Abstract
Crab shells are an important feedstock for chitin production. However, their highly compact structure significantly limits their use for the production of chitin under mild conditions. Here, a green and efficient approach using a natural deep eutectic solvent (NADES) to produce chitin from crab shells was developed. Its effectiveness in isolating chitin was investigated. The results showed that most proteins and minerals were removed from crab shells and the relative crystallinity of the isolated chitin reached 76%. The quality of the obtained chitin was comparable to chitin isolated by the acid–alkali method. This is the first report on a green method for efficient chitin production from crab shells. This study is expected to open new avenues for green and efficient production of chitin from crab shells.
Similar content being viewed by others
Introduction
The global shellfishery industry generates millions of metric tons of waste every year (Yan and Chen 2015). Crustacean shells are a primary source of this waste, causing serious environmental problems and a significant waste of resources. Crustacean shells contain a useful chemical—chitin (Chen et al. 2021). Chitin has received significant attention due to its beneficial characteristics including biocompatibility, renewability, biodegradability, sustainability, bioactivity, and non-toxicity (Duan et al. 2015; Hong et al. 2018; Zhang and Rolandi 2017). It can be applied in many industries, including biomedicine, agriculture, water treatment, and cosmetics (Krajewska 2004; Zhang and Rolandi 2017).
In crustacean shells, the chitin nanofibrils are complex, containing proteins to form long chitin-protein fibers embedded in the mineral matrix (Raabe et al. 2005). Compared to shrimp shell structure, the structure of a crab shell is even more complex and compact because of its higher mineral content. Thus, it is extremely difficult to use biological methods, such as enzymatic reactions and microbial fermentation, to dissociate the chitin-protein-mineral complex to produce chitin from crab shells. Moreover, the conventional acid–alkali approach for chitin production is considered harmful to the environment. Hence, a green method for isolating chitin from crab shells is urgently needed.
Natural deep eutectic solvents (NADESs) are being increasingly considered as green and sustainable solvents. NADESs consist of natural compounds, especially primary metabolites including organic acids, sugars, and amino acids (Dai et al. 2013; Guo et al. 2019; Paiva et al. 2014). NADESs are distinguished from conventional solvents due to their favorable properties including biocompatibility, sustainability, biodegradability, non-volatility, low cost, and simple preparation methods (Xin et al. 2017).
Here, we developed a chitin isolation method using a NADES consisting of choline chloride and malic acid, to separate chitin from crab shells. In our previous work, we developed a NADES-based approach to isolate chitin from shrimp shells (Huang et al. 2018). To the best of our knowledge, this is the first report on a green chemical approach for chitin production from crab shells. The isolated chitin was characterized and the reusability of the NADES was evaluated.
Results and discussion
Preparation of the NADES
The NADES used in this study was composed of choline chloride and malic acid, associated by hydrogen bonding to form a eutectic mixture.
Chitin isolation from crab shells
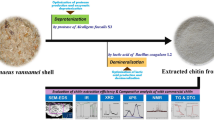
A schematic diagram of the NADES-based chitin isolation is presented in Fig. 1A. Chitin isolation by the NADES was evaluated with various crab shell/NADES ratios (1:10, 1:20, and 1:30) and microwave irradiation times (3, 5, 7, 9, and 11 min). As seen in Fig. 1C, the demineralization and deproteinization efficiency increased with the crab shell/NADES ratio from 1:10 to 1:30. Time for microwave irradiation is another factor that can influence the chitin isolation. The demineralization and deproteinization efficiency increased when the irradiation time was increased. This result indicates that a larger amount of NADES and a longer irradiation time are beneficial for the removal of minerals and proteins from crab shells. The maximal demineralization and deproteinization efficiency reached 99.8% and 92.3%, respectively, at the crab shell/NADES ratio of 1:30 after 11 min of microwave irradiation.
