Abstract
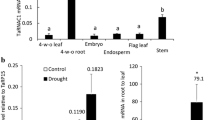
Optimization of Acidothermus cellulolyticus endoglucanase (E1) gene expression in transgenic potato (Solanum tuberosum L.) was examined in this study, where the E1 coding sequence was transcribed under control of a leaf specific promoter (tomato RbcS-3C) or the Mac promoter (a hybrid promoter of mannopine synthase promoter and cauliflower mosaic virus 35S promoter enhancer region). Average E1 activity in leaf extracts of potato transformants, in which E1 protein was targeted by a chloroplast signal peptide and an apoplast signal peptide were much higher than those by an E1 native signal peptide and a vacuole signal peptide. E1 protein accumulated up to 2.6% of total leaf soluble protein, where E1 gene was under control of the RbcS-3C promoter, alfalfa mosaic virus 5′-untranslated leader, and RbcS-2A signal peptide. E1 protein production, based on average E1 activity and E1 protein accumulation in leaf extracts, is higher in potato than those measured previously in transgenic tobacco bearing the same transgene constructs. Comparisons of E1 activity, protein accumulation, and relative mRNA levels showed that E1 expression under control of tomato RbcS-3C promoter was specifically localized in leaf tissues, while E1 gene was expressed in both leaf and tuber tissues under control of Mac promoter. This suggests dual-crop applications in which potato vines serve as enzyme production `bioreactors' while tubers are preserved for culinary applications.
Similar content being viewed by others
References
An G, Ebert PR, Ha S-B: Binary vector. In: Gelvin SB, Schilperoort RA (Eds.) Plant Molecular Biology Mannual, pp. 1-19. Kluwer Academic Publishers, Dordrecht, Netherlands (1988).
Baker JO, Adney WS, Nieves RA, Thomas SR, Wilson DB, Himmel ME: A new thermostable endoglucanase, Acidothermus cellulolyticus E1: synergism with Trichoderma reesei CBHI and comparison to Thermomonospora fusca E5. Appl Bioch Biotechnol 45-46: 245-256 (1994).
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-254 (1976).
Comai L, Moran P, Maslyar D: Novel and useful properties of chimeric plant promoter combining CaMV 35S and MAS elements. Plant Mol Biol 15: 373-381 (1990).
Conrad U, Fiedler U: Compartment-specific accumulation of recombinant immunoglobulins in plant cells: an essential tool for antibody production and immunomodulation of physiological functions and pathogen activity. Plant Mol Biol 38: 101-109 (1998).
Cornelissen BJC, Hooft van Huijsduijnen RAM, Bol JF: A tobacco mosaic virus-induced tobacco protein is homologous to the sweet-tasting protein thaumatin. Nature 321: 531-532 (1986).
Dai Z, Ku MSB, Zhang D, Edwards GE: Effects of growth regulators on the induction of crassulacean acid metabolism in the facultative halophyte Mesembryanthemum crystallinum L. Planta 192: 287-294 (1994).
Dai Z, An G: Induction of nopaline synthase promoter activity by H2O2 has no direct correlation with salicylic acid. Plant Physiol 109: 1191-1197 (1995).
Dai Z, Hooker BS, Quesenberry RD, Gao J: Expression of Trichoderma reesei exo-cellobiohydrolase I in transgenic tobacco leaves and calli. Appl Biochem Biotech 77-79: 689-699 (1999).
Dai Z, Hooker, BS, Anderson DB, Thomas SR: Expression of Acidothermus cellulolyticuls endoglucanase E1 in transgenic tabacco: biochemical characteriscs and physilogical effects. Transgenic Research 9: 43-54 (2000).
Duff SJ, Murray WD: Bioconversion of forest products industry waste cellulosics to fuel ethanol: a review. Bioresource Technol 55: 1-33 (1996).
Gallie DR, Sleat DE, Watts JW, Turner PC, Wilson TMA: A comparison of eukaryotic viral 50-leader sequences as enhancers of mRNA expression in vivo. Nucl Acids Res 15: 8693-8711 (1987).
Gallie DR: Controlling gene expression in transgenics. Curr Opin Plant Biol 1: 166-172 (1998).
Herbers K, Wilke I, and Sonnewald U: A thermostable xylanase from Clostridium thermocellum expressed at high levels in the apoplast of transgenic tobacco has no detrimental effects and is easily purified. Biot/technology 13: 63-66 (1995).
