Summary
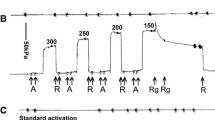
In this article, we have applied a crosslinking technique with a water-soluble carbodiimide to single glycerol-extracted muscle fibres from the rabbit. We have measured the stiffness of the fibres in a relaxing solution at high salt concentration. These fibres were crosslinked to varying extents in the rigor state. The relaxing solution caused uncrosslinked crossbridge heads (S1) to detach. High salt concentrations were used because the fibres were not activated by the crosslinked crossbridges under these conditions, although they were at physiological ionic strength. We found (1) a linear correlation between the extent of S1 crosslinking to thin filaments and the stiffness and (2) that the stiffness in the relaxing solution of muscle fibres with all the S1 heads crosslinked to thin filaments was the same as the rigor stiffness of the fibres before crosslinking. We conclude that the sarcomere compliance is mostly a property of the crossbridges (with more than 65% of the crossbridge compliance in the S1 portions and less than 35% in the S2 portion) and little of other sarcomere structures. In an earlier paper [Kimura & Tawada,Biophys. J. 603–10 (1984)], we demonstrated that the S2 portion of the crossbridge was stiff. It then follows that the crossbridge compliance, and thus the sarcomere compliance, is a property of the S1 heads. Assuming that the S1 portion of the crossbridges in rigor strained muscle fibres is bent, we calculated the Young's modulus of the S1 portion and found that it is about 102 MN m−2. Because this order of magnitude is reasonable in terms of globular protein elasticity, bending is likely to be the nature of the S1 compliance in rigor muscle fibres.
Similar content being viewed by others
References
Botts, J., Takashi, R., Torgerson, P., Hozumi, T., Muhlard, A., Mornet, D. &Morales, M. F. (1984) On the mechanism of energy transduction in myosin subfragment 1.Proc. natn. Acad. Sci. U.S.A. 81, 2060–4.
Cooke, R. (1972) A new method for producing myosin subfragment 1.Biochem. Biophys. Res. Commun. 49, 1021–8.
Cooke, R. (1981) Stress does not alter the conformation of a domain of the myosin cross-bridge in rigor muscle fibres.Nature, Lond. 294, 570–1.
Craig, R., Szent-Györgyi, A. G., Beese, L., Flicker, P., Vibert, P. &Cohen, C. (1980) Electron microscopy of thin filaments decorated with a Ca2+-regulated myosin.J. molec. Biol. 140, 35–55.
Cross, R. A., Bardsley, R. G., Ledward, D. A., Small, J. V. &Sobieszek, A. (1984) Conformational stability of the myosin rod.Eur. J. Biochem. 145, 305–10.
Eisenberg, E. &Hill, T. L. (1978) A cross-bridge model of muscle contraction.Prog. Biophys. molec. Biol. 33, 55–82.
Eisenberg, E. &Hill, T. L. (1985) Muscle contraction and free energy transduction in biological systems.Science, N.Y. 227, 999–1006.
Elliott, A. &Offer, G. (1978) Shape and flexibility of the myosin molecule.J. molec. Biol. 123, 505–19.
Ford, L. E., Huxley, A. F. &Simmons, R. M. (1977) Tension responses to sudden length change in stimulated frog muscle fibres near slack length.J. Physiol., Lond. 269, 441–515.
Ford, L. E., Huxley, A. F. &Simmons, R. M. (1981) The relation between stiffness and filament overlap in stimulated frog muscle fibres.J. Physiol., Lond. 311, 219–49.
Goldman, Y. E. &Simmons, R. M. (1977) Active and rigor muscle stiffness.J. Physiol., Lond. 269, 55–7p.
Güth, K. &Kuhn, H. J. (1978) Stiffness and tension during and after sudden length changes of glycerinated rabbit psoas fibers.Biophys. Struct. Mech. 4, 223–36.