A Schematic diagram of the chitin isolation from crab shells by the NADES. B SEM images of the crab shells, chitin isolated using the acid–alkali, and chitin isolated using the NADES. C Demineralization and deproteinization efficiency at crab shell/NADES ratios of 1:10, 1:20, and 1:30. D Reusability of the NADES. E FT-IR spectra of the crab shells, chitin isolated by acid–alkali, and chitin isolated by the NADES; XRD curves of the crab shells, CaCO3, chitin isolated by acid–alkali, and chitin isolated by the NADES; TG curves of the crab shells, chitin isolated by acid–alkali, and chitin isolated by the NADES
Scanning electron microscopy
The morphologies of the isolated chitin obtained by the NADES and acid–alkali, and the raw crab shells were observed by SEM. As seen in Fig. 1B, remarkable morphological changes were observed between the isolated chitin and raw crab shell. The morphologies of the chitin isolated by the NADES and acid–alkali were similar. The surface of the raw crab shells was rough with pores observed. In contrast, in the chitin isolated by NADES treatment, pores were clearly observed among the microfibrils due to the absence of proteins and minerals.
Fourier transform infrared spectroscopy
Fourier transform infrared (FT-IR) spectra of the crab shells, and the chitin extracted using the NADES and acid–alkali are presented in Fig. 1E. The peaks of isolated chitin at 3447 cm−1 and 3268 cm−1 can be attributed to symmetric stretching vibration of NH2 and OH groups, respectively (Zhu et al. 2017). The amide I band split at 1660 cm−1 and 1626 cm−1 can be assigned to the existence of the intermolecular (–CO··HN–) and the intramolecular hydrogen bond (–CO··HOCH2–) (Rinaudo 2006). The splitting of the amide-I band was not observed in the crab shells because the amide peaks of protein overlapped with the chitin amide-I bands, indicating isolated chitin was free from protein. These absorbance peaks are the typical feature of α-chitin. No apparent differences were observed between the spectra of the chitin obtained from the NADES and acid–alkali method, suggesting chitin isolated by the NADES and acid–alkali possessed the same chemical structure as the chitin isolated by acid–alkali.
X-ray diffraction
X-ray diffraction (XRD) was used to investigate the crystal structure of the crab shells, calcium carbonate, and isolated chitin. As shown in Fig. 1E, the chitin isolated by the NADES and acid–alkali presented diffraction peaks at approximately at 9.20°, 12.76°, 19.29°, and 26.26°, which confirmed the crystal type of the α-chitin (Zhu et al. 2017). This result indicated that the crystal structure of chitin was not changed by the NADES treatment. The diffraction peak of calcium carbonate at 29.55° was not shown in the samples isolated by the NADES and acid–alkali, indicating NADES can remove calcium carbonate. The XRD pattern of the chitin isolated by the NADES was similar to that of the chitin isolated by the acid–alkali method, suggesting complete removal of calcium carbonate. The CrI was calculated using Segal’s method. The CrI of the chitin isolated by the NADES was 75.55%, while that of the crab shells was 33.13%. The increase of CrI indicated that the NADES-based method can effectively remove minerals and proteins from crab shells.
Thermogravimetric analysis
The results of thermogravimetric analysis (TGA) of crab shells before and after treatment are presented in Fig. 1E. The initial decomposition temperature range of raw crab shells was approximately from 150 to 300 °C, which can be assigned to the presence of proteins. Compared with the crab shells, the chitin extracted by the NADES did not show mass loss between 150 and 330 °C, suggesting proteins were not present along with chitin after treatment with NADES. The absence of mass loss between 600 and 750 °C indicates the isolated chitin was free from calcium carbonate. Additionally, the thermal stability of the chitin isolated by the NADES was similar to that of chitin obtained by acid–alkali.
Reuse of the NADES
When the NADES, consisting of choline chloride and malic acid, is used in chitin isolation, some malic acid is consumed in removal of calcium carbonate. Hence, after each cycle, a certain amount of malic acid needs to be added to maintain the ratio of choline chloride/malic acid. As present in Fig. 1D, the NADES could be used three times without a remarkable decrease in the demineralization and deproteinization efficiency. After three times, the NADES was too viscous to be further used because proteins isolated from crab shells increased the viscosity of the NADES.
