Abstract
Under conditions of intense anthropogenic impact, the territory of modern Donbass is geochemically contrasting environment. Concentrations of rare earth elements (Sc, La, Ce, Nd, Sm, Eu, Tb, Dy, Yb) in the natural ecosystems of the Northern Azov region (Central Donbass) were determined by indicator plants using neutron activation analysis. An open landscape experiment was carried out using moss Ceratodon purpureus (Hedw.) Brid. which has a high information response in the assessment of technogenic impacts. It allowed to establish the levels of regional contamination with rare earth elements, to identify correlation groups of related processes in the structural and functional features of indicator. The identified localities of geochemical heterogeneities in ecotopes are responsible for the adaptation of plants under stressful conditions. The most informative structural units of plants indicating the contamination with rare earth elements are abnormalities in the structure (terates, teratomorphs), violation of the morphogenesis processes of the embryonic apparatus of plants and areas of vegetative organs. Correlation analysis and principle component analysis were applied to reveal the relationship between the elements and abnormalities in the structure of plants. Relative accumulation factor, contamination factor and enrichment factor were calculated to evaluate the level of environment pollution and to identify the origin of elements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Neutron activation analysis is a modern technique, which allows performing environmental diagnostics [1, 2] and environmental expertise [3–5]. High-precision data about the state of the environment are of great demand especially in the regions where the level of anthropogenic load is very high [6, 7] and current state of pollution levels have not been previously established because of the lack of regular ecological surveys, danger or risk during periods of active social and military conflicts [8–10]. Such a region in Eastern Europe is the territory of modern Central Donbass (the natural geographical zone of the Northern Azov region). Therefore, organization of environmental surveys within the monitoring network and using proven indicator systems [11] have determined the time, space and tools for conducting a diagnostic experiment in this region.
Earlier, the geochemical contrast in the basic elements-pollutants that are characterized by technophilic properties and are typical for industrial activity [12–14] was shown. There is a strong opinion that rare earth elements (REEs) are involved in the biogeochemical cycle within background concentrations [15] and are not associated with sharp anthropogenic disturbances in landscapes [16]. On the other hand, high concentrations can cause inhibitory effects and ecological risks, such as reduced biogeochemical cycling and macrofauna diversity [17]. It should be mentioned that level of REEs can be high even in the areas where industrial development complex is not presented due to influence of industries located in the near vicinity [18]. Presence of high concentrations of REEs in mining, urban and industrial areas results in moderate to high ecological risk, however, the health risk of REEs for human beings are usually negligible [17, 19, 20].
Therefore, the main task of the study was to establish possible diagnostic properties of REEs in connection with the existing anthropogenic transformations on the territory of the Donetsk economic region. Since the nature of morphogenetic transformations in indicator plants (terates, abnormalities, somatic mutations, morphogenetic and phenotypic plasticity) remains an open question [21, 22], there is a need for a detailed study of those causes that were not previously taken into account. Previously, biological indicators were used by several research groups to assess the level of air pollution with REEs [23–25].
MATERIALS AND METHODS
Monitoring Network
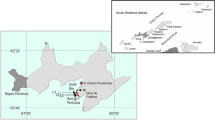
Moss specimens Ceratodon purpureus (Hedw.) Brid were sampled in the buffer zone of the Republican Landscape Park “Donetsky Kryazh”. In November 2018, the samples of live moss were transplanted to the test plots (Fig. 1). The choice of the exposure sites is explained in previously published papers [12, 26]. Among 113 monitoring sites, 24 model trial areas [12, 13] were selected in a certain pattern and according to the existing recommendations [27, 28] in order to get the most complete picture of the environmental situation on the territory of Central Donbass. Sampling sites are characterized by the same natural and climatic characteristics, geobotanical description of the terrain, and uniform landscape units [29, 30]. This arrangement of monitoring network nodes allows visualization—detailed geostrategic and cartographic analysis, as well as to establish cause-and-effect relationships and patterns in the development of natural ecosystems [31]. The authors had developed a project in GIS ArcView 10.4, which allowed to link 113 nodes (trial areas) of the monitoring network with a common information center with a geolocation accuracy of ±5 m. The mechanism of constructing the observation network, including the formation of the base part on topographic maps of 1 : 10 000 and 1 : 100 000 scaling was described earlier [26]. The WGS 1984 Web Mercaror public coordinate system and the Spatial Analyst and 3D Analyst modules were used.
Indicator Plants in the Experiment
For ingredient analysis in order to establish the regional background for rare earth elements, moss species Ceratodon purpureus (Hedw.) Brid. was used [12]. The purpose of the moss transplants growth experiments is to use a single population of the indicators growing on the territory of the reserve in the zone of minimal anthropogenic impact. Moss samples were planted at monitoring sites, grown for 6 months and further used for elemental analysis. Besides, structural and functional features of indicator plant species at exposure sites were established [14, 30, 32–34]. For the analytical program, coefficients and indices for the following parameters: IETp—index of embryonic teratogenesis (frequency of occurrence of terat variants of plant embryos); IMHp—index of morphological heterogeneity (the total sum of abnormalities in the structure of the leaf, shoot and inflorescences for all indicator species on the same accounting platform); TNm—total number of mosses (species diversity of mosses); GTm—general teratogenesis of mosses (frequency of occurrence of moss terates for one trial area); PDI—pollen deformation index (percentage of atypical pollen grains); OTp—overall teratogenicity (characterization of the terrain by the frequency of occurrence of atypical structures of mosses and flowering plants at a specific monitoring point); OTvp—overall teratogenicity of the vegetative sphere (frequency of occurrence of abnormalities in the structure of plants for vegetative organs); OTgp—teratogenicity of the generative sphere (abnormalities in the generative sphere of indicator plants) were calculated. The paper presents the parameters of the state of plants by flowering plants: Berteroa incana (L.) DC., Capsella bursa-pastoris (L.) Medik., Centaurea diffusa Lam., Cichorium intybus L., Diplotaxis muralis (L.) DC., Echium vulgare L., Plantago lanceolata L., Plantago major L., Reseda lutea L., Senecio vulgaris L., Tanacetum vulgare L., Tragopogon major Jacq., Tripleurospermum inodorum (L.) Sch. Bip. The method of obtaining ecological scales and establishing the anatomical and morphological structure of plants was described in previous publications [14, 32, 35] on the basis of existing methods in structural botany [36, 37].
Neutron Activation Analysis (NAA)
Moss elemental composition was determined using neutron activation analysis on the REGATA facility of the IBR-2 reactor (JINR, Dubna, Russia). The content of Dy was determined by samples irradiation for 3 min at a thermal neutron flux of 1.2 × 1012 n cm–2 s–1 and measured for 15 min immediately after irradiation. To determine the content of elements with long-lived isotopes: Sc, La, Ce, Nd, Sm, Eu, Tb, and Yb samples were irradiated for 4 days at a neutron flux of 1.1 × 1011 cm–2 s–1. Gamma spectra of induced activity were obtained after 4 and 20 days using three Canberra HPGe detectors with an efficiency of 55–100% and resolution of 1.8–2.0 at 1332 keV 60Co total-absorption peak [38, 39].
