Abstract
Summary
Pregnancy and lactation cause major changes in calcium homeostasis and bone metabolism. This population-based cohort study presents the physiological changes in biochemical indices of calcium homeostasis and bone metabolism during pregnancy and lactation
Introduction
We describe physiological changes in calcium homeostasis, calcitropic hormones and bone metabolism during pregnancy and lactation.
Methods
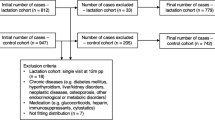
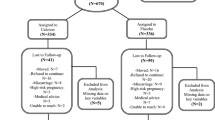
We studied 153 women planning pregnancy (n = 92 conceived) and 52 non-pregnant, age-matched female controls. Samples were collected prior to pregnancy, once each trimester and 2, 16 and 36 weeks postpartum. The controls were followed in parallel.
Results
P-estradiol (E2), prolactin and 1,25-dihydroxyvitamin D (1,25(OH)2D) increased (p < 0.001) during pregnancy, whereas plasma levels of parathyroid hormone (P-PTH) and calcitonin decreased (p < 0.01). Insulin-like growth factor I (IGF-I) was suppressed (p < 0.05) in early pregnancy but peaked in the third trimester. Postpartum, E2 was low (p < 0.05); prolactin decreased according to lactation status (p < 0.05). 1,25(OH)2D was normal and IGF-I was again reduced (p < 0.05). P-PTH and calcitonin increased postpartum. From early pregnancy, markers of bone resorption and formation rose and fall, respectively (p < 0.001). From the third trimester, bone formation markers increased in association with IGF-I changes (p < 0.01). Postpartum increases in bone turnover markers were associated with lactation status (p < 0.001). During lactation, plasma phosphate was increased, whereas calcium levels tended to be decreased which may stimulate PTH levels during and after prolonged lactation.
Conclusion
The increased calcium requirements in early pregnancy are not completely offset by increased intestinal calcium absorption caused by high 1,25(OH)2D since changes in bone markers indicated a negative bone balance. The rise in bone formation in late pregnancy may be initiated by a spike in IGF-I levels. The high bone turnover in lactating women may be related to high prolactin and PTH levels, low E2 levels and perhaps increased parathyroid hormone-related protein levels.





Similar content being viewed by others
References
Naylor KE, Iqbal P, Fledelius C, Fraser RB, Eastell R (2000) The effect of pregnancy on bone density and bone turnover. J Bone Miner Res 15:129–137
Cross NA, Hillman LS, Allen SH, Krause GF, Vieira NE (1995) Calcium homeostasis and bone metabolism during pregnancy, lactation, and postweaning: a longitudinal study. Am J Clin Nutr 61:514–523
Kent GN, Price RI, Gutteridge DH, Allen JR, Rosman KJ, Smith M, Bhagat CI, Wilson SG, Retallack RW (1993) Effect of pregnancy and lactation on maternal bone mass and calcium metabolism. Osteoporos Int 3(Suppl 1):44–47
Ulrich U, Miller PB, Eyre DR, Chesnut CH III, Schlebusch H, Soules MR (2003) Bone remodeling and bone mineral density during pregnancy. Arch Gynecol Obstet 268:309–316
Paoletti AM, Orru M, Floris L, Guerriero S, Ajossa S, Romagnino S, Melis GB (2003) Pattern of bone markers during pregnancy and their changes after delivery. Horm Res 59:21–29
Haliloglu B, Ilter E, Aksungar FB, Celik A, Coksuer H, Gunduz T, Yucel E, Ozekici U (2011) Bone turnover and maternal 25(OH) vitamin D3 levels during pregnancy and the postpartum period: should routine vitamin D supplementation be increased in pregnant women? Eur J Obstet Gynecol Reprod Biol 158:24–27
Kovacs CS (2011) Calcium and bone metabolism disorders during pregnancy and lactation. Endocrinol Metab Clin North Am 40:795–826
Favus M (2006) Primer on the metabolic bone diseases and disorders of mineral metabolism, vol 6. American Society for Bone and Mineral Research, Washington, DC, pp. 50–132
