Abstract
Painful diabetic neuropathy (PDN) is a diabetes mellitus complication. Unfortunately, the mechanisms underlying PDN are still poorly understood. Adenosine triphosphate (ATP)-gated P2X7 receptor (P2X7R) plays a pivotal role in non-diabetic neuropathic pain, but little is known about its effects on streptozotocin (STZ)-induced peripheral neuropathy. Here, we explored whether spinal cord P2X7R was correlated with the generation of mechanical allodynia (MA) in STZ-induced type 1 diabetic neuropathy in mice. MA was assessed by measuring paw withdrawal thresholds and western blotting. Immunohistochemistry was applied to analyze the protein expression levels and localization of P2X7R. STZ-induced mice expressed increased P2X7R in the dorsal horn of the lumbar spinal cord during MA. Mice injected intrathecally with a selective antagonist of P2X7R and P2X7R knockout (KO) mice both presented attenuated progression of MA. Double-immunofluorescent labeling demonstrated that P2X7R-positive cells were mostly co-expressed with Iba1 (a microglia marker). Our results suggest that P2X7R plays an important role in the development of MA and could be used as a cellular target for treating PDN.
摘要
目 的
探讨嘌呤能离子通道型受体 7 (P2X7R) 在链脲佐菌素 (STZ) 诱导的 1 型糖尿病 (T1DM) 小鼠早期发生的机械痛性神经病变中的作用。
创新点
探讨脊髓 P2X7R 在 T1DM 产生痛性神经病变的早期阶段所起的作用, 将有望为研究糖尿病痛性神经病变药物提供新的靶点。
方 法
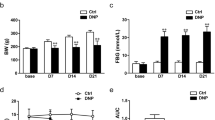
本实验采用健康雄性 C57BL 小鼠与 P2X7R KO 小鼠(体重 20∼30 克, 从 20 点至次日 8 点隔夜禁食 12 小时)为研究对象, 连续三天腹腔注射 STZ (浓度为 100 mg/kg), 从而制备 T1DM 动物模 型。如果空腹血糖 >11.1 mmol/L 且三周后小鼠机 械痛阈值明显下降, 则表示模型制备成功。在小鼠机械痛阈下降的对应时间点, 取腰段脊髓背角, 采用蛋白质印迹法 (western blot) 和免疫组化方法测定 P2X7R 的表达情况。同时, 在发生机械痛阈值下降的第 3 周时间鞘内给予其拮抗剂 A740003, 并进行机械刺激从而观察其痛阈值的变化。
结 论
STZ 诱导的 T1DM 动物模型在早期表现为显著的机械诱发痛, 并伴随脊髓背角 P2X7R 表达上调; 在痛阈下降期鞘内给予其拮抗剂 A740003 可抑制糖尿病小鼠的痛行为。 与腹腔注射 STZ 形成 T1DM 的 C57BL/6 小鼠相比, P2X7R 基因敲除的糖尿病小鼠早期机械痛阈值下降的时间延迟, 程度减轻。因此, 我们推测 P2X7R 可能参与了 STZ 诱导的糖尿病小鼠早期机械痛性神经病变。
Similar content being viewed by others
References
Bele T, Fabbretti E, 2015. P2X receptors, sensory neurons and pain. Curr Med Chem, 22(7):845–850. https://doi.org/10.2174/0929867321666141011195351
Berger JR, Choi D, Kaminski HJ, et al., 2013. Importance and hurdles to drug discovery for neurological disease. Ann Neurol, 74(3):441–446. https://doi.org/10.1002/ana.23997
Bernier LP, Ase AR, Séguéla P, 2018. P2X receptor channels in chronic pain pathways. Br J Pharmacol, 175(12):2219–2230. https://doi.org/10.1111/bph.13957
Burnstock G, 2014. Purinergic signalling: from discovery to current developments. Exp Physiol, 99(1):16–34. https://doi.org/10.1113/expphysiol.2013.071951
Cheng KI, Wang HC, Chuang YT, et al., 2014. Persistent mechanical allodynia positively correlates with an increase in activated microglia and increased P-p38 mitogen-activated protein kinase activation in streptozotocin-induced diabetic rats. Eur J Pain, 18(2):162–173. https://doi.org/10.1002/j.1532-2149.2013.00356.x
Chessell IP, Hatcher JP, Bountra C, et al., 2005. Disruption of the P2X7 purinoceptor gene abolishes chronic inflammatory and neuropathic pain. Pain, 114(3):386–396. https://doi.org/10.1016/j.pain.2005.01.002
Deuchars SA, Atkinson L, Brooke RE, et al., 2001. Neuronal P2X7 receptors are targeted to presynaptic terminals in the central and peripheral nervous systems. J Neurosci, 21(18):7143–7152. https://doi.org/10.1523/JNEUROSCI.21-18-07143.2001
Falk S, Schwab SD, Frøsig-Jorgensen M, et al., 2015. P2X7 receptor-mediated analgesia in cancer-induced bone pain. Neuroscience, 291:93–105. https://doi.org/10.1016/j.neuroscience.2015.02.011
