Abstract
Background
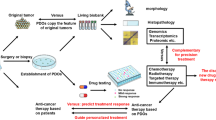
Organoids are three-dimensional in vitro models of human disease developed from benign and malignant gastrointestinal tissues with tremendous potential for personalized medicine applications. We sought to determine whether gastric cancer patient-derived organoids (PDOs) could be safely established from endoscopic biopsies for rapid drug screening.
Methods
Patients underwent esophagogastroduodenoscopy (EGD) for surveillance or staging and had additional forceps biopsies taken for PDO creation. Cancer tissues from operative specimens were also used to create PDOs. To address potential tumor heterogeneity, we performed low-coverage whole-genome sequencing of endoscopic-derived PDOs with paired surgical PDOs and whole-tumor lysates. The stability of genomic alterations in endoscopic organoids was assessed by next-generation sequencing and nested polymerase chain reaction (PCR) assay. The feasibility and potential accuracy of drug sensitivity screening with endoscopic-derived PDOs were also evaluated.
Results
Gastric cancer PDOs (n = 15) were successfully established from EGD forceps biopsies (n = 8) and surgical tissues (n = 7) from five patients with gastric adenocarcinoma. Low-coverage whole-genomic profiling of paired EGD and surgical PDOs along with whole-tumor lysates demonstrated absence of tumor heterogeneity. Nested PCR assay identified similar KRAS alterations in primary tumor and paired organoids. Drug sensitivity testing of endoscopic-derived PDOs displayed standard dose–response curves to current gastric cancer cytotoxic therapies.
Conclusions
Our study results demonstrate the feasibility of developing gastric cancer PDOs from EGD biopsies. These results also indicate that endoscopic-derived PDOs are accurate surrogates of the primary tumor and have the potential for drug sensitivity screening and personalized medicine applications.





Similar content being viewed by others
References
Prasad V, Mailankody S. Research and development spending to bring a single cancer drug to market and revenues after approval. JAMA Intern Med 2017; 177:1569–1575.
Bartfeld S, Clevers H. Organoids as model for infectious diseases: culture of human and murine stomach organoids and microinjection of Helicobacter pylori. J Vis Exp 2015; 105. https://doi.org/10.3791/53359.
Sato T, Vries RG, Snippert HJ, et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 2009; 459:262–265.
Boj SF, Hwang C, Baker LA, et al. Organoid models of human and mouse ductal pancreatic cancer. Cell 2015; 160:324–338.
van de Wetering M, Francies HE, Francis JM, et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 2015; 161:933–945.
Andersson-Rolf A, Zilbauer M, Koo BK, Clevers H. Stem cells in repair of gastrointestinal epithelia. Physiology 2017; 32:278–289.
Tiriac H, Bucobo JC, Tzimas D, et al. Successful creation of pancreatic cancer organoids by means of EUS-guided fine-needle biopsy for personalized cancer treatment. Gastrointest Endosc 2018; 87(6):1474–1480.
Pauli C, Hopkins BD, Prandi D, et al. Personalized in vitro and in vivo cancer models to guide precision medicine. Cancer Discov 2017; 7:462–477.
Barker N, Huch M, Kujala P, et al. Lgr5+ve stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell 2010; 6:25–36.
Wilhelm F, Böger C, Krüger S, Behrens HM, Röcken C. Troy is expressed in human stomach mucosa and a novel putative prognostic marker of intestinal type gastric cancer. Oncotarget 2017; 8:50557–50569.
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014; 513:202–209.
Vlachogiannis G, Hedayat S, Vatsiou A, et al. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science 2018;359:920–926.
Dedhia PH, Bertaux-Skeirik N, Zavros Y, Spence JR. Organoid models of human gastrointestinal development and disease. Gastroenterology 2016; 150:1098–1112.
Sato T, Clevers H. Snapshot: growing organoids from stem cells. Cell 2015; 161:1700–1700e1.
Hidalgo M, Amant F, Biankin AV, et al. Patient-derived xenograft models: an emerging platform for translational cancer research. Cancer Discov 2014; 4:998–1013.
Weeber F, Ooft SN, Dijkstra KK, Voest EE. Tumor organoids as a pre-clinical cancer model for drug discovery. Cell Chem Biol 2017; 24:1092–1100.
Acknowledgment
We would like to thank Drs. Hans Clevers and Georg Busslinger at the Hubrecht Institute for their instruction on developing organoids. These studies were presented in part at 2018 SSO Annual Symposium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None of the authors has a conflict of interest to disclose.
Rights and permissions
About this article
Cite this article
Gao, M., Lin, M., Rao, M. et al. Development of Patient-Derived Gastric Cancer Organoids from Endoscopic Biopsies and Surgical Tissues. Ann Surg Oncol 25, 2767–2775 (2018). https://doi.org/10.1245/s10434-018-6662-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6662-8