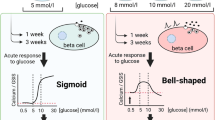
Abstract
The prolonged exposure of pancreatic islets and isolated beta cells to elevated glucose concentrations induces a state of unresponsiveness to glucose (desensitization). However, an increased sensitivity to glucose (detected by a shift to the left of the dose-response curve of glucose-induced insulin release) has been also reported after chronic exposure to glucose, making the overall response less comprehensible. In vitro models have many theoretical and practical advantages in better understanding the effects of the prolonged glucose stimulation; moreover, they are also suitable for studying the mechanisms responsible of the observed alterations. We have performed a time-course study of the effect of the exposure to glucose at high concentration on the secretory behaviour of beta cells. Rat pancreatic islets exposed for 30 min to high glucose (300 mg/dl) showed increased basal insulin secretion (175±29 vs 44±8 pg/islet (per 30 min;n=5,P<0.002) as the only difference from control islets (exposed to 100 mg/dl). After 3 h exposure to high glucose, also increased sensitivity to glucose was observed, as indicated by a shift to the left of the glucose dose-response curve (EC50 123±10 and 177±11 mg/dl, respectively;n=5,P<0.05). After 6 h exposure to high glucose, besides the two alterations already described, also a decrease in glucose-induced insulin release was observed (688±104 vs 1184±34 pg/islet per 30 min;n=5,P<0.01). We studied the mechanism responsible for these alterations and we found that the “supersensitivity” to glucose may be related to alterations in the “glucose-sensing” mechanism of beta cells, in particular in glucose phosphorylation. In contrast, in islets desensitized to glucose our data suggest that ion flux and consequent membrane potential changes play a key role in determining the secretory defect. Since a normal response to glyburide was observed, a proximal signal defect for closure of potassium channels is more likely than an intrinsic defect in the channel. In conclusion, our data show what the prolonged stimulation of beta cells with glucose at high concentration induces a series of distinct secretory abnormalities, with a pattern of response that leads first to increased sensitivity and then to decreased responsiveness to glucose.
Similar content being viewed by others
References
Grodsky GM, Bolaffi JL (1992) Desensitization of the insulinsecreting beta cell. J Cell Biochem 48: 3–11
Robertson RP (1989) Type II diabetes, glucose “non-sense” and islet desensitization. Diabetes 38: 1501–1505
Rossetti L, Giaccari A, De Fronzo RA (1990) Glucose toxicity. Diabetes Care 13: 610–630
Hoenig M, MacGregor LC, Matschinsky FM (1986) In vitro exhaustion of pancreatic β-cells. Am J Physiol 250: E502–E511
Purrello F, Vetri M, Gatta C, Gullo D, Vigneri R (1989) Effects of high glucose on insulin secretion by isolated rat islets and purified B-cells and possible role of glycosylation. Diabetes 38: 1417–1422
Kaiser N, Corcos AP, Sarel I, Cerasi E (1991) Monolayer culture of adult rat pancreatic islets on extracellular matrix: modulation of β-cell function by chronic exposure to high glucose. Endocrinology 129: 2067–2076
Eizirik DL, Korbutt GS, Hellerstrom C (1992) Prolonged exposure of human pancreatic islets to high glucose concentration in vitro impairs the β-cell function. J Clin Invest 90: 1263–1268
Leahy JL, Cooper HE, Deal DA, Weir GC (1986) Chronic hyperglycemia is associated with impaired glucose influence on insulin secretion. J Clin Invest 77: 908–915
Savage PJ, Bennion LJ, Flock EV, Nagulesperan M, Mott D, Roth J, Unger RH, Bennet PH (1979) Diet-induced improvement of abnormalities in insulin and glucagon secretion and in insulin receptor binding in diabetes mellitus. J Clin Endocrinol Metab 48: 999–1007
Kosaka K, Kuzuya T, Akanuma J, Hagura R (1980) Increase in insulin response after treatment of overt maturity onset diabetes is independent of the mode of treatment. Diabetologia 18: 23–28
Andrews WJ, Vasquez P, Nagulesperan M, Klimes I, Foley J, Unger RH, Reaven GM (1984) Insulin therapy in obese non insulin dependent diabetes induces improvements in insulin action and secretion that are maintained for two weeks after insulin withdrawn. Diabetes 33: 634–642
Robertson RP, Olson LK, Hui-Jian Z (1994) Differentiating glucose toxicity from glucose desensitization: a new message from the insulin gene. Diabetes 43: 1085–1089
Viitanen PV, Geiger PJ, Erickson-Viitanen S, Bessman SP (1984) Evidence for functional hexokinase compartmentation in rat skeletal muscle mitochondria. J Biol Chem 259: 9679–9686
Gots RE, Gorin FA, Bessman SP (1972) Kinetic enhancement of bound hexokinase activity by mitochondrial respiration. Biochem Biophys Res Commun 49: 1249–1255
Adams V, Bosch W, Hammerle Th, Brdiczka D (1988) Activation of low Km hexokinases in purified hepatocytes by binding to mitochondria. Biochim Biophys Acta 932: 195–205
Purrello F, Buscema M, Rabuazzo AM, Caltabiano V, Forte F, Vinci C, Vetri M, Vigneri R (1993) Glucose modulates glucose transporter affinity, glucokinase activity and secretory response in rat pancreatic β-cells. Diabetes 42: 199–205
Purrello F, Vetri M, Vinci C, Gatta C, Buscema M, Vigneri R (1990) Chronic exposure to high glucose and impairment of K+ channel function in perifused rat pancreatic islets. Diabetes 39: 397–399
Anello M, Rabuazzo AM, Degano C, Caltabiano V, Patanè G, Vigneri R, Purrello F (1996) Fast reversibility of glucose-induced desensitization in rat pancreatic islets: evidence for an involvement of ionic fluxes. Diabetes 45: 502–506
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Purrello, F., Rabuazzo, A.M., Anello, M. et al. Effects of prolonged glucose stimulation on pancreatic beta cells: from increased sensitivity to desensitization. Acta Diabetol 33, 253–256 (1996). https://doi.org/10.1007/BF00571559
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00571559