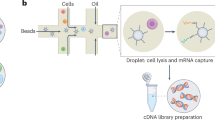
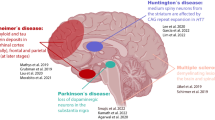
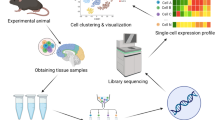
Abstract
Technical and experimental advances in microaspiration techniques, RNA amplification, quantitative real-time polymerase chain reaction (qPCR), and cDNA microarray analysis have led to an increase in the number of studies of single-cell gene expression. In particular, the central nervous system (CNS) is an ideal structure to apply single-cell gene expression paradigms. Unlike an organ that is composed of one principal cell type, the brain contains a constellation of neuronal and noneuronal populations of cells. A goal is to sample gene expression from similar cell types within a defined region without potential contamination by expression profiles of adjacent neuronal subpopulations and noneuronal cells. The unprecedented resolution afforded by single-cell RNA analysis in combination with cDNA microarrays and qPCR-based analyses allows for relative gene expression level comparisons across cell types under different experimental conditions and disease states. The ability to analyze single cells is an important distinction from global and regional assessments of mRNA expression and can be applied to optimally prepared tissues from animal models as well as postmortem human brain tissues. This focused review illustrates the potential power of single-cell gene expression studies within the CNS in relation to neurodegenerative and neuropsychiatric disorders such as Alzheimer's disease (AD) and schizophrenia, respectively.
Similar content being viewed by others
References
Ginsberg, S. D., Hemby, S. E., Lee, V. M-Y., Eberwine, J. H., and Trojanowski, J. Q. 2000. Expression profile of transcripts in Alzheimer's disease tangle-bearing CA1 neurons. Ann. Neurol. 48:77–87.
Ginsberg, S. D., Crino, P. B., Hemby, S. E., Weingarten, J. A., Lee, V. M-Y., Eberwine, J. H., and Trojanowski, J. Q. 1999. Predominance of neuronal mRNAs in individual Alzheimer's disease senile plaques. Ann. Neurol. 45:174–181.
Ginsberg, S. D. and Che, S. 2002. RNA amplification in brain tissues. Neurochem. Res. 27:981–992.
Mufson, E. J., Counts, S. E., and Ginsberg, S. D. 2002. Single cell gene expression profiles of nucleus basalis cholinergic neurons in Alzheimer's disease. Neurochem. Res. 27:1035–1048.
Hemby, S. E., Ginsberg, S. D., Brunk, B., Arnold, S. E., Trojanowski, J. Q., and Eberwine, J. H. 2002. Gene expression profile for schizophrenia: discrete neuron transcription patterns in the entorhinal cortex. Arch. Gen. Psychiat. 59:631–640.
Hemby, S. E., Trojanowski, J. Q., and Ginsberg, S. D. 2003. Neuron-specific age-related decreases in dopamine receptor subtype mRNAs. J. Comp. Neurol. 456:176–183.
Van Deerlin, V. M. D., Ginsberg, S. D., Lee, V. M-Y., and Trojanowski, J. Q. (2002). The use of fixed human post mortem brain tissue to study mRNA expression in neurodegenerative diseases: applications of microdissection and mRNA amplification. Pages 201–235, in Microarrays for the neurosciences: an essential guide, Geschwind, D. H. and Gregg, J. P, (eds.), MIT Press: Boston.
Van Deerlin, V. M. D., Ginsberg, S. D., Lee, V. M-Y., and Trojanowski, J. Q. (2000). Fixed post mortem brain tissue for mRNA expression analysis in neurodegenerative diseases. Pages 118–128, in Geschwind, D. H. (ed.). DNA microarrays: the new frontier in gene discovery and gene expression analysis, Society for Neuroscience: Washington, DC.
Bahn, S., Augood, S. J., Ryan, M., Standaert, D. G., Starkey, M., and Emson, P. C. 2001. Gene expression profiling in the postmortem human brain—no cause for dismay. J. Chem. Neuroanat. 22:79–94.