Spectra analysis was performed using Genie2000 software by Canberra company, while calculation of the metal content was carried out using the software “Concentration” developed in FLNP. Quality control of the analytical measurements was ensured by simultaneous irradiation of certified reference materials: NIST 1633c (Trace Elements in Coal Fly Ash), NIST 1632c (Trace Elements in Coal (Bituminous), BCR-667 (Estuarine Sediment) and FFA-1 (Fine Fly Ash). The recovery of the elements from the reference material ranged from 97 to 102% (Table 1).
Assessment of the State of Ecosystems and Their Connections
The basic statistical analysis and correlation analysis were performed using Origin Pro 2018 (Northampton, Massachusetts, USA) and Statistica 12 (StatSoft, Tulsa, OK, USA). Principal component analysis (PCA) was carried out in R.
The relative accumulation factor (RAF), contamination factor (CF) and Enrichment Factor (EF) were calculated to quantify the level of environment pollution.
The RAF, used to assess the element uptake by the mosses, was calculated using the formula given below:
where Ci is the value of the element concentration (mg/kg) in exposed moss, C0 is the initial element concentration (mg/kg) in moss before exposure.
RAF values higher than 0.5 indicated insignificant elemental enrichment in the moss, and values greater than one indicated significant elemental enrichment.
The CF defined as the ratio between the measured concentration (Ci) and the background value of metal i (Ci background) were calculated for each of the sites:
The contamination factor is categorized as follows: CFi < 1—no contamination factor; 1 ≤ CFi < 2—suspected contamination factor; 2 ≤ CFi ˂ 3.5—slight contamination factor; 3.5 ≤ CFi < 8—moderate contamination factor; 8 ≤ CFi < 27 severe contamination factor; CFi ≥ 27 extreme contamination factor [40].
The EF, a means of quantifying the enrichment of a potentially contaminant-derived element in an environmental sample relative to a user-defined background composition [41], was calculated according to the following equation:
where Mx and Alx are the soil sample concentrations of the heavy metal and Al, while Mb and Alb are their background concentrations. Upper continental crust values were used as reference values [42].
The EF for each trace metal could be categorized into five levels of pollution. EF < 2 represents zero or minimal enrichment, suggestive of zero or minimal pollution; 2 ≤ EF < 5 indicates moderate pollution; 5 ≤ EF < 20 indicates significant pollution; 20 ≤ EF < 40 indicates strong pollution and EF ≥ 40 indicates extreme pollution [43].
The characteristics of structural polymorphism of plants were presented in a cartographic equivalent to understand the distribution of data throughout the territory of Donbass.
The sampling map was built in the QGIS geographic information system. Layers from OpenStreetMap (OSM) were used for construction.
RESULTS AND DISCUSSIONS
According to NAA results the cases of sharp changes in the concentrations of rare earth elements in Donbass were observed (Fig. 2, Table S1). Such cases are typical for territories with a high level of anthropogenic transformation of natural ecosystems.
According to the obtained results the highest concentrations of the determined elements are characteristic for the exposure sites affected by industrial and urbanized objects. In the most of the cases, both pollution sources coincided geographically, since industrial enterprises are usually city-forming centers, it has historically developed on the territory of Donbass since the beginning of mining.
Thus, the highest concentrations of Sc (8.1 mg/kg), La (30.4 mg/kg), Ce (61 mg/kg), Nd (29 mg/kg), Sm (5.1 mg/kg), Eu (0.89 mg/kg), Tb (0.59 mg/kg), Dy (3.3 mg/kg), and Yb (1.9 mg/kg) were determined in mosses exposed in the zone of influence of the of industrial enterprises: Debaltsevo plant of metallurgical engineering, Uglegorsk thermal power plant (site 1), Voroshilovsky District in the Donetsk, impact of the Makeevka Metallurgical Plant (site 13); Leninsky District in the Donetsk—mining and metallurgical enterprises (site 19); Kirovsky District in the Donetsk—mining and processing enterprises (site 20). Also, high concentrations of rare earth elements were found at places subjected to high anthropogenic transformation and pollution as a result of hostilities: Peski settlement, Yasinovatskaya agglomeration (site 11); Spartak settlement, Yasinovatskaya agglomeration (site 12). Sites 4 and 5 are located in the exposure zone of a large metallurgical plant in Yenakievo, however, the ingredient analysis of mosses it was possible to perform at a significant (more than 8 km) distance from the sources of emissions. Thus, a high concentration of Sc (6.3 mg/kg) was recorded in mosses exposed at site 5. This fact proves a relatively small area of contamination with rare earth elements of technogenic origin. Therefore, it is recommended to conduct examination of pollution using bioindication no further than 1.5–2 km from the source of pollution. This primarily concerns are the cities of Donetsk, Makeevka, Enakievo, Gorlovka, Yasinovataya and Zugres.
The lowest concentrations of rare earth elements were determined at sites 7, and 23 (Zuevka urban-type settlement—Zuevka Landscape Park; Petrovsky District, Donetsk city—recreation zone).
Based on the obtained data it can be concluded that conditions of biogeochemical contrast have formed on the territory of Central Donbass [13]. This had previously been established for technophilic elements in the Northern Azov region [12].
RAF values calculated for moss samples indicate in most cases, accumulation of elements in comparison with control (Fig. 3). Thus, no accumulation or slight accumulation of Sc was observed at sites 18, 22 and 23, while high accumulation of Sc was characteristic for sites 1 (3.3) and 20 (3.0). For other rare earth elements almost the same pattern was observed, the lowest accumulation of the elements was observed at sites 18, 22, 23 for all elements, site 5 for Dy and Eu, and sites 3, 6, 7, 9, 10, 15, 17 for Dy. The RAF values for mosses exposed at sites 4, 13–15 lied between 1 and 2, while at other sites they were higher than 2. In general, the mean RAF values changed in the following order: Ce > La > Sm > Sc > Eu = Tb = Yb > Dy. This sequence reflects the pattern and intensity of the particular rare earth elements involvement of in biogeochemical cycles. This is important for establishing a cause-and-effect relationship in the diagnosis of plant communities, landscapes and local ecosystem functioning. Among rare earth elements, Ce, La and Sm are of the greatest importance in migration processes, whereas Dy is of the lowest. The increased concentrations of the studied elements correspond to territories located in the area affected by metallurgical plants and non-ferrous metal processing enterprises.
Mining and processing of alkaline rocks, coal combustion, oil refining, disposal of medical and electrical equipment are important sources of rare earth elements emission [44]. Coal fly ash is one of the largest industrial waste streams in the world, which contains significant concentrations of rare earth elements [45]. The carbonate rocks and alkaline igneous rocks are the leading natural sources of REEs [46].