Harrington, W. F. (1971) A mechanochemical mechanism for muscle contraction.Proc. natn. Acad. Sci. U.S.A. 68, 685–9.
Harrington, W. F. (1979) On the origin of the contractile force in skeletal muscle.Proc. natn. Acad. Sci. U.S.A. 76, 5066–70.
Heaphy, S. &Tregear, R. (1984) Stoichiometry of covalent actin-subfragment 1 complexes formed on reaction with a zero-length cross-linking compound.Biochemistry 23, 2211–14.
Heuser, J. E. &Cooke, R. (1983) Actin-myosin interactions visualized by the quick-freeze, deep-etch replica technique.J. molec. Biol. 169, 97–122.
Highsmith, S., Akasaka, K., Konrad, M., Goody, R., Holmes, K., Wade-Jardetzky, N. &Jardetzky, O. (1979) Internal motions in myosin.Biochemistry 18, 4238–44.
Huxley, A. F. (1957) Muscle structure and theories of contraction.Prog. Biophys. biophys. Chem. 7, 255–318.
Huxley, A. F. (1974) Review lecture on muscular contraction.J. Physiol., Lond. 243, 1–43.
Huxley, A. F. &Simmons, R. M. (1971) Proposed mechanism of force generation in striated muscle.Nature, Lond. 233, 533–8.
Huxley, H. E., Simmons, R. M., Faruqi, A. R., Kress, M., Bordas, J. &Koch, M. H. J. (1983) Changes in the X-ray reflections from contracting muscle during rapid mechanical transients and their structural implications.J. molec. Biol. 169, 469–506,
Kimura, M. &Tawada, K. (1984) Is the SII portion of the cross-bridge in glycerinated rabbit psoas fibers compliant in the rigor state?Biophys. J. 45, 603–10.
Knight, P. &Trinick, J. (1984) Structure of the myosin projections on native thick filaments from vertebrate skeletal muscle.J. molec. Biol. 177, 461–82.
Love, A. E. H. (1934)A Treatise on the Mathematical Theory of Elasticity, P. 329. London: Cambridge University Press.
Matsubara, I., Goldman, Y. E. &Simmons, R. M. (1984) Changes in the lateral filament spacing of skinned muscle fibers when cross-bridges attach.J. molec. Biol. 173, 15–33.
McCammon, J. A. (1984) Protein dynamics.Rep. Prog. Phys. 47, 1–46.
Mendelson, R. A. &Wagner, P. D. (1984) X-ray scattering by single-headed heavy meromyosin: Cleavage of the myosin head from the rod does not change its shape.J. molec. Biol. 177, 153–71.
Mornet, D., Bertrand, R., Pantel, P., Audemard, E. &Kassab, R. (1981) Structure of the actin-myosin interface.Nature, Lond. 292, 301–6.
Naylor, G. R. S. &Podolsky, R. J. (1981) X-ray diffraction of strained muscle fibers in rigor.Proc. natn. Acad. Sci. U.S.A. 78, 5559–63.
Perry, S. V. (1955) Myosin adenosine triphosphatase.Meth. Enzym. 2, 582–8.
Prince, H. P., Trayer, H. R., Henry, G. D., Trayer, I. P., Dalgarno, D. C., Levine, B. A., Cary, P. D. &Turner, C. (1981) Proton nuclear-magnetic-resonance spectroscopy of myosin subfragment 1 isozymes.Eur. J. Biochem. 121, 213–9.
Suezaki, Y. &Gō, N. (1976) Fluctuations and mechanical strength of α-helices of polyglycine and poly(L-alanine).Biopolymers 15, 2137–53.
Sutoh, K. (1983) Mapping of actin-binding sites on heavy chain of myosin subfragment 1.Biochemistry 22, 1579–85.
Sutoh, K., Yamamoto, K. &Wakabayashi, T. (1984) Electron microscopic visualization of the SH1 thiol of myosin by the use of an avidin-biotin system.J. molec. Biol. 178, 323–39.