Leonard, S., Logel, J., Luthman, D., Casanova, M., Kirch, D., and Freedman, R. 1993. Biological stability of mRNA isolated from human postmortem brain collections. Biol. Psychiatry 33:456–466.
von Bertalanffy, L. and Bickis, I. 1956. Identification of cytoplasmic basophilia (ribonucleic acid) by fluorescence microscopy. J. Histochem. Cytochem. 4:481–493.
Mikel, U. V. and Becker Jr., R. L. 1991. A comparative study of quantitative stains for DNA in image cytometry. Analyt. Quant. Cytol. Histol. 13:253–260.
Mai, J. K., Schmidt-Kastner, R., and Tefett, H-B. 1984. Use of acridine orange for histologic analysis of the central nervous system. J. Histochem. Cytochem. 32:97–104.
Sarnat, H. B., Curry, B., Rewcastle, N. B., and Trevenen, C. L. 1986. Cytoplasmic RNA in nervous system tumors in children: a fluorochromic histochemical study using acridine orange. Can. J. Neurol. Sci. 13:31–41.
Ginsberg, S. D., Crino, P. B., Lee, V. M-Y., Eberwine, J. H., and Trojanowski, J. Q. 1997. Sequestration of RNA in Alzheimer's disease neurofibrillary tangles and senile plaques. Ann. Neurol. 41:200–209.
Ginsberg, S. D., Galvin, J. E., Chiu, T-S., Lee, V. M-Y., Masliah, E., and Trojanowski, J. Q. 1998. RNA sequestration to pathological lesions of neurodegenerative disorders. Acta Neuropathol. 96:487–494.
Sarnat, H. B., Curry, B., Rewcastle, N. B., and Trevenen, C. L. 1987. Gliosis and glioma distinguished by acridine orange. Can. J. Neurol. Sci. 14:31–35.
Dickey, C. A., Loring, J. F., Montgomery, J., Gordon, M. N., Eastman, P. S., and Morgan, D. 2003. Selectively reduced expression of synaptic plasticity-related genes in amyloid precursor protein + presenilin-1 transgenic mice. J. Neurosci. 23:5219–5226.
Mandel, S., Weinreb, O., and Youdim, M. 2003. Using cDNA microarray to assess Parkinson's disease models and the effects of neuroprotective drugs. Trends Pharmacol. Sci. 24:184–191.
Grunblatt, E., Mandel, S., Maor, G., and Youdim, M. B. 2001. Gene expression analysis in N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mice model of Parkinson's disease using cDNA microarray: effect of R-apomorphine. J. Neurochem. 78:1–12.
Zhao, X., Lein, E. S., He, A., Smith, S. C., Aston, C., and Gage, F. H. 2001. Transcriptional profiling reveals strict boundaries between hippocampal subregions. J. Comp. Neurol. 441:187–196.
Reyes, T. M., Walker, J. R., DeCino, C., Hogenesch, J. B., and Sawchenko, P. E. 2003. Categorically distinct acute stressors elicit dissimilar transcriptional profiles in the paraventricular nucleus of the hypothalamus. J. Neurosci. 23:5607–5616.
Loring, J. F., Wen, X., Lee, J. M., Seilhamer, J., and Somogyi, R. 2001. A gene expression profile of Alzheimer's disease. DNA Cell Biol. 20:683–695.
Colangelo, V., Schurr, J., Ball, M. J., Pelaez, R. P., Bazan, N. G., and Lukiw, W. J. 2002. Gene expression profiling of 12633 genes in Alzheimer hippocampal CA1: transcription and neurotrophic factor down-regulation and up-regulation of apoptotic and pro-inflammatory signaling. J. Neurosci. Res. 70:462–473.
Malaspina, A., Kaushik, N., and de Belleroche, J. 2001. Differential expression of 14 genes in amyotrophic lateral sclerosis spinal cord detected using gridded cDNA arrays. J. Neurochem. 77:132–145.
Carmel, J. B., Galante, A., Soteropoulos, P., Tolias, P., Recce, M., Young, W., and Hart, R. P. 2001. Gene expression profiling of acute spinal cord injury reveals spreading inflammatory signals and neuron loss. Physiol. Genomics 7:201–213.