Consequently, the primary cause of emissions from coal mining and combustion are superimposed the results of metallurgical activities in Donbass: the preparation of parts for mechanical engineering, microelectronics, smelting of many types of steel and fittings for building materials in Donetsk, Makeevka and Khartsyzsk. In Albania [23] and North Macedonia [24], wind-blowing metal-enriched soils was considered as the main sources of REEs emissions. In case of the present study the level of REEs was not influenced by geology since the 6-month growth of bryophytes is completely geochemically dependent on the level of elements in the air and precipitations. High levels of REEs were determined in the moss samples collected in certain regions of Serbia characterized by the extensive exploitation of coal and mineral deposits, and their further manufacturing or combustion [25].
The correlation analysis was performed to reveal the relationship between the elements and to associate them with sources of emission (Fig. 4). Strong positive correlation was obtained between all determined elements, with r values higher than 0.75. Thus, it can be suggested that they have a common source of emission. Coal mining and processing can be considered as an important source of rare earth elements emissions in the atmosphere. The distribution of elements in coal (and in rocks) is predominantly controlled by REE-bearing trace phases such as monazite (Ce,La, Nd,Th)PO4), allanite (Ce,Ca,Y)2(Al,Fe3+)3(SiO4)3(OH)), rhabdophane (Ce,La,Y)PO4·H2O), florencite (CeAl3(PO4)2(OH)6), and Ce–Nd-bearing carbonates [47].
Therefore, in the case of analysis of the content of rare earth elements in the biosubstrates of indicator plants, significant exceedances of the background geochemical load on ecosystems was observed. According to these data, geochemical traces of a high level of technogenic load on natural systems at coke-chemical production enterprises and places of open waste rock storage areas (slagheaps) have been established: Voroshilovsky and Leninsky districts of Donetsk, Tsentralnogorodskoy district of Makeevka and western districts of Enakievo.
The enrichment factor was applied to differentiate the sources of elements (anthropogenic or natural). EFs close to 1 indicate a crustal origin, whereas metals with values greater than 2 are considered to have anthropogenic source [48]. Based on the EF values between 1 and 3 obtained for the sampling sites, the level of contamination can be characterized as uncontaminated to moderately contaminated (Fig. 5). EF values for Sc and Dy (except site 8) at all exposure sites were lower than 2.0, that suggest that it is a lithophilic element. EF values for La and Ce at sites 1, 20 and 22 were higher than 2.0, while at other sites here were no enrichment in these elements. For sites 20 and 22 EFs values high than 2.0 were obtained for Sm, Eu, Tb, Yb and Nd, pointing at mentioned sites moderate pollution. Moderate pollution with Sm, Eu, Yb and Nd was characteristic for sites 1, 3, 6–10, 15 and 17.
The CFs of all determined elements for sites 18, 22 and 23 were between 1 and 2, indicating suspected contamination. Slight contamination was characteristic for sites 2–10, 14–17, 19 and 21, while sites 1, 11–13 and 20 were subjected to moderate pollution (Fig. 6).
Principal component analysis allowed to identify, which abnormalities in the structure of plants (Table S2) are associated with rare earth elements. On the PCA biplots presented in Fig. 7, the component weights for the two main components are shown, which allows comparing the distances between the variables.
The strong correlation was observed between all determined rare earth elements and two indices IETp and IMHp. The main contributors to this component are sites 20, 1, 12, and 13. A strong correlation of ТТp, TTgp and GТm was observed for samples exposed at sites 2 and 4. The studied rare earth elements have a high positive correlation with each other and are concomitant agents for the main pollutants, which means they also have indicative properties in the diagnosis of the state of ecosystems.
Of all the botanical criteria in the structure of plants, the IETp and IMHp indices have the greatest positive correlation primarily with Sc and Ce. This pattern is explained by the greater physiological activity of protein structures in the embryonic apparatus of the plant and the growing points (meristems) of shoots and inflorescences of indicator plants (Berteroa incana, Capsella bursa-pastoris, Cichorium intybus, Plantago major, Reseda lutea, Tragopogon major, Tripleurospermum inodorum). Sc and Ce elements apparently have a significant impact on changing the normal development of plant organism growth processes or are concomitant of the main pollutants in an industrially developed environment. With such patterns, the survival strategy of species can also change: typical native species behave like weeds (form more seeds and dissemination elements), and weeds feel like species that historically existed here (form stable populations in disturbed habitats). Such conclusions were made on the basis of long-term field observations. The obtained data always need to be explained, for example, by the conducted ingredient analysis of the main pollutants in the indicative phytosubstrates. Additional pollution by La, Sm and Tb enhances the effect of total mineral pollution of the plant organism and stimulates the manifestations of more terat forms of plants.
In the experiment, 8 indices of plant condition were used, 8 of which determine the factor of disadvantage, and 1 indicator (TNm) reflects the coefficient of biodiversity by the species composition of mosses. According to Figs. 7 and 8, it was found that the species richness index of TNm is maximally opposed to the indicator of the manifestation of terat in flowering plants and mosses (TTp) and the atypical structure of plants in vegetative organs (TTv)—a strong negative correlation. On this ground, it is concluded that species diversity is an indicator of favourable conditions and has a negative correlation with high levels of pollution by technophilic elements.
Index of embryonic teratogenesis—frequency of occurrence of terat variants of plant embryos (IETp) and index of morphological heterogeneity—the total sum of abnormalities in the structure of the leaf, shoot and inflorescences for all indicator species on the same accounting platform (IMHp) have patterns on maps in the same plane that are characteristic for data distribution with clearly defined areas by sources of pollution and the nature of distribution of elements and their compounds for adjacent territories (Figs. 9, 10). The greatest contribution to the IETp are from plants Capsella bursa-pastoris, Diplotaxis muralis, Reseda lutea, Tragopogon major, other plant species to the IMHp indicator: Berteroa incana, Centaurea diffusa, Cichorium intybus, Echium vulgare, Plantago lanceolata, Plantago major, Senecio vulgaris, Tanacetum vulgare, Tripleurospermum inodorum.
The biogeochemical role of rare earth elements in relation to physiological significance for the state of plants, animals and humans has not been reliably established [15, 49]. There is evidence that all of these elements are associated with the mineral extraction [50, 51], which is relevant for 90% of the territory of Donbass.
