Summary
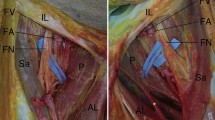
The sympathetic innervation in false tendons as a whole and the distribution of the terminal sympathetic nerve fibers in the conduction tissue in the bundle branches is unclear. Therefore, in the present study, false tendons and bundle branch regions of the bovine heart were examined using tyrosine hydroxylase (TH) immunohistochemistry and the glyoxylic acid induced catecholamine (CA) fluorescence method for demonstration of sympathetic nerve fibers. Acetylcholinesterase (AChE) histochemistry was also applied. Some of the nerve fascicles in the false tendons were found to contain large numers of sympathetic nerve fibers and such nerve fibers formed plexuses in the walls of arteries and arterioles in these structures. In both false tendons and bundle branches sympathetic nerve fibers 1) were non-homogeneously distributed in the conduction tissue, most regularly occurring in the channels of extracellular space that are present within the bundles of Purkinje fibres. and 2) showed the same pattern of distribution in relation to Purkinje fibre bundle surfaces as the AChE-positive nerve branches. The observations show that there is a substantial sympathetic innervation in false tendons. The final distribution of the nerve fibers in these structures and in the bundle branches are discussed in relation to what is known of tissue morphology and the occurrence of sympathetic nerve influences in these regions. In the present study, previous CA-fluorescence observations of a “marked” sympathetic innervation in bundle branch regions, in terms of the presence of sympathetic nerve fibers in nerve fascicles and vessel walls, were also corroborated by the application of TH-immunohistochemistry.
Similar content being viewed by others
References
Bojsen-Möller F, Tranum-Jensen J (1971) On nerves and nerve endings in the conducting system of the moderator band (septomarginal trabecula). J Anat 108:387–395
Cameron JS, Han J (1982) Effects of epinephrine on automaticity and the incidence of arrhythmias in Purkinje fibers surviving myocardial infarction. J Pharmacol Exp Ther 223:573–579
Canale E, Campbell GR, Uehara Y, Fujiwara T, Smolich JJ (1983a) Sheep cardiac Purkinje fibers: Configurational changes during the cardiac cycle. Cell Tissue Res 232:97–110
Canale E, Fujiwara T, Campbell GR (1983b) The demonstration of close nerve-Purkinje fibre contacts in false tendons of sheep heart. Cell Tissue Res 230:105–111
Carlsson L, Abrahamsson T, Almgren O (1986) Release of noradrenaline in myocardial ischemia—importance of local inactivation by neuronal and extraneronal mechanisms. J Cardiovasc Pharmacol 8:545–553
Dart AM, Schömig A, Dietz R, Mayer E, Kübler W (1984) Release of endogenous catecholamines in the ischemic myocardium of the rat. Part B: Effect of sympathetic nerve stimulation. Circ Res 55:702–706
De la Torre JC, Surgeon JW (1976) A methodologial approach to rapid and sensitive monoamine histofluorescence using a modified glyoxylic acid technique: the SPG method. Histochemistry 49:81–93
Ellison JP (1974) The adrenergic cardiac nerves of the cat. Am J Anat 139:209–225
Felten SY, Peterson RG, Shea PA, Besch Jr HR, Felten DL (1982) Effects of streptozotocin diabetes on the noradrenergic innervation of the rat heart: a longitudinal histofluorescence and neurochemical study. Brain Res Bull 8:593–607
Forsgren S (1986) The distribution of sympathetic nerve fibres in the AV node and AV bundle of the bovine heart. Histochem J 18:625–638
Forsgren S (1987) Marked sympathetic innervation in the regions of the bundle branches shown by catecholamine histofluorescence. J Mol Cell Cardiol 19:555–568
Friedman PL, Stewart JR, Fenoglio JJ Jr, Wit AL (1973) Survival of subendocardial Purkinje fibers after extensive myocardial infarction in dogs. Circ Res 33:597–611
Gilmour RF, Zipes DP (1984) Evidence for prejunctional and postjunctional antagonism of the sympathetic neuroeffector junction by acetylcholine in canine cardiac Purkinje fibers. J Am Coll Cardol 3:760–765
Gomori G (1952) Microscopic histochemistry—principles and practise. Chicago: University of Chicago Press, p 211
Jensen H, Holtet L, Hoen R (1978) Nerve-Purkinje fiber relationship in the moderator band of bovine and caprine heart. Cell Tissue Res 188:11–18
Karnovsky MJ (1964) The localization of cholinesterase activity in rat cardiac muscle by electron microscopy. J Cell Biol 23:217–232
Kent KM, Epstein SE, Cooper T, Jacobowitz DM (1974) Cholinergic innervation of the canine and human ventricular conducting system. Circulation 50:948–955
Lazzara R, El-Sherif N, Scherlag BJ (1973) Electrophysiological properties of canine Purkinje cells in one-day-old myocardial infarction. Circ Res 33:722–734
Levitt M, Spector S, Sjoerdsma A, Udenfriend S (1965) Elucidation of the rate limiting step in norepinephrine biosynthesis using the perfused guinea-pig heart. J Pharmac Exp Ther 148:1–8
Papka RE (1981) Development of innervation to the ventricular myocardium of the rabbit. J Mol Cell Cardiol 13:217–228
Reder RF, Danilo Jr P, Rosen MR (1984) Developmental changes in apha-adrenergic effects on canine Purkinje fiber automaticity. Dev Pharmacol Ther 7:94–108
Rosen MR, Hordof JP, Illvento JP, Danilo P (1977) Effects of adrenergic amines on electrophysiological properties and automaticity of neonatal and adult canine Purkinje fibers. Circ Res 40:390–400
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Forsgren, S. The distribution of terminal sympathetic nerve fibers in bundle branchès and false tendons of the bovine heart. Anat Embryol 177, 437–443 (1988). https://doi.org/10.1007/BF00304741
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00304741