Mechanism
Chitin isolation from crab shells using a green approach is extremely difficult because crab shells have a much higher mineral content than other crustacean shells, such as shrimp shells. Because the minerals in crab shells are mostly in the form of crystalline calcium carbonate, when the NADES was applied to the crab shells, minerals were removed by malic acid, leaving chitin and proteins. In crab exoskeletons, the minerals are deposited within the chitin–protein matrix and form a well-defined hierarchical organization (Chen et al. 2008). Thus, the strong internal structure of crab shells was weakened after the removal of minerals. NADESs are capable of breaking hydrogen bonds and have a very high ionic strength (Sharma et al. 2013). The strong hydrogen-bond network between chitin and proteins was weakened due to competing hydrogen bond formed between the chloride ions of the NADES and the hydroxyl groups, and the proteins were removed by dissolution because of the hydrogen-bond interaction with the NADES (Li et al. 2013; Sharma et al. 2013). As a result, chitin was isolated from the crab shells.
Materials and methods
Materials
Choline chloride was purchased from Yuanye Bio-Technology Co., Ltd. Malic acid, HCl, and NaOH were purchased from Sinopharm Chemical Reagent Co., Ltd.
Preparation of the NADES
Choline chloride and malic acids were mixed with a molar ratio of 1:1 at 80 °C until formation of a homogeneous liquid.
Isolation of chitin from crab shells
Chitin isolation from crab shells by the NADES was performed as follows. The ground crab shells and NADES were mixed at various shell/NADES ratios (1:10, 1:20, and 1:30). Next, the shell/NADES mixture was heated by microwave irradiation at 700 W. The isolated chitin was then separated from the mixture by centrifugation. The sample was then rinsed with distilled water followed by drying in a vacuum oven.
For comparison with the NADES-based chitin isolation, acid–alkali-based chitin isolation from crab shells was conducted. Demineralization was performed by treating the crab shells with a 5% (w/v) HCl solution with a HCl solution-to-shell ratio of 10 ml/g at room temperature for 1 h. Next, the demineralized sample was collected by centrifugation followed by deproteinization with 10% (w/v) NaOH with a NaOH solution-to-shell ratio of 10 ml/g at 95 °C for 1 h. The isolated chitin was then rinsed with distilled water followed by drying in a vacuum oven.
The mineral content was measured by heating the samples (1–2 g) at 525 °C to a constant weight in a muffle furnace. The demineralization efficiency was calculated using the following equation:
where M0 and M are the mineral contents of the crab shells and isolated chitin, respectively.
The protein content was determined by the Bradford method (Bradford 1976). Briefly, Coomassie Brilliant Blue G-250 (100 mg) was dissolved in ethanol (95%, 50 ml). Then, phosphoric acid (85%, 100 ml) was added, and the total volume of the solution was adjusted to 1 L with distilled water. Next, a 0.5 g sample was added to 5% NaOH, and the mixture was stirred at 95 °C for 1 h, followed by filtering and dilution. Then, the resulting sample was added to the solution, and the absorbance was measured at 595 nm after 2 min. The deproteinization efficiency was calculated using the following equation:
where P0 and P are the protein contents of the crab shells and isolated chitin, respectively.
To evaluate reusability, the NADES was recycled three times without purification after chitin isolation.
Characterization
The surface morphologies of the crab shells and isolated chitin were observed by SEM (JEOL JSM-840) with an acceleration voltage of 20 kV. Before observation, all the samples were coated with platinum by vacuum sputtering. The FT-IR spectra were recorded on a FT-IR spectrometry (Thermo Scientific Nicolet iS10) over the frequency range of 4000–500 cm−1 with a resolution of 4 cm−1. The XRD patterns were recorded on an X-ray diffractometer (Rigaku Miniflex 600) using Cu Kα radiation at 40 kV. The diffraction data were collected at a scanning rate of 5° min−1 from 2θ = 5 − 60°. The relative crystallinity index (CrI) was calculated by the Segal method:
where I110 is the peak intensity of the diffraction at 2θ ≈ 20°, which represents both the crystalline and amorphous region material, and Iam is the diffraction intensity of the amorphous fraction at 2θ ≈ 18°. TGA was performed under a nitrogen atmosphere at a heating rate of 10 °C min−1 by a thermogravimetric analyzer (NETZSCH TG 209 F3).