Yoshihara, T., Ishigaki, S., Yamamoto, M., Liang, Y., Niwa, J., Takeuchi, H., Doyu, M., and Sobue, G. 2002. Differential expression of inflammation-and apoptosis-related genes in spinal cords of a mutant SOD1 transgenic mouse model of familial amyotrophic lateral sclerosis. J. Neurochem. 80:158–167.
Lock, C., Hermans, G., Pedotti, R., Brendolan, A., Schadt, E., Garren, H., Langer-Gould, A., Strober, S., Cannella, B., Allard, J., Klonowski, P., Austin, A., Lad, N., Kaminski, N., Galli, S. J., Oksenberg, J. R., Raine, C. S., Heller, R., and Steinman, L. 2002. Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat. Med. 8:500–508.
Mycko, M. P., Papoian, R., Boschert, U., Raine, C. S., and Selmaj, K. W. 2003. cDNA microarray analysis in multiple sclerosis lesions: detection of genes associated with disease activity. Brain 126:1048–1057.
Mimmack, M. L., Ryan, M., Baba, H., Navarro-Ruiz, J., Iritani, S., Faull, R. L., McKenna, P. J., Jones, P. B., Arai, H., Starkey, M., Emson, P. C., and Bahn, S. 2002. Gene expression analysis in schizophrenia: reproducible up-regulation of several members of the apolipoprotein L family located in a high-susceptibility locus for schizophrenia on chromosome 22. Proc. Natl. Acad. Sci. USA 99: 4680–4685.
Middleton, F. A., Mirnics, K., Pierri, J. N., Lewis, D. A., and Levitt, P. 2002. Gene expression profiling reveals alterations of specific metabolic pathways in schizophrenia. J. Neurosci. 22: 2718–2729.
Hakak, Y., Walker, J. R., Li, C., Wong, W. H., Davis, K. L., Buxbaum, J. D., Haroutunian, V., and Fienberg, A. A. 2001. Genome-wide expression analysis reveals dysregulation of myelination-related genes in chronic schizophrenia. Proc. Natl. Acad. Sci. USA 98:4746–4751.
Mirnics, K., Middleton, F. A., Marquez, A., Lewis, D. A., and Levitt, P. 2000. Molecular characterization of schizophrenia viewed by microarray analysis of gene expression in prefrontal cortex. Neuron 28:53–67.
Ginsberg, S. D. 2001. Gene expression profiling using single cell microdissection combined with cDNA microarrays. Pages 61–70, in Geschwind, D. H. (ed.). DNA microarrays: the new frontier in gene discovery and gene expression analysis., Society for Neuroscience Press, Washington DC.
Bonner, R. F., Emmert-Buck, M., Cole, K., Pohida, T., Chuaqui, R., Goldstein, S., and Liotta, L. A. 1997. Laser capture microdissection: molecular analysis of tissue. Science 278:1481–1483.
Emmert-Buck, M. R., Bonner, R. F., Smith, P. D., Chuaqui, R. F., Zhuang, Z., Goldstein, S. R., Weiss, R. A., and Liotta, L. A. 1996. Laser capture microdissection. Science 274:998–1001.
Fend, F., Emmert-Buck, M. R., Chuaqui, R., Cole, K., Lee, J., Lotto, L. A., and Raffeld, M. 1999. Immuno-LCM: laser capture microdis-section of immunostained frozen sections for mRNA analysis. Am. J. Pathol. 154:61–66.
Goldsworthy, S. M., Stockton, P. S., Trempus, C. S., Foley, J. F., and Maronpot, R. R. 1999. Effects of fixation on RNA extraction and amplification from laser capture microdissected tissue. Mol. Carcinog. 25:86–91.
Simone, N. L., Remaley, A. T., Charboneau, L., Petricoin, E. F., 3rd, Glickman, J. W., Emmert-Buck, M. R., Fleisher, T. A., and Liotta, L. A. 2000. Sensitive immunoassay of tissue cell proteins procured by laser capture microdissection. Am. J. Pathol. 156: 445–452.
Suarez-Quian, C. A., Goldstein, S. R., Pohida, T., Smith, P. D., Peterson, J. I., Wellner, E., Ghany, M., and Bonner, R. F. 1999. Laser capture microdissection of single cells from complex tissues. BioTechniques 26:328–335.
