Abstract
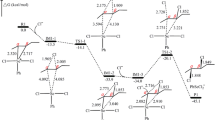
Rate constants and activation parameters of two-step azidation of isomeric dicyanobenzenes with dimethylammonium azide in DMF at 70–100°C were determined. Tetrazole rings are formed from cyano groups in dicyanobenzenes in a stepwise mode following the 1,3-dipolar cycloaddition pattern. The rate of azidation of isomeric dicyanobenzenes is considerably higher than the rate of subsequent azidation of intermediate cyanophenyltetrazolides. The azidation rate constant decrease in going from m-dicyanobenzene to its para and ortho isomers.
Similar content being viewed by others
References
Ostrovskii, V.A., Koldobskii, G.I., and Trifonov, R.E., Comprehensive Heterocyclic Chemistry III, Katritzky, A.R., Ramsden, C.A., Scriven, E.F.V, and Taylor, R.J.K., Eds., Amsterdam: Elsevier, 2008, vol. 6, p. 257.
Koldobskii, G.I., Ostrovskii, V.A., and Poplavskii, V.S., Khim. Geterotsikl. Soedin., 1981, p. 1299.
Noodleman, L., Lovell, T., Han, W.-G., Li, J., and Himo, F., Chem. Rev., 2004, vol. 104, p. 459.
Ostrovskii, V.A., Poplavskii, V.S., Koldobskii, G.I., and Erusalimskii, G.B., Khim. Geterotsikl. Soedin., 1992, p. 1214.
Titova, I.E., Poplavskii, V.S., Koldobskii, G.I., Ostrovskii, V.A., Nikolaev, V.D., and Erusalimskii, G.B., Khim. Geterotsikl. Soedin., 1986, p. 1086.
Emmanuel’, N.M. and Knorre, D.G., Kurs khimicheskoi kinetiki (Lectures on Chemical Kinetics), Moscow: Vysshaya Shkola, 1984, p. 184.
Spiridonov, V.P. and Lopatkin, A.A., Matematicheskaya obrabotka fiziko-khimicheskikh dannykh (Mathematical Processing of Physicochemical Data), Moscow: Mosk. Gos. Univ., 1970, p. 98.
Shchipanov, V.P., Khim. Geterotsikl. Soedin., 1983, p. 1130.
Becker, H., Einführung in die Elektronentheorie organisch-chemischer Reaktionen, Berlin: Wissenschaften, 1974, 3rd ed. Translated under the title Vvedenie v elektronnuyu teoriyu organicheskikh reaktsii, Moscow: Mir, 1977, p. 145.
Hoffmann, R.W., Auf Klärung von Reaktions-mechanismen, Stuttgart: Georg Thieme, 1976. Translated under the title Mekhanizmy khimicheskikh reaktsii, Moscow: Khimiya, 1979, p. 45.
Reichardt, C., Solvents and Solvent Effects in Organic Chemistry, Weinheim: VCH, 1988, 2nd ed. Translated under the title Rastvoriteli i effekty sredy v organicheskoi khimii, Moscow: Mir, 1991, p. 132.
Rudakov, O.B., Vostrov, I.A., Fedorov, S.V., Filippov, A.A., Selemenev, V.F., and Pridantsev, A.A., Sputnik khromatografista. Metody zhidkostnoi khromatografii (Chromatographist’s Companion. Methods of Liquid Chromatography), Voronezh: Vodolei, 2004, p. 333.
Koguro, K., Oga, T., Mitsui, S., and Orita, R., Synthesis, 1998, no. 6, p. 910.
Kruszewski, J., Kaczmarek, J., Bartkowiak, R., and Grzonka, Z., Pol. J. Chem., 1980, vol. 54, p. 925.
Popova, E.A., Ivanova, A.V., Trifonov, R.E., Popov, E.V., Zubarev, V.Yu., Tselinskii, I.V., and Ostrovskii, V.A., Russ. J. Org. Chem., 2007, vol. 43, p. 591.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Full Member of the Russian Academy of Sciences O.N. Chupakhin on his 75th anniversary
Original Russian Text © E.A. Popova, Yu.N. Pavlyukova, E.V. Popov, V.A. Ostrovskii, R.E. Trifonov, 2009, published in Zhurnal Organicheskoi Khimii, 2009, Vol. 45, No. 6, pp. 902–906.
Rights and permissions
About this article
Cite this article
Popova, E.A., Pavlyukova, Y.N., Popov, E.V. et al. Kinetics of azidation of isomeric benzenedicarbonitriles. Russ J Org Chem 45, 890–894 (2009). https://doi.org/10.1134/S1070428009060153
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428009060153