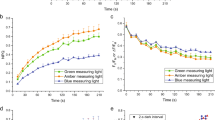
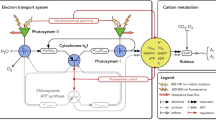
Abstract
A number of useful photosynthetic parameters are commonly derived from saturation pulse-induced fluorescence analysis. We show, that qP, an estimate of the fraction of open centers, is based on a pure ‘puddle’ antenna model, where each Photosystem (PS) II center possesses its own independent antenna system. This parameter is incompatible with more realistic models of the photosynthetic unit, where reaction centers are connected by shared antenna, that is, the so-called ‘lake’ or ‘connected units’ models. We thus introduce a new parameter, qL, based on a Stern–Volmer approach using a lake model, which estimates the fraction of open PS II centers. We suggest that qL should be a useful parameter for terrestrial plants consistent with a high connectivity of PS II units, whereas some marine species with distinct antenna architecture, may require the use of more complex parameters based on intermediate models of the photosynthetic unit. Another useful parameter calculated from fluorescence analysis is ΦII, the yield of PS II. In contrast to qL, we show that the ΦII parameter can be derived from either a pure ‘lake’ or pure ‘puddle’ model, and is thus likely to be a robust parameter. The energy absorbed by PS II is divided between the fraction used in photochemistry, ΦII, and that lost non-photochemically. We introduce two additional parameters that can be used to estimate the flux of excitation energy into competing non-photochemical pathways, the yield induced by downregulatory processes, ΦNPQ, and the yield for other energy losses, ΦNO.
Similar content being viewed by others
References
Adams WI and Demmig-Adams B (2004) Chlorophyll fluorescence as a tool to monitor plant response to the environment. In: Papageorgiou G and Govindjee (eds) Chlorophyll Fluorescence: The Signature of Green Plant Photosynthesis. Advances in Photosynthesis and Respiration. Kluwer Academic Publishers, Dordrecht, The Netherlands (in press)
Andrews JR (1995) Characterization of chilling effects on photosynthetic performance of maize crops during early season growth using chlorophyll fluorescence. J Exp Bot 46: 1195–1203
Baker NR (1991) A possible role for Photosystem II in environmental perturbations of photosynthesis. Physiol Plant 81: 563–570
Baker NR, Oxborough K and Andrews JR (1995) Operation of an alternate electron transfer acceptor to CO2 in maize crops during periods of low temperatures. In: Mathis P (ed) Photosynthesis: From Light to Biosphere, Vol IV, pp 771–776. Kluwer Academic Publishers, Dordrecht, The Netherlands
Baker NR, Oxborough K, Lawson T and Morison JI (2001) High resolution imaging of photosynthetic activities of tissues, cells and chloroplasts in leaves. J Exp Bot 52: 615–621
Barber J (2003) Photosystem II: the engine of life. Q Rev Biophys 36: 71–89
Blankenship RE (2002) Molecular Mechanisms of Photosynthesis.Blackwell Scientific, Oxford
Bradbury M and Baker NR (1983) Analysis of the induction of chlorophyll fluorescence in leaves and isolated thylakoids: contributions of photochemical and non-photochemical quenching. Proc R Soc London B 220: 251–264
Bradbury M and Baker N (1984) A quantitative determination of photochemical and non-photochemical quenching during the slow phase of the chlorophyll fluorescence induction curve of bean leaves. Biochim Biophys Acta 765: 275–281
Butler WL (1978) Energy distribution in the photochemical apparatus of photosynthesis. Ann Rev Plant Physiol 29: 345–378
Demmig-Adams B, Adams III W, Barker D, Logan B, Bowing D and Verhoeven A (1996) Using chlorophyll fluorescence to assess the fraction of absorbed light allocated to thermal dissipation of excess excitation. Physiol Plant 98: 253–264
Duysens LNM (1986) Introduction to (Bactrerio)chlorophyll emission: a historical perspective. In: Govindjee, Amesz J and Fork DC (eds) Light Emission by Plants and Bacteria, pp 3–26.Academic Press, Orlando, Florida
Edwards GE and Baker NR (1993) Can CO2 assimilation in maize leaves be predicted accurately from chlorophyll fluorescence analysis? Photosynth Res 37: 89–102
Genty B, Briantais J-M and Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990: 87–92
Genty B, Harbinson J, Briantais J-M and Baker NR (1990) The relationship between non-photochemical quenching of chlorophyll fluorescence and the rate of Photosystem 2 photochemistry in leaves. Photosynth Res 25: 249–257
Govindjee (1995) Sixty-three years since Kautsky: chlorophyll a fluorescence. Aust J Plant Physiol 22: 20–29
Hoppe W, Lohmann W, Markl H and Zeigler H (eds) (1983) Biophysics. Springer-Verlag, Berlin
Joliot A and Joliot P (1964) Etude cinétique de la reaction photochimique libérant l'oxygèn au cours de la photosynthèse. CR Acad Sci (Paris) 143: 4622–4625