Crino, P. B., Khodakhah, K., Becker, K., Ginsberg, S. D., Hemby, S., and Eberwine, J. H. 1998. Presence and phosphorylation of transcription factors in dendrites. Proc. Natl. Acad. Sci. USA 95:2313–2318.
Che, S. and Ginsberg, S. D. 2004. Amplification of transcripts using terminal continuation. Lab. Invest. 84:233–242.
Kacharmina, J. E., Crino, P. B., and Eberwine, J. 1999. Preparation of cDNA from single cells and subcellular regions. Methods Enzymol. 303:3–18.
Sambrook, J., Fritsch, E., and Maniatis, T. 1989. Molecular cloning: a laboratory manual. Second edition. Cold Spring Harbor Laboratory Press, Cold Spring Harbor.
Kadkol, S. S., Gage, W. R., and Pasternack, G. R. 1999. In situ hybridization—theory and practice. Mol. Diagn. 4:169–183.
Hashimoto, T., Volk, D. W., Eggan, S. M., Mirnics, K., Pierri, J. N., Sun, Z., Sampson, A. R., and Lewis, D. A. 2003. Gene expression deficits in a subclass of GABA neurons in the prefrontal cortex of subjects with schizophrenia. J. Neurosci. 23:6315–6326.
Brail, L. H., Jang, A., Billia, F., Iscove, N. N., Klamut, H. J., and Hill, R. P. 1999. Gene expression in individual cells: analysis using global single cell reverse transcription polymerase chain reaction (GSC RT-PCR). Mutat. Res. 406:45–54.
Holland, P. M., Abramson, R. D., Watson, R., and Gelfand, D. H. 1991. Detection of specific polymerase chain reaction product by utilizing the 5′—3′ exonuclease activity of Thermus aquaticus DNA polymerase. Proc. Natl. Acad. Sci. USA 88:7276–7280.
Cardullo, R. A., Agrawal, S., Flores, C., Zamecnik, P. C., and Wolf, D. E. 1988. Detection of nucleic acid hybridization by nonradiative fluorescence resonance energy transfer. Proc. Natl. Acad. Sci. USA 85:8790–8794.
Bustin, S. A. 2002. Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J. Mol. Endocrinol. 29:23–39.
Eberwine, J., Yeh, H., Miyashiro, K., Cao, Y., Nair, S., Finnell, R., Zettel, M., and Coleman, P. 1992. Analysis of gene expression in single live neurons. Proc. Natl. Acad. Sci. USA 89:3010–3014.
Eberwine, J., Kacharmina, J. E., Andrews, C., Miyashiro, K., McIntosh, T., Becker, K., Barrett, T., Hinkle, D., Dent, G., and Marciano, P. 2001. mRNA expression analysis of tissue sections and single cells. J. Neurosci. 21:8310–8314.
Brown, P. O. and Botstein, D. 1999. Exploring the new world of the genome with DNA microarrays. Nat. Genet. 21(Suppl):33–37.
Eisen, M. B. and Brown, P. O. 1999. DNA arrays for analysis of gene expression. Methods Enzymol. 303:179–205.
Lockhart, D. J., Dong, H., Byrne, M. C., Follettie, M. T., Gallo, M. V., Chee, M. S., Mittmann, M., Wang, C., Kobayashi, M., Horton, H., and Brown, E. L. 1996. Expression monitoring by hybridization to high-density oligonucleotide arrays. Nat. Biotechnol. 14:1675–1680.
Gygi, S. P., Rochon, Y., Franza, B. R., and Aebersold, R. 1999. Correlation between protein and mRNA abundance in yeast. Mol. Cell. Biol. 19:1720–1730.
Ginsberg, S. D., Schmidt, M. L., Crino, P. B., Eberwine, J. H., Lee, V. M-Y., and Trojanowski, J. Q., 1999. Molecular pathology of Alzheimer's disease and related disorders. Pages 603–653, in Cerebral cortex, vol. 14: neurodegenerative and age-related changes in structure and function of cerebral cortex, Peters, A. and Morrison, J. H. (eds.), Kluwer Academic/Plenum, New York.