Black AJ, Topping J, Durham B, Farquharson RG, Fraser WD (2000) A detailed assessment of alterations in bone turnover, calcium homeostasis, and bone density in normal pregnancy. J Bone Miner Res 15:557–563
More C, Bhattoa HP, Bettembuk P, Balogh A (2003) The effects of pregnancy and lactation on hormonal status and biochemical markers of bone turnover. Eur J Obstet Gynecol Reprod Biol 106:209–213
Ritchie LD, Fung EB, Halloran BP, Turnlund JR, Van Loan MD, Cann CE, King JC (1998) A longitudinal study of calcium homeostasis during human pregnancy and lactation and after resumption of menses. Am J Clin Nutr 67:693–701
Specker BL, Tsang RC, Ho ML (1991) Changes in calcium homeostasis over the first year postpartum: effect of lactation and weaning. Obstet Gynecol 78:56–62
Greer FR, Tsang RC, Searcy JE, Levin RS, Steichen JJ (1982) Mineral homeostasis during lactation- relationship to serum 1,25-dihydroxyvitamin D, 25-hydroxyvitamin D, parathyroid hormone and calcitonin. Am J Clin Nutr 36:431–437
Krebs NF, Reidinger CJ, Robertson AD, Brenner M (1997) Bone mineral density changes during lactation: maternal, dietary, and biochemical correlates. Am J Clin Nutr 65:1738–1746
Cross NA, Hillman LS, Allen SH, Krause GF (1995) Changes in bone mineral density and markers of bone remodeling during lactation and postweaning in women consuming high amounts of calcium. J Bone Miner Res 10:1312–1320
Moller UK, Vieth SS, Mosekilde L, Rejnmark L (2012) Changes in bone mineral density and body composition during pregnancy and postpartum. A controlled cohort study. Osteoporos Int 23:1213–1223
Drinkwater BL, Chesnut CH III (1991) Bone density changes during pregnancy and lactation in active women: a longitudinal study. Bone Miner 14:153–160
Kaur M, Godber IM, Lawson N, Baker PN, Pearson D, Hosking DJ (2003) Changes in serum markers of bone turnover during normal pregnancy. Ann Clin Biochem 40:508–513
Seriwatanachai D, Thongchote K, Charoenphandhu N, Pandaranandaka J, Tudpor K, Teerapornpuntakit J, Suthiphongchai T, Krishnamra N (2008) Prolactin directly enhances bone turnover by raising osteoblast-expressed receptor activator of nuclear factor kappaB ligand/osteoprotegerin ratio. Bone 42:535–546
Wilson SG, Retallack RW, Kent JC, Worth GK, Gutteridge DH (1990) Serum free 1,25-dihydroxyvitamin D and the free 1,25-dihydroxyvitamin D index during a longitudinal study of human pregnancy and lactation. Clin Endocrinol (Oxf) 32:613–622
Reddy GS, Norman AW, Willis DM, Goltzman D, Guyda H, Solomon S, Philips DR, Bishop JE, Mayer E (1983) Regulation of vitamin D metabolism in normal human pregnancy. J Clin Endocrinol Metab 56:363–370
Hillman L, Sateesha S, Haussler M, Wiest W, Slatopolsky E, Haddad J (1981) Control of mineral homeostasis during lactation: interrelationships of 25-hydroxyvitamin D, 24,25-dihydroxyvitamin D, 1,25-dihydroxyvitamin D, parathyroid hormone, calcitonin, prolactin, and estradiol. Am J Obstet Gynecol 139:471–476
Kent GN, Price RI, Gutteridge DH, Rosman KJ, Smith M, Allen JR, Hickling CJ, Blakeman SL (1991) The efficiency of intestinal calcium absorption is increased in late pregnancy but not in established lactation. Calcif Tissue Int 48:293–295
Kalkwarf HJ, Specker BL, Heubi JE, Vieira NE, Yergey AL (1996) Intestinal calcium absorption of women during lactation and after weaning. Am J Clin Nutr 63:526–531
Vargas Zapata CL, Donangelo CM, Woodhouse LR, Abrams SA, Spencer EM, King JC (2004) Calcium homeostasis during pregnancy and lactation in Brazilian women with low calcium intakes: a longitudinal study. Am J Clin Nutr 80:417–422
Møller UK, Streym S, Heickendorff L, Mosekilde L, Rejnmark L (2012) Effects of 25OHD concentrations on chances of pregnancy and pregnancy outcomes: a cohort study in healthy Danish women. Eur J Clin Nutr 66(7):862–868
