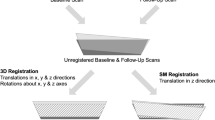
Abstract
Micro-computed tomography (micro-CT) is a widely used technique to track bone structural and mineral changes in small animals in vivo. Precise definition of volumes of interest (VOIs) in follow-up scans is required to accurately quantify these changes. To improve precision, VOIs can be transferred from baseline images onto follow-ups using image registration. We studied the performance of a registration procedure applied to in vivo data sets of anabolic and osteoporotic bone changes in mice. Micro-CT image data from two separate CD1 mouse data sets were studied. The first included a group treated with parathyroid hormone (PTH) and control and the second, an ovariectomy (OVX) group and control. Micro-CT was performed once per week for 4 weeks at the proximal tibia starting at treatment onset (PTH data set) or after surgery (OVX data set). A series consisting entirely of user-defined VOIs and a registered series where VOIs defined at baseline were transferred to follow-ups were created. Standard bone structural and mineral measurements were calculated. Image registration resulted in a 13–56 % reduction in precision error. Significant effects of registration to detect PTH-induced changes in BV/TV and trabecular BMD were observed. When changes were very pronounced or small, the qualitative improvement observed for the registered data set did not reach statistical significance. This study documents an increase in long-term precision of micro-CT measurements with image registration. Sensitivity to detect changes was improved but not uniform for all parameters. Future study of this technique on images with a smaller voxel size (<19 μm) may capture the effect in greater detail, in particular for trabecular thickness, where changes may be too small to be observed with the voxel size used here. Our results document the value of registration and indicate that the magnitude of improvement depends on the model and treatment chosen.





Similar content being viewed by others
References
Seeman E, Delmas PD (2006) Bone quality—the material and structural basis of bone strength and fragility. N Engl J Med 354:2250–2261
Waarsing JH, Day JS, van der Linden JC, Ederveen AG, Spanjers C, De Clerck N, Sasov A, Verhaar JA, Weinans H (2004) Detecting and tracking local changes in the tibiae of individual rats: a novel method to analyse longitudinal in vivo micro-CT data. Bone 34:163–169
Ruegsegger P, Koller B, Muller R (1996) A microtomographic system for the nondestructive evaluation of bone architecture. Calcif Tissue Int 58:24–29
Feldkamp LA, Goldstein SA, Parfitt AM, Jesion G, Kleerekoper M (1989) The direct examination of three-dimensional bone architecture in vitro by computed tomography. J Bone Miner Res 4:3–11
Brouwers JEM, Lambers FM, Gasser JA, van Rietbergen B, Huiskes R (2008) Bone degeneration and recovery after early and late bisphosphonate treatment of ovariectomized Wistar rats assessed by in vivo micro-computed tomography. Calcif Tissue Int 82:202–211
Buie HR, Moore CP, Boyd SK (2008) Postpubertal architectural developmental patterns differ between the L3 vertebra and proximal tibia in three inbred strains of mice. J Bone Miner Res 23:2048–2059
Campbell GM, Buie HR, Boyd SK (2008) Signs of irreversible architectural changes occur early in the development of experimental osteoporosis as assessed by in vivo micro-CT. Osteoporos Int 19:1409–1419
David V, Laroche N, Boudignon B, Lafage-Proust MH, Alexandre C, Rüegsegger P, Vico L (2003) Noninvasive in vivo monitoring of bone architecture alterations in hindlimb-unloaded female rats using novel three-dimensional microcomputed tomography. J Bone Miner Res 18:1622–1631
Gasser JA, Ingold P, Grosios K, Laib A, Hammerle S, Koller B (2005) Noninvasive monitoring of changes in structural cancellous bone parameters with a novel prototype micro-CT. J Bone Miner Metab 23(Suppl):90–96
Klinck J, Boyd SK (2008) The magnitude and rate of bone loss in ovariectomized mice differs among inbred strains as determined by longitudinal in vivo micro-computed tomography. Calcif Tissue Int 83:70–79
Boyd SK, Moser S, Kuhn M, Klinck RJ, Krauze PL, Müller R, Gasser JA (2006) Evaluation of three-dimensional image registration methodologies for in vivo micro-computed tomography. Ann Biomed Eng 34:1587–1599
Brouwers JEM, Van Rietbergen B, Huiskes R, Ito K (2009) Effects of PTH treatment on tibial bone of ovariectomized rats assessed by in vivo micro-CT. Osteoporos Int 20:1823–1835
Campbell GM, Bernhardt R, Scharnweber D, Boyd SK (2011) The bone architecture is enhanced with combined PTH and alendronate treatment compared to monotherapy while maintaining the state of surface mineralization in the OVX rat. Bone 49:225–232
Schulte FA, Lambers FM, Kuhn G, Muller R (2011) In vivo micro-computed tomography allows direct three-dimensional quantification of both bone formation and bone resorption parameters using time-lapsed imaging. Bone 48:433–442
Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Muller R (2010) Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res 25:1468–1486