Hoekema A, Hirsch PR, Hooykaas PJJ, Schilperoort RA: A binary vector strategy based on separation of vir-and T-region of the Agrobacterium tumefaciens Ti-plasmid. Nature 303: 179-181 (1983).
Holmes-Davis R, Comai L: Nuclear matrix attachment regions and plant gene expression. Trends Plant Sci 3: 91-97 (1998).
Jensen LG, Olsen O, Kops O, Wolf N, Thomsen KK, Wettstein DV: Transgenic barley expressing a protein-engineered, thermostable (1, 3-1,4)-β-glucanase during germination. Proc Natl Acad Sci USA 93: 3487-3491 (1996).
Jobling SA, Gehrke L: Enhanced translation of chimaeric messenger RNAs containing a plant viral untranslated leader sequence. Nature 325: 622-625 (1987).
Koziel MC, Carozzi NB, Desai N: Optimizing expression of transgenes with an emphasis on post-transcriptional events. Plant Mol Biol 32: 393-405 (1996).
Liu J-H, Selinger B, Cheng K-J, Beauchemin KA, Moloney MM: Plant seed oil-bodies as an immobilization matrix for a recombinant xylanase from the rumen fungus Neocallimastix patriciarum. Mol Breed 3: 463-470 (1997).
Manzara T, Carrasco P, Gruissem W: Developmental and organ-specific changes in promoter DNA-protein interactions in the tomato rbcS gene family.Plant Cell 3: 1305-1316 (1991).
Matsuoka K, Bassham DC, Raikhel NV, Nakamura K: Different sensitivity to wortmannin of two vacuolar sorting signals indicates the presence of distinct sorting machineries in tobacco cells. J Cell Biol 130: 1307-1318 (1995).
Matsuoka K, Nakamura K: Fropeptide of a precursor to a plant vacuole protein required for vacuolar targeting. Proc Natl Acad Sci USA 88: 834-838 (1991).
Murashige T, Skoog F: A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15: 473-497 (1962).
Nakamura K, Matsuoka K, Mukumoto F, Watanabe N: Processing and transport to the vacuole of a precursor to sweet potato sporamin in transformed tobacco cell line BY-2. J Exp Bot 44 (suppl): 331-338 (1993).
Sakon J, Adney WS, Himmel ME, Thomas SR, Karplus PA: Crystal structure of thermostable family 5 endocellulase E1 from Acidothermus cellulolyticus in a complex with cellotetraose. Biochemistry 35: 10648-10660 (1996).
Sambrook J, Fritsch EF, Maniatis T: Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY (1989).
Schouten A, Roosien J, van Engelen FA, de Jong GAM, Borst-Vrenssen AWM, Zilverentant JF, Bosch D, Stiekema WJ, Gommer FJ, Schots A, Baker J: The C-terminal KDEL sequence increases the expression level of a single-chain antibody designed to be targeted to both the cytosol and the secretory pathway in transgenic tobacco. Plant Mol Biol 30: 781-793 (1996).
Spiker S, Thompson WF: Nuclear matrix attachment regions and transgene expression in plants. Plant Physiol 110: 15-21 (1996).
Sugita M, Manzara T, Pichersky E, Cashmore A, Gruissem W: Genomic organization, sequence analysis and expression of all five genes encoding the small subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase from tomato. Mol Gen Genet 209: 247-256 (1987).
Tucker MP, Mohagheghi A, Grohman K, Himmel ME: Ultrathermostable cellulases from acidothermus cellulolyticus: Comparison of temperature optima with previously reported cellulases. Bio/technology 7: 817-820 (1989).
Verwoerd TC, van Paridon PA, van Ooyen AJJ, van Lent JWM, Hoekema A, Pen J: Stable accumulation of Aspergillus niger phytase in transgenic tobacco leaves. Plant Physiol 109: 1199-1203 (1995).
Wenzler N, Mignery G, May G, Park W: A rapid and efficient transformation method for the production of large numbers of transgenic potato plants. Plant Sci 63: 79-85 (1989).
Ziegelhoffer T, Will J, Austin-Phillips S: Expression of bacterial cellulase genes in transgenic alfalfa (medicago sativa L.), potato (Ssolanum tuberosum L.) and tobacco (Nicotiana tabacum L.). Mol Breed 5: 309-318 (1999).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dai, Z., Hooker, B.S., Anderson, D.B. et al. Improved plant-based production of E1 endoglucanase using potato: expression optimization and tissue targeting. Molecular Breeding 6, 277–285 (2000). https://doi.org/10.1023/A:1009653011948
Issue Date:
DOI: https://doi.org/10.1023/A:1009653011948