The strongest positive correlation between the indicators of teratogenesis TTp and TTgp is explained by a unified vector of structural transformation of plants (Berteroa incana, Centaurea diffusa, Cichorium intybus, Echium vulgare, Plantago major, Reseda lutea, Tanacetum vulgare, Tripleurospermum inodorum). At high exposure to pollutants, changes at the cellular level are observed, which consequently cause changes in the tissues of generative structures, ultimately this process is expressed in organ modification. As the result the general morphology of plant changes (Capsella bursa-pastoris, Cichorium intybus, Diplotaxis muralis, Echium vulgare, Plantago lanceolata, Reseda lutea, Senecio vulgaris, Tragopogon major, Tripleurospermum inodorum) is an object of express diagnostics of the state of unfavourable ecosystems. Therefore, both indicators are informative for determining the quality of natural environments, since they complement each other. However, the TTp criterion carries more general information about mosses (Ceratodon purpureus (Hedw.) Brid, Bryum argenteum Hedw., Brachythecium campestre (Müll.Hal.) Bruch et al., Bryum caespiticium Hedw., Bryum capillare Hedw., Pylaisia polyantha (Hedw.) Schimp., Amblystegium subtile (Hedw.) Schimp.) and flowering plants (Berteroa incana, Centaurea diffusa, Cichorium intybus, Echium vulgare, Plantago lanceolata, Senecio vulgaris, Tanacetum vulgare, Tripleurospermum inodorum), and the TTgp indicator is associated with changes in the generative sphere, primarily for the formation of plant embryos and seeds (Capsella bursa-pastoris, Diplotaxis muralis, Plantago major, Reseda lutea, Tragopogon major). Numerous cases of polymorphism, dystopias, oligomerizations and prolifications in plant organisms were noted.
Sc, La, Ce, Nd, Dy, Yb in present research have revealed concomitant patterns of concentration associated with metallurgical production, modification of foundry products. In some sources [52, 53] high migration abilities of these elements in metal dust was mentioned, that results in mechanical irritation of the human respiratory system [54, 55].
Among botanical features, GTm and PDI are less correlated with any factor and other botanical indices. This result shows that informative indicators for moss teratogenesis (Aulacomnium palustre (Hedw.) Schwägr., Brachythecium mildeanum (Schimp.) Schimp., Brachythecium salebrosum (F. Weber and D. Mohr) Bruch et al., Grimmia pulvinata (Hedw.) Sm., Orthotrichum pallens Bruch ex Brid., Orthotrichum pumilum Sw. ex anon., Plagiomnium cuspidatum (Hedw.) T. Kop., Pleurozium schreberi (Brid.) Mitt., Syntrichia ruralis (Hedw.) F. Weber and Mohr, Tortula mucronifolia Schwägr., Weissia controversa Hedw.) and plant pollen deformation (Berteroa incana, Centaurea diffusa, Cichorium intybus, Echium vulgare, Senecio vulgaris, Tanacetum vulgare, Tragopogon major, Tripleurospermum inodorum) depend on other factors. Apparently, the pollen and atypical structure of mosses depend on more toxic elements in the air and soil of the industrial region.
The obtained data on hyperfunctions of plant organs and morphostructural abnormalities by the type of overgrowth of some tissues. This effect can be explained by the peculiarities of some low-toxic rare earth elements (Sm, Eu, Yb) to metabolism stimulation in the plant organism, which is consistent with the published data [56, 57]. At the same time, Sc, La, Ce, Nd, Tb are characterized by increased toxicity, which explains the appearance of morphogenetic abnormalities of flowering plants. However, in literature, effects of these elements on the physiological properties are presented fragmentarily. It can be suggested that the main biogeochemical effect occurs as a result of associated contamination with elements having much more significant volumes of substance transfer in natural environments, for example, Zn, Mn, Al, Ni, which were presented in previous publications [12, 13]. In such cases, physiological toxicity of technophilic elements increases, which necessarily affects the state of living organisms.
CONCLUSIONS
(1) Various landscape systems of Donbass differ according to concentrations of rare earth elements in moss indicators. Determined elements have a reliable positive correlation with combined contamination, which proves a single source of their entry into biogeochemical cycles—the impact of industrial facilities in the Donbass. The highest concentrations were determined for Ce 12.6–61 mg/kg, while the lowest for Yb 0.52–1.9 mg/kg.
(2) Using mosses, the levels of accumulation and contamination with rare earth elements were established, which allowed to determine the relationship with other functional characteristics of the ecosystem—the atypical structure of the plant organism, the peculiarities of the manifestation of terat forms: dystopias, oligomerizations and prolifications in Capsella bursa-pastoris, Cichorium intybus, Diplotaxis muralis, Echium vulgare, Plantago major, Reseda lutea, Senecio vulgaris, Tragopogon major. The biodiversity index for mosses is defined as an indicator with the most negative correlation with pollution criteria, therefore it is considered an indicator of favorable conditions for the open steppe landscapes of the Northern Azov region.
(3) The most informative indicators are those that appear under the influence of even insignificant, but reliable fluctuations in the concentration of low-toxic rare earth elements. For the association of pollutants with a predominance of Sc, the embryonic apparatus of plants is more susceptible to change Berteroa incana, Capsella bursa-pastoris, Echium vulgare, Plantago major, Reseda lutea, Senecio vulgaris, Tanacetum vulgare, Tragopogon major; Ce influences the structure of the leaf, shoot and inflorescences Centaurea diffusa, Cichorium intybus, Diplotaxis muralis, Plantago lanceolata, Tripleurospermum inodorum. For Sc and Ce, the synergistic effect of pollutants in combination with La, Sm and Tb on accumulation in plant tissues is characteristic.
(4) According to contamination factor and enrichment factor values rare earth elements contribute to slight or moderate pollution of the natural systems of Donbass. The combination of the joint intake of rare earth elements enhances the mineral background in the structures of plant organisms and provokes appearance of abnormalities, neoplasms.
(5) A full-scale monitoring experiment using neutron activation analysis of phytoindicator samples allowed to study biogeochemical characteristics of rare earth elements, that for the natural ecosystems of the Donbass was done for the first time.
REFERENCES
S. S. Pavlov, A. Yu. Dmitriev, and M. V. Frontasyeva, “Automation system for neutron activation analysis at the reactor IBR-2, Frank Laboratory of Neutron Physics, Joint Institute for Nuclear Research, Dubna, Russia,” J. Radioanal. Nucl. Chem. 309, 27–38 (2016). https://doi.org/10.1007/s10967-016-4864-8
A. A. Antsiferova, M. Yu. Kopaeva, V. N. Kochkin, A. A. Reshetnikov, and P. K. Kashkarov, “Neurotoxicity of silver nanoparticles and non-linear development of adaptive homeostasis with age,” Micromachines 14, 984 (2023). https://doi.org/10.3390/mi14050984
L. H. Khiem, K. Sera, T. Hosokawa, L. D. Nam, and N. H. Quyet, “Active moss biomonitoring technique for atmospheric elemental contamination in Hanoi using proton induced X-ray emission,” J. Radioanal. Nucl. Chem. 325, 515–525 (2020). https://doi.org/10.1007/s10967-020-07253-y
A.O lacel, S. Ujeniuc, R. Suvaila, B. Alexandrescu, and I. Pojar, “Isotopic patterns via neutron irradiation and gamma spectrometry of environmental samples,” Chem. Phys. Impacts 4, 100065 (2022).
