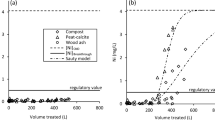
Abstract
In this investigation, the capacity of peat to treat two different landfill leachates was determined. Freundlich isotherms showed that, for the tested metals (Cd, Cr, Cu, and Pb), an increased time of contact increased the level of adsorption over the entire range of influent values studied. In addition, metal interaction may play an important role in the adsorption of metals from leachate, as the various metal ions compete for the available adsorption sites on the peat. Column studies using two leachates, one from a municipal refuse fill (Al, Ca, Cd, Fe, Mg, Na, and Pb), and one from a fill which receives mainly oil and fly ash (Al, Ca, Cr, Fe, Mg, Mn, Na, Pb, and V), were conducted to establish design parameters for full-scale design. The efficiency of treatment was evaluated as a function of factors important for developing filter design criteria. Further considerations were given to filter longevity and ease of replacement, as well as for the ultimate disposal of the peat from the filters. For the municipal leachate a compaction density of 0.12 g mL−1 gave the best results as clogging occurred at higher densities, whereas 0.18 g mL−1 provided the best removal for the industrial leachate. While peat is incapable of adequately removing metals to acceptable levels for direct discharge, peat can provide substantial removal as a pretreatment process at low hydraulic loadings. Desorption of up to 50% was found when deionized water was applied to spent columns so ultimate disposal of the peat would require a dry environment.
Similar content being viewed by others
References
Alesii, B. A., Fueller, W. H., and Boyle, M. V.: 1980, J. Env. Qual. 9, 119.
Asplund, D., Ekman, E., and Thun, R.: 1972, in Fourth Int. Peat Cong. Proc., Vol. 5, pp. 358–371.
Benefield, L. D., Judkins, J. F., and Weard, B. L.: 1982, Process Chemistry for Water and Wastewater Treatment, Prentice-Hall, Inc., Englewood Cliffs, NJ. pp. 510.
Cameron, R. D.: 1978, Can. J. Civil Eng. 5, 83.
Campbell, D. J. V., Parker, A., Rees, J. F., and Ross, C. A. M.: 1983, Waste Man. & Res. 1, 31.
Chian, E. S. K. and DeWalle, F. B.: 1976, J. Env. Eng. Div.: ASCE 102, 41.
Keenan, J. D., Steiner, R. L., and Fungaroli, A. A.: 1984, JWPCF 56, 27.
Lidkea, T. R.: 1974, ‘Treatment of Sanitary Landfill Leachate with Peat’, M. S. Thesis. University of British Columbia, 61 pp.
McLellan, J. K. and Rock, C. A.: 1986, J. Int. Peat Society 1, 1.
Rock, C. A., Brooks, J. L., Bradeen, S. B., and Structemeyer, R. A.: 1984, J. Env. Qual. 13, 518.
Rock, C. A., Greer, T. F., Fiola, J. W., and Woodard, F. E.: 1985, J. New Eng. Water Poll. Cont. Assoc. 19, 32.
Ruel, M., Chornet, S., Coupal, B., Aitcin, P., and Cossette, M.: 1973, in Radforth (ed.), Muskeg and the Northern Environment, pp. 221–246.
Schnitzer, M. and Khan, S. U.: 1978, ‘Soil Organic Matter’, Developments in Soil Science 8, Elsevier Scientific Publ. Co., Amsterdam.
Soltanpour, P. N., Jones Jr., J. B., and Workman, S. M.: 1982, in Page (ed.) Methods of Soil Analysis, Part 2, Second Edition, American Soc. Agronomy, Madison Wl.
Zhipei, Z., Junlun, Y., Zenghni, W., and Piya, C.: 1984, in Seventh Int. Peat Cong. Proc., Vol. 3, pp. 147–152.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McLellan, J.K., Rock, C.A. Pretreating landfill leachate with peat to remove metals. Water Air Soil Pollut 37, 203–215 (1988). https://doi.org/10.1007/BF00226492
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00226492