biotechnol Biochem 63:1203–1208
Pamilo P, O’Neill RJW (1997) Evolution of the Sry genes. Mol Biol Evol 14:49–55
Park JM, Lee JY, Moon HM, Lee BJ (1995) Molecular cloning of cDNAs encoding precursors of frog skin antimicrobial peptides from Rana rugosa. Biochim Biophys Acta 1264:23–25
Patrzykat A, Gallant JW, Seo J-K, Pytyck J, Douglas SE (2003) Novel antimicrobial peptides derived from flatfish genes. Antimicrob Agents Chemother 47:2464–2470
Pupko T, Sharan R, Hasegawa M, Shamir R, Graur D (2003) Detecting excess radical replacements in phylogenetic trees. Gene 319:127–135
Rozas J, Sánchez-DelBarrio JC, Messeguer X, Rozas R (2003) DnaSP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics 19:2496–2497
Simmaco M, Mignogna G, Barra D, Bossa F (1994) Antimicrobial peptides from skin secretions of Rana esculenta. Molecular cloning of cDNAs encoding esculentin and brevinins and isolation of new active peptides. J Biol Chem 269:11956–11961
Simmaco M, Mignogna G, Barra D (1998) Antimicrobial peptides from amphibian skin: What do they tell us? Biopolymers 47:435–450
Smith NGC, Hurst LD (1999) The effect of tandem substitutions on the correlation between synonymous and nonsynonymous rates in rodents. Genetics 153:1395–1402
Swofford DL, (2002) PAUP*. Phylogenetic analysis using parsimony (*and other methods), version 4.0b10. Sinauer Associates, Sunderland, MA
Tani A, Ogawa T, Nose T, Nikandrov NN, Deshimaru M, Chijiwa T, Chang C–C, Fukumaki Y, Ohno M (2002) Characterization, primary structure and molecular evolution of anticoagulant protein from Agkistrodon actus venom. Toxicon 40:803–813
Tsunoyama K, Bellgard MI, Gojobori T (2001) Intragenic variation of synonymous substitution rates is caused by nonrandom mutations at methylated CpG. J Mol Evol 53:456–464
Vanhoye D, Bruston F, Nicolas P, Amiche M (2003) Antimicrobial peptides from hylid and ranin frogs originated from a 150-million year old ancestral precursor with a conserved signal peptide but a hypermutable antimicrobial domain. Eur J Biochem 270:2068–2081
Vouille V, Amiche M, Nicolas P (1997) Structure of genes for dermaseptins B, antimicrobial peptides from frog skin. Exon 1-encoded prepropeptide is conserved in genes for peptides of highly different structures and activities. FEBS Lett 414:27–32
Wechselberger C (1998) Cloning of cDNAs encoding new peptides of the dermaseptin-family. Biochim Biophys Acta 1388:279–283
Wolfe KH, Sharp PM (1993) Mammalian gene evolution: nucleotide sequence divergence between mouse and rat. J Mol Evol 37:441–456
Wolfe KH, Sharp PM, Li W-H (1989) Mutation rates differ among regions of the mammalian genome. Nature 337:283–285
Yang Z, (1997) PAML: a program package for phylogenetic analysis by maximum likelihood. Comput Appl Biosci 13:555–556
Yang Z, Nielsen R, Goldman N, Pederson A-MK (2000) Codon-substitution models for heterogeneous selection pressure at amino acid sites. Genetics 155:431–449
Yeaman MR, Yount NY (2003) Mechanisms of antimicrobial peptide action and resistance. Pharmacol Rev 55:27–55
Zhang J (2000) Rates of conserved and radical nonsynonymous nucleotide substitutions in mammalian nuclear genes. J Mol Evol 50:56–68
Acknowledgments
This work was supported by a National Science Foundation Graduate Research Fellowship. I thank M.S. Blouin, C.D. Criscione, C.A. Palmer, and R.A. Watts for suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
[Reviewing Editor: Dr. Willie J. Swanson]
Rights and permissions
About this article
Cite this article
Tennessen, J.A. Enhanced Synonymous Site Divergence in Positively Selected VertebrateAntimicrobial Peptide Genes. J Mol Evol 61, 445–455 (2005). https://doi.org/10.1007/s00239-004-0330-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-004-0330-2