Abstract
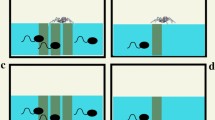
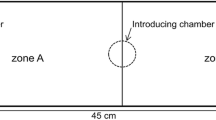
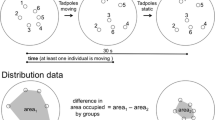
Predator–prey relationship was studied in three sympatric species of anuran tadpoles. The study design consisted of allowing predaceous Hoplobatrachus tigerinus tadpoles to devour prey tadpoles (Sphaerotheca breviceps and Bufo melanostictus) placed in a plastic tub (five tadpoles of each species, stage ~27) in 30 min. In trials without refugia, more tadpoles of Bufo fell prey compared to Sphaerotheca. In contrast, provision of refugia using hydrilla plant reversed predation risk of the two species. The swimming speed (V max = 64.55 ± 1.45 cm/s) of Hoplobatrachus tadpoles was much higher compared to the prey species (Bufo: 3.6 ± 0.4 cm/s; Sphaerotheca: 27.6 ± 1.6 cm/s). Poor swimming ability may account for the observed vulnerability of the Bufo tadpoles to predation especially in clear waters; refugia overcame predation to some extent. On the other hand, Sphaerotheca tadpoles that swim faster than the toad tadpoles were less vulnerable in open areas; refugia actually hindered swimming and increased predation. Experiments with association choice tests show that predaceous tadpoles detect prey based on both visual and chemical cues. On the other hand, the prey tadpoles detected predator based exclusively on chemical rather than visual cues. The antipredator defense strategy of the toad tadpoles is manifested in the form of reduced movements, remaining still for longer times and, increased burst speed. The present findings also suggest that in both prey species predator detection has a genetic basis since naive tadpoles with no prior exposure to predators exhibit fright response on first encounter with them.

Similar content being viewed by others
References
Blaustein L, Kiflawi M, Eitam A, Mangel M, Cohen JE (2004) Oviposition habitat selection in response to risk of predation in temporary pools: mode of detection and consistency across experimental venue. Oecologia 138:300–305
Buskirk JV, McCollum A (2000) Influence of tail shape on tadpole swimming performance. J Exp Biol 203:2449–2458
Chovanec A (1992) The influence of tadpole swimming behavior on predation by dragonfly nymphs. Amphib-reptil 13:341–349
Eklov P (2000) Chemical cues from multiple predator-prey interactions induce changes in behavior and growth of anuran larvae. Oecologia 123:192–199
Gomes FR, Bevier CR, Navas CA (2002) Environmental and physiological factors influence antipredator behavior in Scinax hiemalis (Anura: Hylidae). Copeia 2002:994–1005
Hickman CR, Stone MD, Mathis A (2004) Priority use of chemical over visual cues for detection of predators by graybelly salamanders, Eurycea multiplicata griseogaster. Herpetologica 60:203–210
Kishida O, Nishimura K (2004) Bulgy tadpoles: inducible defense morph. Oecologia 140:414–421
Laurila A, Kujasalo J, Ranta E (1997) Different antipredator behavior in two anuran tadpoles: effects of predator diet. Behav Ecol Sociobiol 40:329–336
Maerz JC, Panebianco NL, Madison DM (2001) Effects of predator chemical cues and behavioral biorhythms on foraging activity of terrestrial salamanders. J Chem Ecol 7:1333–1344
Mathis A (2003) Use of chemical cues in detection of conspecifics predators and prey by newts, Notophthalmus viridescens. Chemoecology 13:193–197
Mathis A, Smith RJF (1992) Avoidance of areas marked with a chemical alarm substance by fathead minnows (Pimephales promelas) in a natural habitat. Can J Zool 70:11473–11476
Mathis A, Smith RJF (1993) Fathead minnows, Pimephales promelas, learn to recognize northern pike, Esox lucius, as predators on the basis of chemical stimuli from minnows in the pike’s diet. Anim Behav 46:645–656
Mathis A, Vincent F (2000) Differential use of visual and chemical cues in predator recognition and threat-sensitive predator-avoidance responses by larval newts (Notophthalmus viridescens). Can J Zool 78:1646–1652
Mathis A, Murray KL, Hickman CR (2003) Do experience and body size play a role in response of larval ringed salamanders, Ambystoma annulatum, to predator kairomones? Laboratory and field assays. Ethology 109:159–170
Nystrom P, Abjornsson K (2000) Effect of fish chemical cues on interaction between tadpoles and crayfish. Oikos 88:181–190
Petranka JW, Kats LB, Andrew S (1987) Predator-prey interactions among fish and larval amphibians: use of chemical cues to detect predatory fish. Anim Behav 35:420–425
Relyea RA (2002) Costs of phenotypic plasticity. Am Nat 159:272–282
Schmidt BR, Amezquita A (2001) Predator-induced behavioural responses: tadpole of the neotropical frog Phyllomedusa tarsius do not respond to all predators. Herpetol J 11:9–15
Sharma SS, Veeranagoudar DK, Shanbhag BA, Saidapur SK (2008) Activity of Sphaerotheca breviceps tadpoles in response to chemical cues of the predaceous tadpoles Hoplobatrachus tigerinus. J Ethol 26:303–307
Skelly DK (1997) Tadpole communities. Am Sci 85:36–46
Teplitsky C, Plénet S, Joly P (2003) Tadpoles responses to risk of fish introduction. Oecologia 134:270–277
Veeranagoudar DK, Shanbhag BA, Saidapur SK (2004) Mechanism of food detection in the tadpoles of the bronze frog Rana temporalis. Acta Ethol 7:37–41
Whitham J, Mathis A (2000) Effects of hunger and predation risk on foraging behavior of gray belly salamanders, Eurycea multiplicata. J Chem Ecol 26:1659–1665
Wilson DJ, Lefcort H (1993) The effect of predator diet on the alarm response of red-legged frog, Rana aurora, tadpoles. Anim Behav 46:1017–1019
Woody DR, Mathis A (1997) Avoidance of areas labeled with chemical stimuli from damaged conspecifics by adult newts, Notophthalmus viridescens, in a natural habitat. J Herpetol 31:316–318
Woody DR, Mathis A (1998) Acquired recognition of chemical stimuli from an unfamiliar predator: Associative learning by adult newts, Notophthalmus Viridescens. Copeia 1998:1027–1031
Acknowledgments
The study was supported by a grant (No. SP/SO/C-29/2000) from the Department of Science and Technology (DST), New Delhi to SKS and partly by UGC-SAP-II grant (F.3-32/2007). DKV thanks Council of Scientific and Industrial Research (CSIR), New Delhi, for Senior Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saidapur, S.K., Veeranagoudar, D.K., Hiragond, N.C. et al. Mechanism of predator–prey detection and behavioral responses in some anuran tadpoles. Chemoecology 19, 21–28 (2009). https://doi.org/10.1007/s00049-009-0004-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-009-0004-z