Hermann AP, Thomsen J, Vestergaard P, Mosekilde L, Charles P (1999) Assessment of kalcium intake. A quick method compared to a 7 days food diary. Calcif Tissue Int 64(S1):S82
Quadri KH, Bernardini J, Greenberg A, Laifer S, Syed A, Holley JL (1994) Assessment of renal function during pregnancy using a random urine protein to creatinine ratio and Cockcroft–Gault formula. Am J Kidney Dis 24:416–420
Hojskov CS, Heickendorff L, Moller HJ (2010) High-throughput liquid-liquid extraction and LCMSMS assay for determination of circulating 25(OH) vitamin D3 and D2 in the routine clinical laboratory. Clin Chim Acta 411:114–116
Frystyk J, Dinesen B, Orskov H (1995) Non-competitive time-resolved immunofluorometric assays for determination of human insulin-like growth factor I and II. Growth Regul 5:169–176
Schifter S (1993) A new highly sensitive radioimmunoassay for human calcitonin useful for physiological studies. Clin Chim Acta 215:99–109
Ardawi MS, Nasrat HA, BA'Aqueel HS (1997) Calcium-regulating hormones and parathyroid hormone-related peptide in normal human pregnancy and postpartum: a longitudinal study. Eur J Endocrinol 137:402–409
Turner M, Barre PE, Benjamin A, Goltzman D, Gascon-Barre M (1988) Does the maternal kidney contribute to the increased circulating 1,25-dihydroxyvitamin D concentrations during pregnancy? Miner Electrolyte Metab 14:246–252
Kubota M, Ohno J, Shiina Y, Suda T (1982) Vitamin D metabolism in pregnant rabbits: differences between the maternal and fetal response to administration of large amounts of vitamin D3. Endocrinology 110:1950–1956
Kovacs CS, Kronenberg HM (1997) Maternal-fetal calcium and bone metabolism during pregnancy, puerperium, and lactation. Endocr Rev 18:832–872
Novakovic B, Sibson M, Ng HK, Manuelpillai U, Rakyan V, Down T, Beck S, Fournier T, Evain-Brion D, Dimitriadis E, Craig JM, Morley R, Saffery R (2009) Placenta-specific methylation of the vitamin D 24-hydroxylase gene: implications for feedback autoregulation of active vitamin D levels at the fetomaternal interface. J Biol Chem 284:14838–14848
Potashnik G, Lunenfeld E, Levitas E, Itskovitz J, Albutiano S, Yankowitz N, Sonin Y, Levy J, Glezerman M, Shany S (1992) The relationship between endogenous oestradiol and vitamin D3 metabolites in serum and follicular fluid during ovarian stimulation for in-vitro fertilization and embryo transfer. Hum Reprod 7:1357–1360
Kumar R, Abboud CF, Riggs BL (1980) The effect of elevated prolactin levels on plasma 1,25-dihydroxyvitamin D and intestinal absorption of calcium. Mayo Clin Proc 55:51–53
Strewler GJ (2000) The physiology of parathyroid hormone-related protein. N Engl J Med 342:177–185
Bertelloni S, Baroncelli GI, Pelletti A, Battini R, Saggese G (1994) Parathyroid hormone-related protein in healthy pregnant women. Calcif Tissue Int 54:195–197
Hirota Y, Anai T, Miyakawa I (1997) Parathyroid hormone-related protein levels in maternal and cord blood. Am J Obstet Gynecol 177:702–706
Thiebaud D, Janisch S, Koelbl H, Hanzal E, Jacquet AF, Leodolter S, Burckhardt P, Pecherstorfer M (1993) Direct evidence of a parathyroid related protein gradient between the mother and the newborn in humans. Bone Miner 23:213–221
Fraser DR (1980) Regulation of the metabolism of vitamin D. Physiol Rev 60:551–613
Peppone LJ, Hebl S, Purnell JQ, Reid ME, Rosier RN, Mustian KM, Palesh OG, Huston AJ, Ling MN, Morrow GR (2010) The efficacy of calcitriol therapy in the management of bone loss and fractures: a qualitative review. Osteoporos Int 21:1133–1149
Fuglsang J, Lauszus F, Flyvbjerg A, Ovesen P (2003) Human placental growth hormone, insulin-like growth factor I and -II, and insulin requirements during pregnancy in type 1 diabetes. J Clin Endocrinol Metab 88:4355–4361