Glüer CC, Blake G, Lu Y, Blunt BA, Jergas M, Genant HK (1995) Accurate assessment of precision errors: how to measure the reproducibility of bone densitometry techniques. Osteoporos Int 5:262–270
Kohler T, Beyeler M, Webster D, Müller R (2005) Compartmental bone morphometry in the mouse femur: reproducibility and resolution dependence of microtomographic measurements. Calcif Tissue Int 77:281–290
Umoh JU, Sampaio AV, Welch I, Pitelka V, Goldberg HA, Underhill TM, Holdsworth DW (2009) In vivo micro-CT analysis of bone remodeling in a rat calvarial defect model. Phys Med Biol 54:2147–2161
Nishiyama KK, Campbell GM, Klinck RJ, Boyd SK (2009) Reproducibility of bone micro-architecture measurements in rodents by in vivo micro-computed tomography is maximized with 3D image registration. Bone 46:155–161
Buie HR, Campbell GM, Klinck RJ, Macneil JA, Boyd SK (2007) Automatic segmentation of cortical and trabecular bone compartments based on a dual threshold technique for in vivo micro-CT bone analysis. Bone 41:505–515
Hajnal JV, Saeed N, Soar EJ, Oatridge A, Young IR, Bydder GM (1995) A registration and interpolation procedure for subvoxel matching of serially acquired MR images. J Comput Assist Tomogr 19:289–296
Boyd SK, Kuhn M, Moser S, Krauze P, Klinck RJ, Mattmann C, Kuhn A, Müller R (2004) Three-dimensional image registration for longitudinal site-specific measure of bone adaptation. In: Proceedings of the European Society of Biomechanics, s-Hertogenbosch, the Netherlands, July 14–17, 2004, p 514
Hildebrand T, Ruegsegger P (1997) Quantification of bone microarchitecture with the structure model index. Comput Methods Biomech Biomed Eng 1:15–23
Hildebrand T, Rüegsegger P (1997) A new method for the model-independent assessment of thickness in three-dimensional images. J Microsc 185:67–75
Odgaard A (1997) Three-dimensional methods for quantification of cancellous bone architecture. Bone 20:315–328
Odgaard A, Gundersen HJ (1993) Quantification of connectivity in cancellous bone, with special emphasis on 3-D reconstructions. Bone 14:173–182
Glüer CC (1999) Monitoring skeletal changes by radiological techniques. J Bone Miner Res 14:1952–1962
Armitage P, Berry G, Matthews JNS (2008) Statistical methods in biomedical research. Wiley, Hoboken
Fritz V, Louis-Plence P, Apparailly F, Noel D, Voide R, Pillon A, Nicolas JC, Müller R, Jorgensen C (2007) Micro-CT combined with bioluminescence imaging: a dynamic approach to detect early tumor–bone interaction in a tumor osteolysis murine model. Bone 40:1032–1040
Rosol TJ, Tannehill-Gregg SH, LeRoy BE, Mandl S, Contag CH (2003) Animal models of bone metastasis. Cancer 97:748–757
Bing Y, Williams PJ, Niewolna M, Wang Y, Yoneda T (2002) Tumor-derived platelet-derived growth factor-BB plays a critical role in osteosclerotic bone metastasis in an animal model of human breast cancer. Cancer Res 62:917–923
Alexander JM, Bab I, Fish S, Muller R, Uchiyama T, Gronowicz G, Nahounou M, Zhao Q, White DW, Chorev M, Gazit D, Rosenblatt M (2001) Human parathyroid hormone 1–34 reverses bone loss in ovariectomized mice. J Bone Miner Res 16:1665–1673
Hurley MM, Okada Y, Xiao L, Tanaka Y, Ito M, Okimoto N, Nakamura T, Rosen CJ, Doetschman T, Coffin JD (2006) Impaired bone anabolic response to parathyroid hormone in Fgf2–/– and Fgf2+/– mice. Biochem Biophys Res Commun 341:989–994
Pierroz DD, Bonnet N, Baldock PA, Ominsky MS, Stolina M, Kostenuik PJ, Ferrari SL (2010) Are osteoclasts needed for the bone anabolic response to parathyroid hormone? A study of intermittent parathyroid hormone with denosumab or alendronate in knock-in mice expressing humanized RANKL. J Biol Chem 285:28164–28173
Pierroz DD, Bouxsein ML, Rizzoli R, Ferrari SL (2006) Combined treatment with a beta-blocker and intermittent PTH improves bone mass and microarchitecture in ovariectomized mice. Bone 39:260–267
Pauchard Y, Mattmann C, Kuhn A, Gasser JA, Boyd SK (2008) European Society of Biomechanics S.M. Perren Award 2008. Using temporal trends of 3D bone micro-architecture to predict bone quality. J Biomech 41:2946–2953
Acknowledgments
This study was carried out in the Molecular Imaging North Competence Center, funded by the European Regional Development Fund (ERDF) and the Zukunftsprogramm Wirtschaft of Schleswig-Holstein. Additional support came from funding for the Forschergruppe FOR 1586 of the Deutsche Forschungsgemeinschaft.
Ethical standard
All institutional and national guidelines for the care and use of laboratory animals were followed.
Author information
Authors and Affiliations
Corresponding author
Additional information
All authors state that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Campbell, G.M., Tiwari, S., Grundmann, F. et al. Three-dimensional Image Registration Improves the Long-term Precision of In Vivo Micro-Computed Tomographic Measurements in Anabolic and Catabolic Mouse Models. Calcif Tissue Int 94, 282–292 (2014). https://doi.org/10.1007/s00223-013-9809-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-013-9809-4