Harrison, P. J., Barton, A. J., Najlerahim, A., McDonald, B., and Pearson, R. C. 1991. Regional and neuronal reductions of polyadenylated messenger RNA in Alzheimer's disease. Psychol. Med. 21:855–866.
Griffin, W. S., Ling, C., White, C. L., and Morrison-Bogorad, M. 1990. Polyadenylated messenger RNA in paired helical filament-immunoreactive neurons in Alzheimer disease. Alz. Dis. Assoc. Dis. 4:69–78.
Joyce, J. N., Kaeger, C., Ryoo, H., and Goldsmith, S. 1993. Dopamine D2 receptors in the hippocampus and amygdala in Alzheimer's disease. Neurosci. Lett. 154:171–174.
Ryoo, H. L. and Joyce, J. N. 1994. Loss of dopamine D2 receptors varies along the rostrocaudal axis of the hippocampal complex in Alzheimer's disease. J. Comp. Neurol. 348:94–110.
Cataldo, A. M., Barnett, J. L., Berman, S. A., Li, J., Quarless, S., Bursztajn, S., Lippa, C., and Nixon, R. A. 1995. Gene expression and cellular content of cathepsin D in Alzheimer's disease brain: evidence for early up-regulation of the endosomal-lysosomal system. Neuron 14:671–680.
Cataldo, A. M., Hamilton, D. J., Barnett, J. L., Paskevich, P. A., and Nixon, R. A. 1996. Properties of the endosomal-lysosomal system in the human central nervous system: disturbances mark most neurons in populations at risk to degenerate in Alzheimer's disease. J. Neurosci. 16:186–199.
Nixon, R. A., Cataldo, A. M., and Mathews, P. M. 2000. The endosomal-lysosomal system of neurons in Alzheimer's disease pathogenesis: a review. Neurochem. Res. 25:1161–1172.
Goedert, M., Trojanowski, J. Q., and Lee, V. M-Y. 1997. The neurofibrillary pathology of Alzheimer's disease. Pages 613–627, in The Molecular and Genetic Basis of Neurological Disease, Second ed., Rosenberg, R. N., et al., (eds.), Butterworth-Heinemann, Boston.
Selkoe, D. J. 1997. Alzheimer's disease: genotypes, phenotypes, and treatments. Science 275:630–631.
Itagaki, S., McGeer, P. L., Akiyama, H., Zhu, S., and Selkoe, D. 1989. Relationship of microglia and astrocytes to amyloid deposits of Alzheimer disease. J. Neuroimmunol. 24:173–182.
Eikelenboom, P. and Veerhuis, R. 1996. The role of complement and activated microglia in the pathogenesis of Alzheimer's disease. Neurobiol. Aging 17:673–680.
Mesulam, M. M., Mufson, E. J., Levey, A. I., and Wainer, B. H. 1983. Cholinergic innervation of cortex by the basal forebrain: cytochemistry and cortical connections of the septal area, diagonal band nuclei, nucleus basalis (substantia innominata), and hypothalamus in the rhesus monkey. J. Comp. Neurol. 214:170–197.
Bartus, R. T., Dean, R. L., 3rd, Beer, B., and Lippa, A. S. 1982. The cholinergic hypothesis of geriatric memory dysfunction. Science. 217:408–414.
Baxter, M. G. and Chiba, A. A. 1999. Cognitive functions of the basal forebrain. Curr. Opin. Neurobiol. 9:178–183.
Whitehouse, P. J., Price, D. L., Struble, R. G., Clark, A. W., Coyle, J. T., and Delong, M. R. 1982. Alzheimer's disease and senile dementia: loss of neurons in the basal forebrain. Science 215:1237–1239.
Davies, P. and Maloney, A. J. 1976. Selective loss of central cholinergic neurons in Alzheimer's disease. Lancet 2:1403.
Mufson, E. J., Bothwell, M., and Kordower, J. H. 1989. Loss of nerve growth factor receptor-containing neurons in Alzheimer's disease: a quantitative analysis across subregions of the basal forebrain. Exp. Neurol. 105:221–232.