Kato M, Hikosaka K, Hirotsu N, Makino A and Hirose T (2003) The excess light energy that is neither utilized in photosynthesis nor dissipated by photoprotective mechanisms determines the rate of photoinactivation in Photosystem II. Plant Cell Physiol 44: 318–325
Kitajima M and Butler WL (1975) Excitation spectra for Photosystem I and Photosystem II in chloroplasts and the spectral characteristics of the distribution of quanta between the two photosystems. Biochim Biophys Acta 408: 297–305
Krall JP and Edwards GE (1990) Quantum yields of Photosystem II electron transport and carbon dioxide fixation in C4 plants. Aust J Plant Physiol 17: 579–588
Krall JP, Edwards GE and Ku MSB (1991) Quantum yield of Photosystem II and efficiency of CO2 fixation in Flaveria (Asteraceae) species under varying light and CO2. Aust J Plant Physiol 18: 369–383
Kramer DM and Crofts AR (1996) Control of photosynthesis and measurement of photosynthetic reactions in intact plants. In: Baker N (ed) Photosynthesis and the Environment. Advances in Photosynthesis, pp 25–66. Kluwer Academic Publishers, Dordrecht, The Netherlands
Krause G, Vernotte C and Briantais J (1982) Photoinduced quenching of chlorophyll fluorescence in intact chloroplasts and algae.Resolution into two components. Biochim Biophys Acta 679: 116–124
Krause GH and Weis E (1991) Chlorophyll fluorescence and photosynthesis: the basics. Annu Rev Plant Phys Plant Mol Biol 42: 313–349
Laisk A and Edwards GE (1997) CO2 and temperature-dependent induction in C4 photosynthesis: an approach to the hierarchy of rate-limiting processes. Aust J Plant Physiol 24: 505–516
Laisk A, Oja V, Rasulov B, Eichelmann H and Sumberg A (1997) Quantum yields and rate constants of photochemical and nonphotochemical excitation quenching. Plant Physiol 115: 803–815
Lavergne J and Trissl HW (1995) Theory of fluorescence induction in Photosystem II: derivation of analytical expressions in a model including exciton–radical-pair equilibrium and restricted energy transfer between photosynthetic units. Biophys J 68: 2474–2492
Lazár D (1999) Chlorophyll a fluorescence induction. Biochim Biophys Acta 1412: 1–28
Li X-P, Muller-Moule P, Gilmore AM and Niyogi KK (2002) PsbS-dependent enhancement of feedback de-excitation protects Photosystem II from photoinhibition. PNAS 99: 15222–15227
Maxwell K and Johnson GN (2000) Chlorophyll fluorescence – a practical guide. J Exp Bot 51: 659–668
Oxborough K and Baker NR (1997) Resolving chlorophyll fluo-rescenceimages of photosynthetic efficiency into photochemicaland non-photochemical components -calculation of qP and F ′v /F ′m without measuring F ′O Photosynth Res 54: 135–142
Quick W and Horton P (1984) Studies on the induction of chlorophyll fluorescence in barley protoplasts. II Resolution of fluorescence quenching by redox state and the transthylakoid pH gradient. Proc R Soc London B 220: 371–382
Ruuska S, Badger M, Andrews T and von Caemmerer S (2000) Photosynthetic electron sinks in transgenic tobacco with reduced amounts of Rubisco: little evidence for significant Mehler reaction. J Exp Bot 51: 357–368
Schreiber U (1986) Detection of rapid induction kinetics with a new type of high-frequency modulated chlorophyll fluorometer.Photosynth Res 9: 261–272
Schreiber U, Schliwa U and Bilger W (1986) Continuous recording of photochemical and non-photochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer.Photosynth Res 10: 51–62
Schreiber U, Bilger W and Neubauer C (1994) Chlorophyll fluorescence as a nonintrusive indicator for rapid assessment of in vivo photosynthesis. In: Schulze E-D, Caldwell M (eds) Ecophysiology of Photosynthesis, pp 49–70 Springer-Verlag, Berlin
van Kooten O and Snel J (1990) The use of chlorophyll fluorescence nomenclature in plant stress physiology. Photosynth Res 25: 147–150
Vassiliev IR, Kolber Z, Wyman KD, Mauzerall D, Shukla VK and Falkowski PG (1995) Effects of iron limitation on Photosystem II composition and light utilization in Dunaliella tertiolecta. Plant Physiol 109: 963–972
Vavilin DV, Tyystjarvi E and Aro EM (1998) Model for the fluorescence induction curve of photoinhibited thylakoids. Biophys J 75: 503–512
Vredenberg WJ and Duysens LNM (1965) Light-induced changes in absorbancy and fluorescence of chlorophyllous pigments associated with the pigment systems 1 and 2 in blue-green algae.Biochim Biophys Acta 94: 355–370
Weis E and Berry J (1987) Quantum efficiency of PS2 in relation to 'energy' dependent quenching of chlorophyll fluorescence.Biochim Biophys Acta 894: 198–208
Weis E and Lechtenberg D (1989) Fluorescence analysis during steady-state photosynthesis. Phil Trans R Soc London 323: 253–268
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kramer, D.M., Johnson, G., Kiirats, O. et al. New Fluorescence Parameters for the Determination of QA Redox State and Excitation Energy Fluxes. Photosynthesis Research 79, 209–218 (2004). https://doi.org/10.1023/B:PRES.0000015391.99477.0d
Issue Date:
DOI: https://doi.org/10.1023/B:PRES.0000015391.99477.0d