Chellakooty M, Vangsgaard K, Larsen T, Scheike T, Falck-Larsen J, Legarth J, Andersson AM, Main KM, Skakkebaek NE, Juul A (2004) A longitudinal study of intrauterine growth and the placental growth hormone (GH)-insulin-like growth factor I axis in maternal circulation: association between placental GH and fetal growth. J Clin Endocrinol Metab 89:384–391
Lacroix MC, Guibourdenche J, Frendo JL, Muller F, Evain-Brion D (2002) Human placental growth hormone—a review. Placenta 23(Suppl A):S87–S94
Kveiborg M, Flyvbjerg A, Rattan SI, Kassem M (2000) Changes in the insulin-like growth factor-system may contribute to in vitro age-related impaired osteoblast functions. Exp Gerontol 35:1061–1074
Carney SL (1997) Calcitonin and human renal calcium and electrolyte transport. Miner Electrolyte Metab 23:43–47
Dobnig H, Kainer F, Stepan V, Winter R, Lipp R, Schaffer M, Kahr A, Nocnik S, Patterer G, Leb G (1995) Elevated parathyroid hormone-related peptide levels after human gestation: relationship to changes in bone and mineral metabolism. J Clin Endocrinol Metab 80:3699–3707
Sowers M, Eyre D, Hollis BW, Randolph JF, Shapiro B, Jannausch ML, Crutchfield M (1995) Biochemical markers of bone turnover in lactating and nonlactating postpartum women. J Clin Endocrinol Metab 80:2210–2216
Laskey MA, Prentice A, Hanratty LA, Jarjou LM, Dibba B, Beavan SR, Cole TJ (1998) Bone changes after 3 mo of lactation: influence of calcium intake, breast-milk output, and vitamin D-receptor genotype. Am J Clin Nutr 67:685–692
Kovacs CS, Chik CL (1995) Hyperprolactinemia caused by lactation and pituitary adenomas is associated with altered serum calcium, phosphate, parathyroid hormone (PTH), and PTH-related peptide levels. J Clin Endocrinol Metab 80:3036–3042
Lippuner K, Zehnder HJ, Casez JP, Takkinen R, Jaeger P (1996) PTH-related protein is released into the mother's bloodstream during location: evidence for beneficial effects on maternal calcium-phosphate metabolism. J Bone Miner Res 11:1394–1399
Grill V, Hillary J, Ho PM, Law FM, MacIsaac RJ, MacIsaac IA, Moseley JM, Martin TJ (1992) Parathyroid hormone-related protein: a possible endocrine function in lactation. Clin Endocrinol (Oxf) 37:405–410
Sowers MF, Hollis BW, Shapiro B, Randolph J, Janney CA, Zhang D, Schork A, Crutchfield M, Stanczyk F, Russell-Aulet M (1996) Elevated parathyroid hormone-related peptide associated with lactation and bone density loss. JAMA 276:549–554
Caplan RH, Wickus GG, Sloane K, Silva PD (1995) Serum parathyroid hormone-related protein levels during lactation. J Reprod Med 40:216–218
Juppner H, Wolf M, Salusky IB (2010) FGF23: more than a regulator of renal phosphate handling? J Bone Miner Res 25:2091–2097
Mortensen LH, Diderichsen F, Smith GD, Andersen AM (2009) The social gradient in birthweight at term: quantification of the mediating role of maternal smoking and body mass index. Hum Reprod 24:2629–2635
Acknowledgments
We are grateful for the financial support provided from The Danish Agency for Science, Technology and Innovation; The Aarhus University Research Foundation; The AP Moeller Foundation; the Svend Fældings Humanitære Fond; The Lundbeck Foundation; the Faculty of Health Sciences at Aarhus University; The Novo Nordic Foundation and Helga and Peter Kornings Foundation.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Møller, U.K., Streym, S., Mosekilde, L. et al. Changes in calcitropic hormones, bone markers and insulin-like growth factor I (IGF-I) during pregnancy and postpartum: a controlled cohort study. Osteoporos Int 24, 1307–1320 (2013). https://doi.org/10.1007/s00198-012-2062-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-012-2062-2