Bierer, L. M., Haroutunian, V., Gabriel, S., Knott, P. J., Carlin, L. S., Purohit, D. P., Perl, D. P., Schmeidler, J., Kanof, P., and Davis, K. L. 1995. Neurochemical correlates of dementia severity in Alzheimer's disease: relative importance of the cholinergic deficits. J. Neurochem. 64:749–760.
Counts, S. E., Perez, S. E., Ginsberg, S. D., de Lacalle, S., and Mufson, E. J. 2003. Galanin plasticity and Alzheimer's disease. Molecular Interventions 3:23–40.
Cullen, K. M. and Halliday, G. M. 1998. Neurofibrillary degeneration and cell loss in the nucleus basalis in comparison to cortical Alzheimer pathology. Neurobiol. Aging 19:297–306.
Sassin, I., Schultz, C., Thal, D. R., Rub, U., Arai, K., Braak, E., and Braak, H. 2000. Evolution of Alzheimer's disease-related cytoskeletal changes in the basal nucleus of Meynert. Acta Neuropathol. (Berl). 100:259–69.
Struble, R. G., Cork, L. C., Whitehouse, P. J., and Price, D. L. 1982. Cholinergic innervation in neuritic plaques. Science 216: 413–415.
German, D. C., Yazdani, U., Speciale, S. G., Pasbakhsh, P., Games, D., and Liang, C. L. 2003. Cholinergic neuropathology in a mouse model of Alzheimer's disease. J. Comp. Neurol. 462:371–381.
Buxbaum, J. D., Thinakaran, G., Koliatsos, V., O'Callahan, J., Slunt, H. H., Price, D. L., and Sisodia, S. S. 1998. Alzheimer amyloid protein precursor in the rat hippocampus: transport and processing through the perforant path. J. Neurosci. 18:9629–9637.
Sheng, J. G., Price, D. L., and Koliatsos, V. E. 2002. Disruption of corticocortical connections ameliorates amyloid burden in terminal fields in a transgenic model of Abeta amyloidosis. J. Neurosci. 22:9794–9799.
Lazarov, O., Lee, M., Peterson, D. A., and Sisodia, S. S. 2002. Evidence that synaptically released beta-amyloid accumulates as extracellular deposits in the hippocampus of transgenic mice. J. Neurosci. 22:9785–9793.
Lencz, T., Cornblatt, B., and Bilder, R. M. 2001. Neurodevelopmental models of schizophrenia: pathophysiologic synthesis and directions for intervention research. Psychopharmacol. Bull. 35:95–125.
Johns, L. C. and van Os, J. 2001. The continuity of psychotic experiences in the general population. Clin Psychol Rev. 21:1125–1141.
Lewis, D. A. 2002. In pursuit of the pathogenesis and pathophysiology of schizophrenia: where do we stand? Am. J. Psychiatry 159:1467–1469.
Kurtz, M. M., Moberg, P. J., Gur, R. C., and Gur, R. E. 2001. Approaches to cognitive remediation of neuropsychological deficits in schizophrenia: a review and meta-analysis. Neuropsychol. Rev. 11:197–210.
Gur, R. C. and Gur, R. E. 2002. Neuroimaging applications in elderly patients. Am. J. Geriatr. Psychiatry 10:5–11.
Arnold, S. E., Trojanowski, J. Q., Gur, R. E., Blackwell, P., Han, L. Y., and Choi, C. 1998. Absence of neurodegeneration and neural injury in the cerebral cortex in a sample of elderly patients with schizophrenia. Arch. Gen. Psychiatry 55:225–232.
Trojanowski, J. Q. and Arnold, S. E. 1995. In pursuit of the molecular neuropathology of schizophrenia. Arch. Gen. Psychiatry 52: 274–276.
Blalock, E. M., Chen, K. C., Sharrow, K., Herman, J. P., Porter, N. M., Foster, T. C., and Landfield, P. W. 2003. Gene microarrays in hippocampal aging: statistical profiling identifies novel processes correlated with cognitive impairment. J. Neurosci. 23: 3807–3819.
Prolla, T. A. 2002. DNA microarray analysis of the aging brain. Chem. Senses 27:299–306.
Bannon, M. J., Poosch, M. S., Xia, Y., Goebel, D. J., Cassin, B., and Kapatos, G. 1992. Dopamine transporter mRNA content in human substantia nigra decreases precipitously with age. Proc. Natl. Acad. Sci. USA 89:7095–7099.
