Abstract
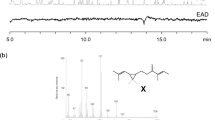
Hexamethylphosphoric triamide (HMPA) is a good solvent for the alkylation of terminal acetylenes; however, it has been proven to be a carcinogen towards rats and should be considered as a serious risk to man when a large-scale synthesis is involved. Thus the search for a safe alternative is important. The physical properties of 1,3-dimethyl-2-imidazolidinone (DMI) are similar to DMPU (1,3-dimethyl-2-oxo-hexahydropyrimidine) and HMPA, but the potential toxicological risk of DMI is less than DMPU and HMPA. When used in the alkylation of terminal acetylenes, DMI is comparable with HMPA, thus it is a good alternative solvent to the carcinogen HMPA in the alkylation because it provides a safer working environment than DMPU and HMPA.
Similar content being viewed by others
References
Ando, T., Yoshida, S., Tatsuki, S., andTakahashi, N. 1977. Sex attractants for male Lepidoptera.Agric. Biol. Chem. 41:1485–1492.
Barker, B.J., Rosenfarb, J., andCaruso, J.A. 1979. Ureas as solvents for chemical investigations.Angew, Chem. Int. Ed. Engl. 18:503–507.
Bengtsson, M., andLiljefors, T. 1988. DMPU: An alternative to HMPT in moth sex pheromone synthesis.Synthesis 00:250–252.
Brandsma, L. 1988. Preparative Acetylenic Chemistry. Elsevier, New York.
Brattesani, D.N., andHeathcock, C.H. 1973. A convenient procedure for the alkylation of acetylenes.Synth. Commun. 3:245–248.
Brown, M.M., Wassom, J.S., Mallino, H.V., Shelby, M.D., andVon Halle, E.S. 1979. Literature survey of bacterial, fungal, andDrosophila assay systems used in the evaluation of selected chemical compounds for mutagenic activity.J. Natl. Cancer Inst. 62:841–871.
Denmark, S.E., andAres, J.J. 1988. Stereoselective alkylation of chiral α-nitro keto imine dianions. Observations on the role of amide bases.J. Am. Chem. Soc. 110:4432–4434.
Green, N., Jacobson, M., Henneberry, T.J., andKishaba, A.N. 1967. Insect sex attractants. VI. 7-dodecen-1-ol acetates and congeners.J. Med. Chem. 10:533–535.
Henrick, C.A. 1977. The synthesis of insect sex pheromones.Tetrahedron 33:1845–1889.
Kochansky, J., Tette, J., Taschenberg, E.F., Cardé, R.T., Kaissling, K.E., andRoelofs, W.L. 1975. Sex pheromone of the moth,Antheraeapolyphemus.J. Insect. Physiol. 21:1977–1983.
Lien, E.J., andKumler, W.D. 1968. Dipole moments and pharmacological activity of cyclic ureas, cyclic thioureas, and the N,N′-dimethylated compounds.J. Med. Chem. 11:214–219.
Lo, C.C., Hung, M.D., Hwang, J.S., andHung, C.C. 1988. An improved method to synthesize the major component of the sex pheromone ofSpodoptera litura (F.).J. Chin. Agric. Chem. Soc. 26:536–544.
Maron, D.M., andAmes, B.N. 1983. Revised methods for theSalmonella mutagenicity test.Mutat. Res. 113:173–215.
Mukaiyama, T., Harada, T., andShoda, S.-I. 1980. An efficient method for the preparation of homoallylic alcohol derivatives by the reaction of allyl iodide with carbonyl compounds in the presence of stannous halide.Chem. Lett. 1507–1510.
Mukhopadhyay, T., andSeebach, D. 1982. 39. Substitution of HMPT by the cyclic urea DMPU as a cosolvent for highly reactive nucleophiles and bases.Helv. Chim. Acta 65:385–391.
Normant, H. 1967. Hexamethylphosphoramide.Angew. Chem. Int. Ed. 6:1046–1067.
Oi, R.,Shimakawa, C., andTakenaka, S. 1988. Ullmann ether synthesis in DMI. Preparation of m-phenoxybenzyl alcohol.Chem. Lett. 899–900.
Sakurai, H., andKondo, F. 1975. Chemistry of organosilicon compounds LXXX. Useful modifications in the preparation of trimethylsilylsodium and trimethylsilylpotassium.J. Organomet. Chem. 92: C46-C48.
Sakurai, H., andKondo, F. 1976. Chemistry of organosilicon compounds XC. The reaction of the trimethylchlorosilane-Hthium-aliphatic ketone mixture in 1,3-dimethyl-2-imidazolidinone.J. Organomet. Chem. 117:149–155.
Schmutz, J.F. 1978. Chronic health hazards: A national challenge.Chem. Eng. News 16:37–39.
Schwarz, M., andWaters, R.M. 1972. Insect sex attractants; XII. an efficient procedure for the preparation of unsaturated alcohols and acetates.Synthesis 567–568.
Seebach, D., Henning, R., andMukhopadhyay, T. 1982. Doubly deprotonated methyl 3-nitropropanoate, and acrylic ester d2-reagent.Chem. Ber. 115:1705–1720.
Sonnet, P.E. 1974. A practical synthesis of the sex pheromone of the pink bollworm.J. Org. Chem. 39:3793–3794.
Spangler, C.W.,Kjell, D.P.,Wellander, L.L., andKinsella, M.A. 1981. Methyltriphenoxyphosphonium iodide (MTPI): Induced dehydration and dehydrohalogenation in aprotic solvents.J. Chem. Soc. 2287–2289.
Tamaki, Y., Noguchi, H., andYushima, T. 1971. Two sex pheromones of the smaller tea tortrix: Isolation, identification, and synthesis.Appl. Entomol. Zool. 6:139–141.
Tamaki, Y., Noguchi, H., Sugic, H., Sato, R., andKariya, A. 1979. Minor components of the female sex-attractant pheromone of the smaller tea tortrix moth (Lepidoptera: Tortricidae): isolation and identification.Appl. Entomol. Zool. 14:101–113.
Warthen, D. 1968. Synthesis ofcis-9-tetradecen-1-ol acetate, the sex pheromone of the fall armyworm.J. Med. Chem. 11:371–373.
Warthen, D., andJacobson, M. 1968. Insect sex attractants. X. 5-Dodecen-1-ol acetates, analogs of the cabbage looper sex attractant.J. Med. Chem. 11:373–374.
You, B.Y. 1988. Unpublished result. Taiwan Agricultural Chemicals and Toxic Substances Research Institute. (TACTRI), Taiwan.
Zulstra, J.A., andVogel, E.W. 1988. The ratio of induced recessive lethals to ring-X loss has prognostic value in terms of functionality of chemical mutagens inDrosophila melanogaster.Mutat. Res. 201:27–38.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lo, C.C., Chao, P.M. Replacement of carcinogenic solvent HMPA by DMI in insect sex pheromone synthesis. J Chem Ecol 16, 3245–3253 (1990). https://doi.org/10.1007/BF00982095
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00982095