Abstract
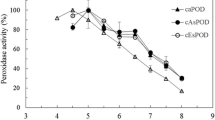
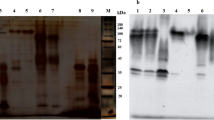
Cell wall-bound and tracheary element-specific peroxidase isoenzymes, designated P5A and P5B, were shown previously to be associated with lignification during the differentiation into tracheary elements of single cells isolated from the mesophyll ofZinnia elegans (Satoet al. Planta 189: 584–589, 1993; Planta 196: 141–147, 1995). Isoenzymes corresponding to P5 (RP5A and RP5B) were present at a relatively high level in the roots ofZinnia elegans. These isoenzymes were purified from theZinnia roots by several column-chromatographic steps. Both RP5A and RP5B had molecular masses of 35 kDa. Purified RP5A and RP5B were cleaved by CNBr and the partial amino acid sequences of these isoenzymes were determined.
Similar content being viewed by others
Abbreviations
- CAPS:
-
3-cyclohexylaminopropanesulfonic acid
- CBB:
-
Coomassie Brilliant Blue R-250
- DAB:
-
diaminobenzidine
- kDa:
-
kilodalton
- PAGE:
-
polyacrylamide gel electrophoresis
- PVDF:
-
polyvinylidene difluoride
- SDS:
-
sodium dodecyl sulfate
- TE:
-
tracheary element
References
Abeles, F.B. andBiles, C.L. 1991. Characterization of peroxidases in lignifying peach fruit endocarp. Plant Physiol.95: 269–273.
Buffard, D., Breda, C., van Huystee, R.B., Asemota, O., Pierre, M., Ha, D.B.D. andEsnault, R. 1990. Molecular cloning of complementary DNAs encoding two cationic peroxidases from cultivated peanut cells. Proc. Natl. Acad. Sci. USA87: 8874–8878.
Church, D.L. andGalston, A.W. 1988. 4-Coumarate: coenzyme A ligase and isoperoxidase expression inZinnia mesophyll cells induced to differentiate into tracheary elements. Plant Physiol.88: 679–684.
Ferrer, M.A., Pedreño, M.A., Muñoz, R. andRos Barceló, A. 1990. Oxidation of coniferyl alcohol by cell wall peroxidases at the expense of indole-3-acetic acid and O2; a model for lignification of plant cell walls in the absence of H2O2. FEBS Lett.276: 127–130.
Fukuda, H. andKomamine, A. 1980. Establishment of an experimental system for the study of tracheary element differentiation from single cells isolated from the mesophyll ofZinnia elegans. Plant Physiol.65: 57–60.
Fukuda, H. andKomamine, A. 1982. Lignin synthesis and its related enzymes as markers of trachearyelement differentiation in single cells isolated from the mesophyll ofZinnia elegans. Planta155: 423–430.
Goldberg, R., Lë, T. andCatesson, A.-M. 1985. Localization and properties of cell wall enzyme activities related to the final stages of lignin biosynthesis. J. Exp. Bot.36: 503–510.
Harkin, J.M. andObst, J.R. 1973. Lignification in trees: Induction of exclusive peroxidase participation. Science180: 296–298.
Hepler, P.K., Rice, R.M. andTerranova, W.A. 1972. Cytochemical localization of peroxidase activity in wound vessel members ofColeus. Can. J. Bot.50: 977–983.
Imberty, A., Goldberg, R. andCatesson, A.-M. 1985. Isolation and characterization ofPopulus isoperoxidases involved in the last step of lignification. Planta164: 221–226.
Laemmli, U.K. 1970. Cleavage of structural proteins during assembly of the head bacteriophage T4. Nature227: 680–685.
Lagrimini, L.M., Burkhart, W., Moyer, M. andRothstein, S. 1987. Molecular cloning of complementary DNA encoding the lignin-forming peroxidase from tobacco: Molecular analysis and tissue-specific expression. Proc. Natl. Acad. Sci. USA84: 7542–7546.
Mäder, M., Nessel, A. andBopp, M. 1977. Über die physiologische Bedeutung der Peroxidase-Isoenzymgruppen des Tabaks anhand einiger biochemischer Eigenschaften II. pH-Optima, Michaelis-Konstanten, Maximale Oxidationsraten. Z. Pflanzenphysiol.82: 247–260.
Mäder, M., Ungemach, J. andSchlo, P. 1980. The role of peroxidase isoenzyme groups ofNicotiana tabacum in hydrogen peroxide formation. Planta147: 467–470.
Masuda, H., Fukuda, H. andKomamine, A. 1983. Changes in peroxidase isoenzyme patterns during tracheary element differentiation in a culture of single cells isolated from the mesophyll ofZinnia elegans. Z. Pflanzenphysiol.112: 417–426.
Mazza, G. andWelinder, K.G. 1980. Covalent structure of turnip peroxidase 7. Cyanogen bromide fragments, complete structure and comparison to horseradish peroxidase C. Eur. J. Biochem.108: 481–489.
McDougall, G.J. 1991. Cell-wall-associated peroxidases and lignification during growth of flax fibres. J. Plant Physiol.139: 182–186.
Pedreño, M.A., Ros Barceló, A., Sabater, F. andMuñoz, R. 1989. Control by pH of cell wall peroxidase activity involved in lignification. Plant Cell Physiol.30: 237–241.
Sato, Y., Sugiyama, M., Górecki, R.J., Fukuda, H. andKomamine, A. 1993. Interrelationship between lignin deposition and the activities of peroxidase isoenzymes in differentiating tracheary elements ofZinnia: analysis using L-α-aminooxy-β-phenyl-propionic acid and 2-aminoindan-2-phosphonic acid. Planta189: 584–589.
Sato, Y., Sugiyama, M., Komamine, A. andFukuda, H. 1995. Separation and characterization of the isoenzymes of wall-bound peroxidase from culturedZinnia cells during tracheary element differentiation. Planta196: 141–147.
Smith, C.G., Rodgers, M.W., Zimmerlin, A., Ferdinando, D. andBolwell, G.P. 1994. Tissue and subcellular immunolocalization of enzymes of lignin synthesis in differentiating and wounded hypocotyl tissue of French bean (Phaseolus vulgaris L.). Planta192: 155–164.
Sugiyama, M., Fukuda, H. andKomamine, A. 1986. Effects of nutrient limitation and gamma irradiation on tracheary element differentiation and cell division in single mesophyll cells ofZinnia elegans. Plant Cell Physiol.27: 601–606.
van Huystee, R.B. 1987. Some molecular aspects of plant peroxidase. Biosynthetic studies. Annu. Rev. Plant Physiol.38: 205–219.
Welinder, K.G. 1979. Amino acid sequence studies of horseradish peroxidase. Amino acid carboxyl terminal, cyanogen bromide and tryptic fragments, the complete sequence, and some structural characteristics of horseradish peroxidase C. Eur. J. Biochem.96: 483–502.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sato, Y., Sugiyama, M., Takagi, T. et al. Purification of cationic peroxidases bound ionically to the cell walls from the roots ofZinnia elegans . J. Plant Res. 108, 463–468 (1995). https://doi.org/10.1007/BF02344235
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02344235