V. Takeshita, G. V. Munhoz-Garcia, A. E. S. Pereira, V. L. Tornisielo, and L. F. Fraceto, “Radiometric strategy to track nanopesticides: An important approach to understand the fate, mechanisms of action and toxicity,” Trends Anal. Chem. 165, 117156 (2023). https://doi.org/10.1016/j.trac.2023.117156
J. C. Massante, “Mining disaster: restore habitats now,” Nature 528, 39 (2015). https://doi.org/10.1038/528039c
Z. Bian, H. Yu, J. Hou, and S. Mu, “Influencing factors and evaluation of land degradation of 12 coal mine areas in Western China,” J. China Coal Soc. 45, 338–350 (2020).
J. Peng, Y. Pan, Y. Liu, H. Zhao, and Y. Wang, “Linking ecological degradation risk to identify ecological security patterns in a rapidly urbanizing landscape,” Habitat Int. 71, 110–124 (2018). https://doi.org/10.1016/j.habitatint.2017.11.010
R. Neamtu, B. Sluser, O. Plavan, and C. Teodosiu, “Environmental monitoring and impact assessment of Prut River cross-border pollution,” Environ. Monit. Assess. 193, 09110 (2021). https://doi.org/10.1007/s10661-021-09110-1
S. A. Yeprintsev, S. V. Shekoyan, L. A. Lepeshkina, A. A. Voronin, and M. A. Klevtsova, “Technologies for creating geographic information resources for monitoring the socio-ecological conditions of cities,” IOP Conf. Ser.: Mat. Sci. Eng. 582, 012012 (2019). https://doi.org/10.1088/1757-899X/582/1/012012
I. T. Bayouli, H. T. Bayouli, A. Dell’Oca, E. Meers, and J. Sun, “Ecological indicators and bioindicator plant species for biomonitoring industrial pollution: Eco-based environmental assessment,” Ecol. Indic. 125, 107508 (2021). https://doi.org/10.1016/j.ecolind.2021.107508
I. I. Zinicovscaia, K. N. Vergel, A. I. Safonov, N. S. Yushin, A. V. Kravtsova, and O. Chaligava, “Using moss Ceratodon purpureus (Hedw.) Brid for assessing the technogenic pollution (Ni, Zn, Mn, Al, Se, Cs, La, and Sm) of transformed ecotopes of Donbass,” Ecosyst. Transform. 6, 22–38 (2023). https://doi.org/10.23859/estr-220726
A. I. Safonov, A. S. Alemasova, I. I. Zinicovscaia, K. N. Vergel, N. S. Yushin, A. V. Kravtsova, and O. Chaligava, “Morphogenetic abnormalities of bryobionts in geochemically contrasting conditions of Donbass,” Geochem. Int. 68, 1032–1044 (2023). https://doi.org/10.1134/S0016702923100117
A. Safonov and A. Glukhov, “Ecological phytomonitoring in Donbass using geoinformational analysis,” BIO Web Conf. 31, 00020 (2021). https://doi.org/10.1051/bioconf/20213100020
D. Li, C. Jiang, C. Jiang, F. Liu, and Q. Zhu, “Geochemical characteristics and migration patterns of rare earth elements in coal mining subsidence lakes under the influence of multiple factors,” Sci. Total Environ. 904, 166668 (2023). https://doi.org/10.1016/j.scitotenv.2023.166668
N. M. Mupatsi and W. Gwenzi, “Chapter 3–High-Technology Rare Earth Elements in the Soil-Plant System: Occurrence, Behaviour, and Fate,” in Emerging Contaminants in the Terrestrial-Aquatic-Atmosphere Continuum (2022), pp. 29–46. https://doi.org/10.1016/B978-0-323-90051-5.00025-0
W. Valter da Silveira Pereira, S. J. Ramos Azevedo, L. C. Melo, Y. N. Dias, G. C. Martins, L. C. Gonçalves Ferreira, and A. R. Fernandes, “Human and environmental exposure to rare earth elements in gold mining areas in the northeastern Amazon,” Chemosphere 340, 139824 (2023). https://doi.org/10.1016/j.chemosphere.2023.139824
A. da Silva de Freitas, L. L. de Oliveira Pompermayer, A. D.de Oliveira Santos, M. T. Lima do Nascimento, T. Dillenburg Saint’Pierre, R. A. Hauser-Davis, J. A. Baptista Neto, and E. Monteiro da Fonseca, “Rare earth elements as sediment contamination tracers in a coastal lagoon in the state of Rio de Janeiro, Brazil,” J. Trace Elem. Miner. 4, 100068 (2023). https://doi.org/10.1016/j.jtemin.2023.100068
Y. W. Shen, C. X. Zhao, H. Zhao, S. F. Dong, Q. Guo, J. J. Xie, M. L. Lv, and C. G. Yuan, “Insight study of rare earth elements in PM2.5 during five years in a Chinese inland city: Composition variations, sources, and exposure assessment,” J. Environ. Sci. 138, 439—449 (2024). https://doi.org/10.1016/j.jes.2023.04.015
Q. Liu, H. Shi, Y. An, J. Ma, W. Zhao, Y. Qu, H. Chen, L. Liu, and F. Wu, “Source, environmental behavior and potential health risk of rare earth elements in Beijing urban park soils,” J. Hazard. Mater. 445, 130451 (2023). https://doi.org/10.1016/j.jhazmat.2022.130451
A. Zaghloul, M. Saber, S. Gadow, and F. Awad, “Biological indicators for pollution detection in terrestrial and aquatic ecosystems,” Bull. Natl. Res. Centre 44, 385 (2020).
P. K. Rai, C. Sonne, and K.-H. Kim, “Heavy metals and arsenic stress in food crops: Elucidating antioxidative defense mechanisms in hyperaccumulators for food security, agricultural sustainability, and human health,” Sci. Total Environ. 874, 162327 (2023). https://doi.org/10.1016/j.scitotenv.2023.162327
S. Allajbeu, N. S. Yushin, and F. Qarri, “Atmospheric deposition of rare earth elements in Albania studied by the moss biomonitoring technique, neutron activation analysis and GIS technology,” Environ. Sci. Pollution Res. 23, 14087–14101 (2016). https://doi.org/10.1007/s11356-016-6509-4
K. Bačeva Andonovska, R. Šajn, C. Tănăselia, and T. Stafilov, “Moss as an indicator of rare earth elements across the area of the volcanogenic deposit in the Allchar Locality, North Macedonia,” Air Qual., Atmos. Health 16, 1381–1391 (2023). https://doi.org/10.1007/s11869-023-01348-7
M. Aničić Urošević, M. Krmar, D. Radnović, G. Jovanović, T. Jakšić, P. Vasić, and A. Popović, “The use of moss as an indicator of rare earth element deposition over large area,” Ecol. Indic. 109, 105828 (2020). https://doi.org/10.1016/j.ecolind.2019.105828
A. I. Safonov and E. A. Germonova, “Ecological Phytomonitoring Networks in Donbass,” Probl. Ecol. Nat. Prot. Technogenic Region 3–4, 37–42 (2019).