Bannon, M. J. and Whitty, C. J. 1997. Age-related and regional differences in dopamine transporter mRNA expression in human midbrain. Neurology. 48:969–977.
Emborg, M. E., Ma, S. Y., Mufson, E. J., Levey, A. I., Taylor, M. D., Brown, W. D., Holden, J. E., and Kordower, J. H. 1998. Age-related declines in nigral neuronal function correlate with motor impairments in rhesus monkeys. J. Comp. Neurol. 401: 253–265.
Ma, S. Y., Roytt, M., Collan, Y., and Rinne, J. O. 1999. Unbiased morphometrical measurements show loss of pigmented nigral neurones with ageing. Neuropathol. Appl. Neurobiol. 25:394–399.
Zola-Morgan, S. and Squire, L. R. 1993. Neuroanatomy of memory. Annu. Rev. Neurosci. 16:547–563.
Zola-Morgan, S., Squire, L. R., and Ramus, S. J. 1994. Severity of memory impairment in monkeys as a function of locus and extent of damage within the medial temporal lobe memory system. Hippocampus 4:483–495.
Akil, M., Edgar, C. L., Pierri, J. N., Casali, S., and Lewis, D. A. 2000. Decreased density of tyrosine hydroxylase-immunoreactive axons in the entorhinal cortex of schizophrenic subjects. Biol. Psychiatry 47:361–370.
Volkow, N. D., Wang, G. J., Fowler, J. S., Logan, J., Gatley, S. J., MacGregor, R. R., Schlyer, D. J., Hitzemann, R., and Wolf, A. P. 1996. Measuring age-related changes in dopamine D2 receptors with 11C-raclopride and 18F-N-methylspiroperidol. Psychiatry Res. 67:11–16.
Volkow, N. D., Ding, Y. S., Fowler, J. S., Wang, G. J., Logan, J., Gatley, S. J., Hitzemann, R., Smith, G., Fields, S. D., and Gur, R. 1996. Dopamine transporters decrease with age. J. Nucl. Med. 37:554–559.
Volkow, N. D., Wang, G. J., Fowler, J. S., Ding, Y. S., Gur, R. C., Gatley, J., Logan, J., Moberg, P. J., Hitzemann, R., Smith, G., and Pappas, N. 1998. Parallel loss of presynaptic and postsynaptic dopamine markers in normal aging. Ann. Neurol. 44:143–147.
Rinne, J. O., Lonnberg, P., and Marjamaki, P. 1990. Age-dependent decline in human brain dopamine D1 and D2 receptors. Brain Res. 508:349–352.
Amenta, F., Mignini, F., Ricci, A., Sabbatini, M., Tomassoni, D., and Tayebati, S. K. 2001. Age-related changes of dopamine receptors in the rat hippocampus: a light microscope autoradiography study. Mech. Ageing Dev. 122:2071–2083.
Kemppainen, N., Laine, M., Laakso, M. P., Kaasinen, V., Nagren, K., Vahlberg, T., Kurki, T., and Rinne, J. O. 2003. Hippocampal dopamine D2 receptors correlate with memory functions in Alzheimer's disease. Eur. J. Neurosci. 18:149–154.
Kaasinen, V. and Rinne, J. O. 2002. Functional imaging studies of dopamine system and cognition in normal aging and Parkinson's disease. Neurosci. Biobehav. Rev. 26:785–793.
Colantuoni, C., Purcell, A. E., Bouton, C. M., and Pevsner, J. 2000. High throughput analysis of gene expression in the human brain. J. Neurosci. Res. 59:1–10.
Serafini, T. 1999. Of neurons and gene chips. Curr. Opin. Neurobiol. 9:641–644.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ginsberg, S.D., Elarova, I., Ruben, M. et al. Single-Cell Gene Expression Analysis: Implications for Neurodegenerative and Neuropsychiatric Disorders. Neurochem Res 29, 1053–1064 (2004). https://doi.org/10.1023/B:NERE.0000023593.77052.f7
Issue Date:
DOI: https://doi.org/10.1023/B:NERE.0000023593.77052.f7