Conclusions
In the present study, we developed a NADES-based method for chitin production from crab shells. The experimental results indicated that most protein and minerals were removed, and the quality of the isolated chitin was superior to that isolated by other methods. In addition, the NADES can be reused three times without purification. This method was proved to be green, efficient, facile, and sustainable. The chitin produced by the proposed method was comparable to the chitin isolated by the acid–alkali method. This study provides a method for the sustainable production of chitin from crab shells.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chen P-Y, Lin AY-M, McKittrick J, Meyers MA (2008) Structure and mechanical properties of crab exoskeletons. Acta Biomater 4:587–596
Chen R, Huang W-C, Wang W, Mao X (2021) Characterization of TEMPO-oxidized chitin nanofbers with various oxidation times and its application as an enzyme immobilization support. Mar Life Sci Technol 3:85–93
Dai Y, van Spronsen J, Witkamp G-J, Verpoorte R, Choi YH (2013) Natural deep eutectic solvents as new potential media for green technology. Anal Chim Acta 766:61–68
Duan B, Zheng X, Xia Z, Fan X, Guo L, Liu J, Wang Y, Ye Q, Zhang L (2015) Highly biocompatible nanofibrous microspheres self-assembled from chitin in NaOH/urea aqueous solution as cell carriers. Angew Chem Int Ed 54:5152–5156
Guo N, Ping K, Jiang Y-W, Wang L-T, Niu L-J, Liu Z-M, Fu Y-J (2019) Natural deep eutectic solvents couple with integrative extraction technique as an effective approach for mulberry anthocyanin extraction. Food Chem 296:78–85
Hong M-S, Choi G-M, Kim J, Jang J, Choi B, Kim J-K, Jeong S, Leem S, Kwon H-Y, Hwang H-B, Im H-G, Park J-U, Bae B-S, Jin J (2018) Biomimetic chitin-silk hybrids: an optically transparent structural platform for wearable devices and advanced electronics. Adv Funct Mater 28:1705480
Huang W-C, Zhao D, Guo N, Xue C, Mao X (2018) Green and facile production of chitin from crustacean shells using a natural deep eutectic solvent. J Agric Food Chem 66:11897–11901
Krajewska B (2004) Application of chitin- and chitosan-based materials for enzyme immobilizations: a review. Enzyme Microb Technol 35:126–139
Li C, Li D, Zou S, Li Z, Yin J, Wang A, Cui Y, Yao Z, Zhao Q (2013) Extraction desulfurization process of fuels with ammonium-based deep eutectic solvents. Green Chem 15:2793–2799
Paiva A, Craveiro R, Aroso I, Martins M, Reis RL, Duarte ARC (2014) Natural deep eutectic solvents—solvents for the 21st century. ACS Sustain Chem Eng 2:1063–1071
Raabe D, Sachs C, Romano P (2005) The crustacean exoskeleton as an example of a structurally and mechanically graded biological nanocomposite material. Acta Mater 53:4281–4292
Rinaudo M (2006) Chitin and chitosan: properties and applications. Prog Polym Sci 31:603–632
Sharma M, Mukesh C, Mondal D, Prasad K (2013) Dissolution of alpha-chitin in deep eutectic solvents. RSC Adv 3:18149–18155
Xin R, Qi S, Zeng C, Khan FI, Yang B, Wang Y (2017) A functional natural deep eutectic solvent based on trehalose: Structural and physicochemical properties. Food Chem 217:560–567
Yan N, Chen X (2015) Don’t waste seafood waste. Nature 524:155–157
Zhang X, Rolandi M (2017) Engineering strategies for chitin nanofibers. J Mater Chem B 5:2547–2559
Zhu P, Gu Z, Hong S, Lian H (2017) One-pot production of chitin with high purity from lobster shells using choline chloride-malonic acid deep eutectic solvent. Carbohydr Polym 177:217–223
Acknowledgements
This work was supported by China Agriculture Research System (CARS-48), Taishan Scholar Project of Shandong Province (tsqn201812020), Fundamental Research Funds for the Central Universities (201941002).
Author information
Authors and Affiliations
Contributions
WCH conceived the idea and wrote the manuscript. DZ performed the experiments. CX and XM supervised the work. WCH and DZ contributed equally to this work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Animal and human rights statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Edited by Xin Yu.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, WC., Zhao, D., Xue, C. et al. An efficient method for chitin production from crab shells by a natural deep eutectic solvent. Mar Life Sci Technol 4, 384–388 (2022). https://doi.org/10.1007/s42995-022-00129-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42995-022-00129-y