D. Marasova, V. M. Zolotukhin, N. A. Zolotukhina, O. Volkova, and M. Yazevich, “Chemical monitoring of the socio-ecological situation in resource-producing regions,” E3S Web Conf. 315, 02003 (2021). https://doi.org/10.1051/e3sconf.202131502003
S. Tomasini and I. Theilade, “Local ecological knowledge indicators for wild plant management: Autonomous local monitoring in Prespa, Albania,” Ecol. Indic. 101, 1064–1076 (2019). https://doi.org/10.1016/j.ecolind.2019.01.076
T. V. Avraimova and A. I. Safonov, “Ecological developments in Donbass: bibliographic control and promotion of research,” Sci. Tech. Libr. 3, 30–42(2023). https://doi.org/10.33186/1027-3689-2023-3-30-42
A. S. Alemasova and A. I. Safonov, “Heavy metals in phytosubstrates as indicators of anthropogenic air pollution in industrial region,” For. Bull. 26, 5–13 (2022). https://doi.org/10.18698/2542-1468-2022-6-5-13
N. Krutskikh, “Modelling the structure of terrestrial landscapes in urban areas,” Quaestiones Geographicae 40, 39–49 (2021). https://doi.org/10.2478/quageo-2021-0003
A. Safonov, “Ecological scales of indicator plants in an industrial region,” BIO Web Conf. 43, 03002 (2022). https://doi.org/10.1051/bioconf/20224303002
A. I. Safonov, “Abnormalities of embryo structures in Donbass indicator plants 14, 5–18 (2022). https://doi.org/10.22281/2686-9713-2022-3-5-18
A. I. Safonov, “Theratogenesis of indicator plants of industrial Donbass,” Diversity Plant World 1, 4–16 (2019). https://doi.org/10.22281/2686-9713-2019-1-4-16
A. I. Safonov, “Phyto-qualimetry of toxic pressure and the degree of ecotopes transformation in Donetsk region,” Probl. Ecol. Nat. Prot. Technogenic Region 13, 52–59 (2013).
A.D. Bell, Plant Form: An Illustrated Guide to Flowering Plant Morphology (Oxford Univ. Press, New York, 1991).
M. A. Outram, M. Figueroa, J. Sperschneider, S. J. Williams, and P. N. Dodds, “Seeing is believing: exploiting advances in structural biology to understand and engineer plant immunity,” Curr. Opin. Plant Biol. 67, 102210 (2022). https://doi.org/10.1016/j.pbi.2022.102210
I. Zinicovscaia, C. Hramco, O. Chaligava, N. Yushin, D. Grozdov, K. Vergel, and G. Duca, “Accumulation of potentially toxic elements in mosses collected in the Republic of Moldova,” Plants 10, 1–13 (2021). https://doi.org/10.3390/plants10030471
K. Vergel, I. Zinicovscaia, N. Yushin, and S. Gundorina, “Assessment of atmospheric deposition in Central Russia using moss biomonitors, neutron activation analysis and GIS technologies,” J. Radioanal. Nucl. Chem. 325, 807–816 (2020). https://doi.org/10.1007/s10967-020-07234-1
J. A. Fernandez, J. R. Aboal, and A. Carballeira, “Identification of pollution sources by means of moss bags,” Ecotoxicol. Environ. Saf. 59, 76–83 (1994). )https://doi.org/10.1016/j.ecoenv.2004.01.007
C. R. Bern, K. Walton-Day, and D. L. Naftz, “Improved enrichment factor calculations through principal component analysis: Examples from soils near breccia pipe uranium mines, Arizona, USA,” Environ. Pollut. 248, 90–100 (2019). https://doi.org/10.1016/j.envpol.2019.01.122
R. L. Rudnick and S. Gao, “The Composition of the Continental Crust,” in Treatise on Geochemistry Ed. by H. D. Holland and K. K. Turekian (Elsevier-Pergamon, Oxford, 2003), Vol. 3: The Crust, pp. 1–64. https://doi.org/10.1016/b0-08-043751-6/03016-4
L. Chen, H. Zhang, M. Ding, A. T. Devlin, P. Wang, M. Nie, and K. Xie, “Exploration of the variations and relationships between trace metal enrichment in dust and ecological risks associated with rapid urban expansion,” Ecotoxicol. Environ. Saf. 212, 111944 (2021). https://doi.org/10.1016/j.ecoenv.2021.111944
A. Sergeeva, I. Zinicovscaia, D. Grozdov, and N. Yushin, “Assessment of selected rare earth elements, HF, Th, and U in the Donetsk region using moss bags technique,” Atmos. Pollut. Res. 12, 101165 (2021). https://doi.org/10.1016/j.apr.2021.101165
V. Balaram, “Potential future alternative resources for rare earth elements: opportunities and challenges,” Minerals 13, 425 (2023). https://doi.org/10.3390/min13030425
Y. Tao, L. Shen, C. Feng, R. Yang, J. Qu, H. Ju, and Y. Zhang, “Distribution of rare earth elements (REEs) and their roles in plant growth: A review,” Environ. Pollut. 298, 118540 (2022). https://doi.org/10.1016/j.envpol.2021.118540