Guo XX, Wang Y, Wang K, et al., 2018. Stability of a type 2 diabetes rat model induced by high-fat diet feeding with low-dose streptozotocin injection. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 19(7):559–569. https://doi.org/10.1631/jzus.B1700254
He XF, Wei JJ, Shou SY, et al., 2017. Effects of electroacupuncture at 2 and 100 Hz on rat type 2 diabetic neuropathic pain and hyperalgesia-related protein expression in the dorsal root ganglion. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 18(3):239–248. https://doi.org/10.1631/jzus.b1600247
Huang ZX, Lu ZJ, Ma WQ, et al., 2014. Involvement of RVM-expressed P2X7 receptor in bone cancer pain: mechanism of descending facilitation. Pain, 155(4):783–791. https://doi.org/10.1016/j.pain.2014.01.011
Illes P, Khan TM, Rubini P, 2017. Neuronal P2X7 receptors revisited: do they really exist? J Neurosci, 37(30):7049–7062. https://doi.org/10.1523/JNEUROSCI.3103-16.2017
Javed S, Alam U, Malik RA, 2015. Burning through the pain: treatments for diabetic neuropathy. Diabetes Obes Metab, 17(12):1115–1125. https://doi.org/10.1111/dom.12535
Jiang K, Zhuang Y, Yan M, et al., 2016. Effects of riluzole on P2X7R expression in the spinal cord in rat model of neuropathic pain. Neurosci Lett, 618:127–133. https://doi.org/10.1016/j.neulet.2016.02.065
Jiang TF, Hoekstra J, Heng X, et al., 2015. P2X7 receptor is critical in α-synuclein-mediated microglial NADPH oxidase activation. Neurobiol Aging, 36(7):2304–2318. https://doi.org/10.1016/j.neurobiolaging.2015.03.015
Kobayashi K, Takahashi E, Miyagawa Y, et al., 2011. Induction of the P2X7 receptor in spinal microglia in a neuropathic pain model. Neurosci Lett, 504(1):57–61. https://doi.org/10.1016/j.neulet.2011.08.058
Liu SM, Zou LF, Xie JY, et al., 2016. LncRNA NONRATT021972 siRNA regulates neuropathic pain behaviors in type 2 diabetic rats through the P2X7 receptor in dorsal root ganglia. Mol Brain, 9:44. https://doi.org/10.1186/s13041-016-0226-2
Liu WJ, Ao QY, Guo QL, et al., 2017. miR-9 mediates CALHM1-activated ATP-P2X7R signal in painful diabetic neuropathy rats. Mol Neurobiol, 54(2):922–929. https://doi.org/10.1007/s12035-016-9700-1
Lu WN, Albalawi F, Beckel JM, et al., 2017. The P2X7 receptor links mechanical strain to cytokine IL-6 up-regulation and release in neurons and astrocytes. J Neurochem, 141(3):436–448. https://doi.org/10.1111/jnc.13998
Mehta N, Kaur M, Singh M, et al., 2014. Purinergic receptor P2X7: a novel target for anti-inflammatory therapy. Bioorg Med Chem, 22(1):54–88. https://doi.org/10.1016/j.bmc.2013.10.054
Miras-Portugal MT, Sebastian-Serrano A, de Diego García L, et al., 2017. Neuronal P2X7 receptor: involvement in neuronal physiology and pathology. J Neurosci, 37(30):7063–7072. https://doi.org/10.1523/JNEUROSCI.3104-16.2017
Moore RA, Wiffen PJ, Derry S, et al., 2011. Gabapentin for chronic neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev, 3:CD007938. https://doi.org/10.1002/14651858.CD007938.pub2
Ni GL, Cui R, Shao AM, et al., 2017. Salidroside ameliorates diabetic neuropathic pain in rats by inhibiting neuroinflammation. J Mol Neurosci, 63(1):9–16. https://doi.org/10.1007/s12031-017-0951-8
Ochi-Ishi R, Nagata K, Inoue T, et al., 2014. Involvement of the chemokine CCL3 and the purinoceptor P2X7 in the spinal cord in paclitaxel-induced mechanical allodynia. Mol Pain, 10:53. https://doi.org/10.1186/1744-8069-10-53
Pan Q, Li QM, Deng W, et al., 2018. Prevalence of and risk factors for peripheral neuropathy in Chinese patients with diabetes: a multicenter cross-sectional study. Front Endocrinol (Lausanne), 9:617. https://doi.org/10.3389/fendo.2018.00617
Pop-Busui R, Boulton AJM, Feldman EL, et al., 2017. Diabetic neuropathy: a position statement by the American Diabetes Association. Diabetes Care, 40(1):136–154. https://doi.org/10.2337/dc16-2042