Suzuki, S. &Sugi, H. (1983) Extensibility of the myofilaments in vertebrate skeletal muscle as revealed by stretching rigor muscle fibers.J. gen. Physiol. 81, 531–46.
Takahashi, K. (1978) Topography of the myosin molecule as visualized by an improved negative staining method.J. Biochem., Tokyo 83, 905–8.
Tanaka, H., Sugi, H. &Hashizume, H. (1984) Effect of stretch on the equatorial X-ray diffraction pattern from frog skeletal muscle in rigor. InContractile Mechanism in Muscle (edited byPollack, G. H. andSugi, H.), pp. 203–5. New York: Plenum Press.
Tawada, K. &Kimura, M. (1984a) Cross-linking studies related to the location of the rigor compliance in glycerinated rabbit psoas fibers: Is the SII portion of the cross-bridge compliant? InContractile Mechanisms in Muscle (edited byPollack, G. H. andSugi, H.), pp. 385–96. New York: Plenum Press.
Tawada, K. &Kimura, M. (1984b) Stiffness of glycerinated rabbit psoas fibers in rigor: Filament-overlap relation.Biophys. J. 45, 593–602.
Toyoshima, C. &Wakabayashi, T. (1985) Threedimensional image analysis of the complex of thin filaments and myosin molecules from skeletal muscle. IV. Reconstruction from minimal- and high-dose images of the actin-tropomyosin-myosin subfragment-1 complex.J. Biochem., Tokyo 97, 219–43.
Tsong, T. Y., Karr T. &Harrington, W. F. (1979) Rapid helix-coil transitions in the S-2 region of myosin.Proc. natn. Acad. Sci. U.S.A. 76, 1109–13.
Ueno, H., Rodgers, M. E. &Harrington, W. F. (1983) Self-association of a high molecular weight subfragment-2 of myosin induced by divalent metal ions.J. molec. Biol. 168, 207–28.
Wagner, P. D. &Weeds, A. G. (1977) Studies on the role of myosin alkali light chains.J. molec. Biol. 109, 455–73.
Weber, A. &Murray, J. M. (1973) Molecular control mechanisms in muscle contraction.Physiol. Rev. 53, 612–73.
Weber, K. &Osborn, M. (1969) The reliability of molecular weight determinations by dodecyl sulfatepolyacrylamide gel electrophoresis.J. biol. Chem. 244, 4406–12.
Weeds, A. G. &Taylor, R. S. (1975) Separation of subfragment-1 isozymes from rabbit skeletal muscle myosin.Nature, Lond. 257, 54–6.
Winkelmann, D. A., Mekeel, H. &Rayment, I. (1985) Packing analysis of crystalline myosin subfragment-1: Implications for the size and shape of the myosin head.J. molec. Biol. 181, 487–501.
Yamamoto, K., Sekine, T. &Sutoh, K. (1984) Spatial relationship between SH1 and the actin binding site on myosin subfragment-1 surface.FEBS Lett. 176, 75–8.
Yamamoto, T. &Herzig, J. W. (1978) Series elastic properties of skinned muscle fibers in contraction and rigor.Pflügers Arch. 373, 21–4.
Yanagida, T. (1984) Angles of fluorescently labelled myosin heads and actin monomers in contracting and rigor strained muscle fiber. InContractile Mechanisms in Muscle (edited byPollack, G. H. andSugi, H.), pp. 397–411. New York: Plenum Press.
Young, D. M., Himmelfarb, S. &Harrington, W. F. (1965) On the structural assembly of the polypeptide chains of heavy meromyosin.J. biol. Chem. 240, 2428–36.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tawada, K., Kimura, M. Stiffness of carbodiimide-crosslinked glycerinated muscle fibres in rigor and relaxing solutions at high salt concentrations. J Muscle Res Cell Motil 7, 339–350 (1986). https://doi.org/10.1007/BF01753655
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01753655