A. Kolker, C. Scott, J. C. Hower, J. A. Vazquez, C. L. Lopano, and S. Dai, “Distribution of rare earth elements in coal combustion fly ash, determined by SHRIMP-RG ion microprobe,” Int. J. Coal Geol. 184, 1—10 (2017). https://doi.org/10.1016/j.coal.2017.10.002
B. H. Alharbi, M. J. Pasha, M. D. Alotaibi, A. K. Alduwais, and M. A. S. Al-Shamsi, “Contamination and risk levels of metals associated with urban street dust in Riyadh, Saudi Arabia,” Environ. Sci. Pollut. Res. 27, 18475–18487 (2020). https://doi.org/10.1007/s11356-020-08362-7
M.-C. Lafrenière, J.-F. Lapierre, D. E. Ponton, F. Guillemette, and M. Amyot, “Rare earth elements (REEs) behavior in a large river across a geological and anthropogenic gradient,” Geochim. Cosmochim. Acta 353, 129–141 (2023). https://doi.org/10.1016/j.gca.2023.05.019
T. Zerizghi, Q. Guo, R. Wei, Z. Wang, C. Du, and Y. Deng, “Rare earth elements in soil around coal mining and utilization: Contamination, characteristics, and effect of soil physicochemical properties,” Environ. Pollut. 331, Part 2, 121788 (2023). https://doi.org/10.1016/j.envpol.2023.121788
H. Liu, H. Guo, O. Pourret, Z. Wang, M. Liu, W. Zhang, Z. Li, B. Gao, Z. Sun, and P. Laine, “Geochemical signatures of rare earth elements and yttrium exploited by acid solution mining around an ion-adsorption type deposit: role of source control and potential for recovery,” Sci. Total Environ. 804, 150241 (2022). https://doi.org/10.1016/j.scitotenv.2021.150241
L. Dai, L. Deng, W. Wang, Y. Li, L. Wang, T. Liang, X. Liao, J. Cho, C. Sonne, S. S. Lam and J. Rinklebe, “Potentially toxic elements in human scalp hair around China’s largest polymetallic rare earth ore mining and smelting area,” Environ. Int. 172, 107775 (2023). https://doi.org/10.1016/j.envint.2023.107775
S. Cheng, W. Li, Y. Han, Y. Sun, P. Gao, and X. Zhang, “Recent process developments in beneficiation and metallurgy of rare earths: a review,” J. Rare Earths (2023, in press). https://doi.org/10.1016/j.jre.2023.03.017
L. Dai, L. Wang, X. Wan, J. Yang, Y. Wang, T. Liang, H. Song, S. M. Shaheen, V. Antoniadis, and J. Rinklebe, “Potentially toxic elements exposure biomonitoring in the elderly around the largest polymetallic rare earth ore mining and smelting area in China,” Sci. Total Environ. 853, 158635 (2022).https://doi.org/10.1016/j.scitotenv.2022.158635
Z. Li, T. Liang, K. Li, and P. Wang, “Exposure of children to light rare earth elements through ingestion of various size fractions of road dust in REEs mining areas,” Sci. Total Environ. 743, 140432 (2020).https://doi.org/10.1016/j.scitotenv.2020.140432
C. Zhang, Q. Li, M. Zhang, N. Zhang, and M. Li, “Effects of rare earth elements on growth and metabolism of medicinal plants,” Acta Pharm. Sin. B 3, 20—24 (2013). https://doi.org/10.1016/j.apsb.2012.12.005
Y. Tao, L. Shen, C. Feng, R. Yang, J. Qu, H. Ju, and Y. Zhang, “Distribution of rare earth elements (REEs) and their roles in plant growth: a review,” Environ. Pollut. 298, 118540 (2022). https://doi.org/10.1016/j.envpol.2021.118540
Funding
This work was supported by ongoing institutional funding. No additional grants to carry out or direct this particular research were obtained.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Table S1 . The content of rare earth elements (С) in the control and exposed samples of indicator moss Ceratodon purpureus (Hedw.) Brid (µg/g) in the ecotopes of Donbass
Site | Sc | La | Ce | Nd | Sm | Eu | Tb | Dy | Yb | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
С | %* | С | % | С | % | С | % | С | % | С | % | С | % | С | % | С | % | |
1 | 8.1 | 6 | 29.1 | 9 | 54 | 9 | 27 | 12 | 4.5 | 10 | 0.72 | 18 | 0.49 | 5 | 2.9 | 18 | 1.48 | 12 |
2 | 6 | 6 | 17.9 | 9 | 32.6 | 9 | 13.8 | 13 | 3.1 | 10 | 0.41 | 27 | 0.381 | 5 | 2.2 | 18 | 1.34 | 12 |
3 | 4.39 | 6 | 13.6 | 9 | 27.5 | 9 | 13.5 | 12 | 2.4 | 10 | 0.56 | 15 | 0.315 | 5 | 1.26 | 17 | 1.14 | 12 |
4 | 5.6 | 6 | 14.8 | 9 | 24.1 | 9 | 14.1 | 14 | 2.48 | 10 | 0.53 | 28 | 0.307 | 5 | 2.6 | 19 | 0.97 | 12 |
5 | 6.3 | 6 | 14.2 | 9 | 27.7 | 9 | 11.8 | 16 | 2.29 | 10 | 0.32 | 20 | 0.295 | 5 | 1.6 | 19 | 1.12 | 12 |
6 | 5.4 | 6 | 14.9 | 9 | 27.6 | 9 | 14 | 13 | 2.79 | 10 | 0.77 | 15 | 0.4 | 5 | 1.8 | 20 | 1.57 | 11 |
7 | 4.89 | 6 | 12.8 | 9 | 26.6 | 9 | 10.6 | 14 | 2.62 | 9 | 0.57 | 15 | 0.333 | 5 | 1.4 | 21 | 1.15 | 12 |
8 | 5.6 | 6 | 14.1 | 9 | 28.6 | 9 | 15.3 | 13 | 2.74 | 10 | 0.46 | 21 | 0.361 | 5 | 2.6 | 19 | 1.38 | 12 |