Sánchez-Nogueiro J, Marín-Garcia P, Bustillo D, et al., 2014. Subcellular distribution and early signalling events of P2X7 receptors from mouse cerebellar granule neurons. Eur J Pharmacol, 744:190–202. https://doi.org/10.1016/j.ejphar.2014.10.036
Tsuda M, Ueno H, Kataoka A, et al., 2008. Activation of dorsal horn microglia contributes to diabetes-induced tactile allodynia via extracellular signal-regulated protein kinase signaling. GLIA, 56(4):378–386. https://doi.org/10.1002/glia.20623
Vinik A, Emir B, Cheung R, et al., 2013. Relationship between pain relief and improvements in patient function/quality of life in patients with painful diabetic peripheral neuropathy or postherpetic neuralgia treated with pregabalin. Clin Ther, 35(5):612–623. https://doi.org/10.1016/j.clinthera.2013.03.008
Wang DM, Couture R, Hong YG, 2014. Activated microglia in the spinal cord underlies diabetic neuropathic pain. Eur J Pharmacol, 728:59–66. https://doi.org/10.1016/j.ejphar.2014.01.057
Wu B, Peng LC, Xie JY, et al., 2017. The P2X7 receptor in dorsal root ganglia is involved in HIV gp120-associated neuropathic pain. Brain Res Bull, 135:25–32. https://doi.org/10.1016/j.brainresbull.2017.09.006
Yao ZY, Chen WB, Shao SS, et al., 2018. Role of exosome-associated microRNA in diagnostic and therapeutic applications to metabolic disorders. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 19(3):183–198. https://doi.org/10.1631/jzus.B1600490
Ying YL, Wei XH, Xu XB, et al., 2014. Over-expression of P2X7 receptors in spinal glial cells contributes to the development of chronic postsurgical pain induced by skin/muscle incision and retraction (SMIR) in rats. Exp Neurol, 261:836–843. https://doi.org/10.1016/j.expneurol.2014.09.007
Zhang PA, Xu QY, Xue L, et al., 2017. Neonatal maternal deprivation enhances presynaptic P2X7 receptor transmission in insular cortex in an adult rat model of visceral hypersensitivity. CNS Neurosci Ther, 23(2):145–154. https://doi.org/10.1111/cns.12663
Author information
Authors and Affiliations
Contributions
Cheng-ming NI was responsible for drafting the manuscript, analysis and interpretation of data. He-ping SUN collected and analyzed the data. Xiang XU, Bing-yu LING, and Hui JIN contributed analysis of data and manuscript preparation. Yu-qiu ZHANG and Zhi-qi ZHAO helped perform the analysis with constructive discussions. Hong CAO and Lan XU contributed the conception and design of the current study. All authors have read and approved the final manuscript. Therefore, all authors had full access to all the data in the study and take responsibility for the integrity and security of the data.
Corresponding authors
Ethics declarations
Cheng-ming NI, He-ping SUN, Xiang XU, Bing-yu LING, Hui JIN, Yu-qiu ZHANG, Zhi-qi ZHAO, Hong CAO, and Lan XU declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
Additional information
Project supported by the National Natural Science Foundation of China (Nos. 81771208 and 81971043), the Health and Family Planning Commission of Wuxi (No. YGZXM1406), the Wuxi Municipal Bureau on Science and Technology (No. CSE31N1614), the Fundamental Research Fund of Wuxi People’s Hospital (No. RKA201720), and the Technology for Social Development Project of Kunshan (No. KS1539), China
Rights and permissions
About this article
Cite this article
Ni, Cm., Sun, Hp., Xu, X. et al. Spinal P2X7R contributes to streptozotocin-induced mechanical allodynia in mice. J. Zhejiang Univ. Sci. B 21, 155–165 (2020). https://doi.org/10.1631/jzus.B1900456
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1900456