Summary
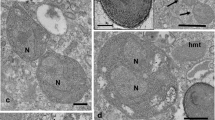
New intracellular bacteria were detected in the cytoplasm ofParamecium calkinsi andP. putrinum. Some of the bacteria were not evenly distributed in the cytoplasm of the host but were found in the center of the cell, eventually near the nuclei, but not in the cortex area, whereas another species was found in the cortex area. These peculiarities of intracellular bacteria localization in the host suggest that the conditions in various parts of the cytoplasm favor bacterial maintenance to different extent. Due to the results obtained by transmission electron microscopy and in situ hybridization using appropriate oligonucleotide probes, the bacteria, three or possibly four species, are Gram-negative and belong to the alpha-subgroup of proteobacteria. Bacteria from one stock ofP. calkinsi were found to be infectious for bacteria-free cells ofP. calkinsi andP. nephridiatum.
Similar content being viewed by others
References
Amann RI, Binder BJ, Oslon RJ, Chrisholm SW, Devereux R, Stahl DA (1990) Combination of 16S rRNA-targeted oligonucleotide probes with flow cytometry for analyzing mixed microbial population. Appl Environ Microbiol 56: 1219–1925
—, Springer N, Ludwig W, Görtz H-D, Scheifer K-H (1991) Identification in situ and phylogeny of uncultured bacteria endosymbionts. Nature 351: 161–164
Brigge T, Fokin SI, Brümmer F, Görtz H-D (1999) Molecular probes for localization of endosymbiotic bacteria in ciliates and toxic dinoflagellates. J Euk Microbiol 46: 11A
Clegg S, Gerlach GT (1987) Enterobacterial fimbriae. J Bacteriol 169: 934–938
Duguid JP (1968) The function of fimbriae. Arch Immunol Therap Exp 16: 173–188
Fokin SI (1989a) Bacterial endobionts of the ciliateParamecium woodruffi I: endobionts of the macronucleus. Cytologia (Sankt Petersburg) 31: 829–844
— (1989b) Bacterial endobionts of the ciliateParamecium woodruffi III: endobionts of the cytoplasm. Cytologia (Sankt Petersburg) 31: 964–970
—, Karpov SA (1995) Bacterial endocytobionts inhabiting the perinuclear space of protista. Endocyt Cell Res 11: 81–94
—, Sabaneyeva EV (1993) Bacterial endocytobionts of the ciliateParamecium calkinsi. Eur J Protistol 29: 390–395
—, Brigge T, Brenner J, Görtz H-D (1996)Holospora species infecting the nuclei ofParamecium appear to belong into two groups of bacteria. Eur J Protistol 32 Suppl 1: 19–24
Görtz H-D, Dieckmann J (1987)Leptomonas ciliatorum n. sp. in the macronucleus of hypotrich ciliate J Protozool 34: 259–265
Gromov BV (1985) Structure of bacteria. LGU, Leningrad
Heckmann K, Görtz H-D (1991) Prokaryotic symbionts of ciliates. In: Balows A, Truper HG, Dworkin M, Harder W, Scheifer K-H (eds) The prokaryotes, 2nd edn. Springer, Berlin Heidelberg New York Tokyo, pp 3865–3890
Moreno E (1998) Genome evolution within the alpha Proteobacteria: why do some bacteria not possess plasmids and others exhibit more than one different chromosome? FEMS Microbiol Rev 22: 255–275
Preer JR, Preer LB (1984) Endosymbionts of protozoa. In: Krieg NR (ed) Bergey's manual of systematic bacteriology, vol 1. Willams and Wilkins, Baltimore, pp 795–811
—, Preer LB, Jurand A (1974) Kappa and other endosymbionts inParamecium aurelia. Bacteriol Rev 38: 113–163
Preer LB (1969) Alpha, an infectious macronuclear symbiont ofParamecium aurelia. J Protozool 16: 570–578
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fokin, S.I., Sabaneyeva, E.V., Borkhsenious, O.N. et al. Paramecium calkinsi andP. putrinum (Ciliophora, Protista) harboring alpha-subgroup bacteria in the cytoplasm. Protoplasma 213, 176–183 (2000). https://doi.org/10.1007/BF01282155
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01282155