9 | 4.35 | 6 | 12.4 | 9 | 21.4 | 9 | 12.1 | 14 | 2.18 | 10 | 0.5 | 18 | 0.276 | 5 | 1.49 | 19 | 1.02 | 12 |
10 | 3.83 | 4 | 10 | 9 | 19.7 | 9 | 13.3 | 16 | 1.94 | 10 | 0.38 | 20 | 0.269 | 5 | 1.8 | 19 | 0.88 | 11 |
11 | 6.45 | 4 | 24.9 | 9 | 47 | 9 | 24 | 14 | 4.2 | 9 | 0.89 | 15 | 0.515 | 5 | 2.9 | 19 | 1.58 | 11 |
12 | 7.16 | 4 | 22.4 | 9 | 43 | 9 | 22 | 14 | 4 | 10 | 0.68 | 19 | 0.511 | 5 | 2.8 | 14 | 1.58 | 11 |
13 | 7.07 | 4 | 19.5 | 9 | 33 | 9 | 24 | 15 | 3.6 | 10 | 0.72 | 18 | 0.468 | 5 | 3.3 | 19 | 1.44 | 11 |
14 | 5.01 | 4 | 14 | 9 | 28.1 | 9 | 12.2 | 16 | 2.61 | 9 | 0.55 | 16 | 0.322 | 5 | 1.6 | 19 | 1.01 | 11 |
15 | 4.35 | 4 | 13.3 | 9 | 24.7 | 9 | 14.7 | 16 | 2.46 | 9 | 0.54 | 16 | 0.3 | 5 | 2 | 19 | 0.86 | 11 |
16 | 6.6 | 4 | 16.4 | 9 | 31.6 | 9 | 16.7 | 16 | 3.04 | 9 | 0.66 | 16 | 0.397 | 5 | 2.2 | 19 | 1.3 | 11 |
17 | 4.88 | 4 | 13.6 | 9 | 23.5 | 9 | 15.7 | 15 | 2.49 | 9 | 0.52 | 16 | 0.316 | 5 | 1.6 | 20 | 1.06 | 11 |
18 | 3.32 | 6 | 9.4 | 9 | 17.8 | 8 | 9.3 | 23 | 1.66 | 10 | 0.26 | 13 | 0.216 | 5 | 1.5 | 20 | 0.66 | 13 |
19 | 5.8 | 6 | 22.2 | 9 | 40 | 8 | 14 | 22 | 3.6 | 10 | 0.37 | 19 | 0.41 | 5 | 1.7 | 19 | 1.62 | 12 |
20 | 7.6 | 6 | 30.4 | 9 | 61 | 8 | 29 | 19 | 5.1 | 10 | 0.86 | 10 | 0.59 | 5 | 2.8 | 19 | 1.9 | 12 |
21 | 1.89 | 6 | 5.1 | 9 | 9.5 | 9 | <DL** | 0.95 | 10 | 0.2 | 17 | 0.13 | 5 | 1.04 | 19 | 0.44 | 16 | |
22 | 4.72 | 6 | 14.4 | 9 | 29.6 | 8 | 15 | 20 | 2.6 | 10 | 0.45 | 13 | 0.32 | 5 | 1.8 | 19 | 0.99 | 13 |
23 | 2.36 | 6 | 6.3 | 9 | 12.6 | 8 | <DL | 1.29 | 10 | 0.225 | 13 | 0.167 | 5 | 0.57 | 24 | 0.52 | 14 | |
24 | 3.52 | 6 | 9.4 | 9 | 20 | 8 | <DL | 1.76 | 10 | 0.29 | 13 | 0.215 | 5 | 1.03 | 20 | 0.67 | 14 | |
Table S2 . Signs of structural organization of flowering plants in the ecotopes of Donbass
IETp | % | IMHp | % | ТNm | % | GТm | % | PDI | % | ТТp | % | ТТvp | % | TTgp | % | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
1 | 16 | 3 | 56.1 | 5 | 15 | 2 | 3.2 | 3 | 17 | 2 | 28 | 7 | 9.7 | 7 | 18 | 7 |
2 | 13.4 | 3 | 33 | 3 | 3.6 | 6 | 39.5 | 4 | 4.1 | 2 | 52.2 | 8 | 11 | 8 | 41.2 | 2 |
3 | 9.1 | 3 | 28.4 | 3 | 20.1 | 6 | 2 | 3 | 5.1 | 1 | 20.6 | 8 | 4.8 | 5 | 15.3 | 7 |
4 | 12.3 | 3 | 25.9 | 3 | 9 | 2 | 39 | 4 | 27.7 | 1 | 39.4 | 8 | 9 | 7 | 30 | 7 |
5 | 13.2 | 1 | 29.7 | 3 | 16 | 2 | 4.5 | 4 | 8.6 | 2 | 19 | 7 | 9.7 | 7 | 9.1 | 5 |
6 | 12.6 | 1 | 29.3 | 5 | 9.5 | 5 | 4 | 4 | 2.3 | 1 | 15.9 | 7 | 7.3 | 7 | 8.9 | 7 |
7 | 10.3 | 3 | 27.2 | 5 | 25.3 | 5 | 7.8 | 4 | 4 | 1 | 4.1 | 8 | 1.4 | 5 | 2.9 | 3 |
8 | 11.1 | 3 | 29.8 | 5 | 26 | 5 | 3 | 3 | 15.6 | 1 | 10.3 | 7 | 4 | 8 | 6 | 7 |
9 | 9.6 | 2 | 20.6 | 3 | 14.4 | 5 | 2.4 | 3 | 12.5 | 1 | 19 | 8 | 5 | 5 | 14 | 3 |
10 | 7.8 | 2 | 19.5 | 3 | 7 | 5 | 33.7 | 4 | 19.1 | 2 | 38 | 8 | 9.8 | 8 | 28 | 5 |
11 | 12 | 2 | 49.3 | 4 | 6.2 | 5 | 38.9 | 4 | 15.9 | 2 | 25.5 | 7 | 10.3 | 7 | 15 | 3 |
12 | 15.7 | 2 | 45.2 | 4 | 16.3 | 6 | 11.3 | 4 | 11.5 | 2 | 30 | 8 | 9.1 | 7 | 21 | 7 |
13 | 15.9 | 1 | 34.1 | 3 | 13.9 | 6 | 6.7 | 4 | 18 | 2 | 16.2 | 8 | 8.2 | 7 | 8 | 5 |
14 | 11.4 | 3 | 29.8 | 3 | 10 | 2 | 30 | 4 | 21.2 | 2 | 14 | 8 | 10 | 8 | 3.9 | 3 |
15 | 9.2 | 3 | 25.2 | 3 | 20.4 | 6 | 6.3 | 3 | 9 | 2 | 10.4 | 8 | 5 | 8 | 5.3 | 2 |
16 | 13.3 | 2 | 32.9 | 5 | 15 | 5 | 6.9 | 3 | 16.7 | 2 | 15 | 7 | 7.3 | 7 | 8 | 2 |
17 | 9.5 | 2 | 24.3 | 3 | 12 | 2 | 20.8 | 4 | 19 | 1 | 10.2 | 7 | 4.6 | 8 | 5 | 2 |
18 | 6 | 2 | 18.5 | 3 | 11.2 | 5 | 7.1 | 3 | 9.3 | 2 | 9.1 | 7 | 4.2 | 8 | 5.1 | 7 |
19 | 11.8 | 2 | 41 | 3 | 17.7 | 5 | 13 | 3 | 15.8 | 2 | 18.7 | 8 | 8.4 | 7 | 10.4 | 3 |
20 | 15.5 | 3 | 63 | 3 | 7.5 | 2 | 7 | 3 | 13.8 | 2 | 17 | 8 | 9.8 | 7 | 7.1 | 5 |
21 | 4.7 | 3 | 10.6 | 5 | 17.8 | 6 | 12.4 | 4 | 5.4 | 2 | 9.3 | 7 | 4 | 7 | 4.8 | 5 |
22 | 9.9 | 1 | 30.4 | 4 | 12 | 2 | 15.2 | 4 | 9.4 | 2 | 18.8 | 7 | 8.6 | 8 | 10.4 | 7 |
23 | 5.1 | 2 | 13.7 | 3 | 15 | 2 | 30 | 3 | 16.9 | 2 | 9.4 | 7 | 3.5 | 8 | 5.5 | 7 |
24 | 7 | 2 | 4.4 | 3 | 25.1 | 2 | 19.6 | 4 | 10.2 | 2 | 7.3 | 8 | 5.1 | 5 | 2.2 | 2 |
Rights and permissions
About this article
Cite this article
Zinicovscaia, I., Safonov, A., Kravtsova, A. et al. Neutron Activation Analysis of Rare Earth Elements (Sc, La, Ce, Nd, Sm, Eu, Tb, Dy, Yb) in the Diagnosis of Ecosystems of Donbass. Phys. Part. Nuclei Lett. 21, 186–200 (2024). https://doi.org/10.1134/S1547477124020158
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1547477124020158