Abstract
Sustainable Urban Drainage Systems (SUDS) are commonly used to control flooding in urban areas. These structures store and treat stormwater runoff. Several studies in high-income countries have reported the presence of pathogens in runoff water, but it is expected that runoff water in developing countries contains higher pathogen concentrations given their lack of resources to properly manage sewage; this could result in higher risks of infection for people interacting with SUDS. In this study, we investigated pathogen concentrations (i.e., Salmonella spp. and E. Coli O157) at the micropool of a SUDS train composed of a grassed swale followed by a dry extended detention basin in Bogotá (Colombia) during a 25-week period. We also estimated the risk of infection with the analyzed pathogens, given the high level of exposure to the detention structure. Additionally, we investigated if any of the physicochemical or meteorological variables were associated with pathogen concentrations at the site. We found that pathogen concentrations greatly exceeded concentrations reported for stormwater runoff in developed countries, namely 1562 CFU/mL, on average, for Salmonella spp. and 9160 CFU/mL, on average, for E. Coli O157. The risk of infection from Salmonella spp. and E. Coli O157 greatly exceeded risks previously reported for recreational waters and SUDS. Pathogen concentrations were associated with precipitation and the concentration of suspended solids in the runoff. Given our findings, it is recommended that SUDS in developing countries should consider potential higher pathogen concentrations in stormwater runoff to reduce exposure.
Article Highlights
Pathogen concentrations in stormwater runoff may be higher in developing countries.
Pathogen concentrations in the SUDS micropool are linked to TSS concentrations.
High precipitation events result in high pathogen concentrations in the SUDS.
High pathogen concentrations in the micropool yield a high infection risk in children.
Understanding pathogen concentrations in stormwater runoff is key to operating SUDS.
Similar content being viewed by others
1 Introduction
Increasing areas of impermeable surfaces due to urbanization impacts stormwater runoff in urban settings, resulting in frequent flooding and decreased quality of stormwater runoff water (Perales-Momparler et al. 2017; Ariza et al. 2019; Bohman et al. 2020; Ishaq et al. 2022). Sustainable Urban Drainage Systems (SUDS) are used as an innovative urban water management strategy to drain stormwater runoff allowing follow-up harvest and reuse (Gülbaz and Kazezyılmaz-Alhan 2018; x et al. 2018; Kourtis et al. 2020; Chen et al. 2021; Kourtis et al. 2021). Given the wide variety of SUDS, these systems are designed and operated depending on the context and the objectives in each case (Dierkes et al. 2015; Jodra-Lopez et al. 2018). Increasing utilization of SUDS has led to the development of many projects in urban communities, resulting in increased human exposure to these systems (Ishaq et al. 2022). However, exposure varies, depending on the type of structure, being more important in systems where stormwater runoff is transported above the ground or stored at the surface.
It has been well established that stormwater runoff contains pathogens (Ahmed et al. 2019) and that these pathogens affect water quality at SUDS (Ishaq et al. 2022), resulting in potential health risks. Pathogens are known to attach to soil particles in stormwater runoff (Soupir et al. 2010), and therefore, pathogen concentrations may be associated with those of suspended solids. There is little information regarding other aspects that may influence the presence of pathogens in SUDS, although it has been reported that seasonality, temperature, and geographic location also have an important effect on pathogen concentrations in stormwater runoff (Ishaq et al. 2022).
The health risks associated with exposure to urban stormwater runoff contaminated with Noroviruses, Enteroviruses, Campylobacter, Salmonella, E. Coli, Giardia, and Cryptosporidium have been previously established in high-income countries, including the USA, Switzerland, Holland, New Zealand, and the United Kingdom (Yeah-King 2009; McBride et al. 2013; Sales-Ortells and Medema 2014, 2015; Schoen et al. 2017; Van Abel et al. 2017; Abia et al. 2018; Allafchi et al. 2019; Taguchi et al. 2020). Despite the fact that there is an increasing number of publications regarding pathogen concentrations in stormwater runoff (Sidhu et al. 2013; Ahmed et al. 2018, 2019; McGinnis et al. 2018; Steele et al. 2018), there is very little information on this issue in low-income countries, most of them located in tropical zones. Tropical zones often show high precipitation and temperatures (Gilarranz et al. 2022; Lembrechts et al. 2022) thus favoring pathogen presence. To our knowledge, there are only a handful of studies that measure pathogen concentrations in stormwater in low-income countries in tropical regions (Vieira et al. 2002; Cardonha et al. 2004; Soupir et al. 2010; Katukiza et al. 2014), and most of the studies report concentrations that are higher than those reported in high-income countries.
Poorly managed combined sewer overflow (CSO) structures often result in surface water contamination (McGinnis et al. 2018). Lack of resources in developing countries can lead to less frequent maintenance of sewer pipes and overflow structures, increasing the chances of surface water and stormwater runoff contamination with CSO during rain events. The latter may lead to higher pathogen concentrations in stormwater runoff, which could lead to higher infection risks at SUDS structures.
A previous study conducted at the San Cristobal Park’s SUDS (Bogotá-Colombia) found high E. Coli concentrations in stormwater runoff ranging between 3,000 and 50,000 MPN/100 mL at the SUDS treatment train inlet (Ramirez Cely 2018; Giraldo et al. 2019). It was also observed that park users (especially children) were interacting with a micropool water in a SUDS train that treats this pathogen-rich stormwater runoff, resulting in potential exposure to the pathogens. Given the scarce information regarding pathogen concentrations in stormwater runoff in low-income countries in the tropics, we wanted to investigate if these concentrations are, in fact, higher than those reported in high-income countries and determine the risks of infection linked to exposure to these pathogens at SUDS structures in these settings, using as a case study the micropool in the SUDS at the San Cristobal Park. Also, we wanted to identify external variables that are associated with the presence of pathogens in these systems and make recommendations regarding the operation of extended dry detention basins in low-income countries. To do so, we measured the concentrations of two pathogens commonly associated with enteric symptoms in children: Salmonella spp. and E. Coli O157. These two pathogens are frequently reported in studies that evaluate risks resulting from exposure to pathogens in runoff water (Harmel et al. 2016; Sharma et al. 2020). We investigated several physicochemical water quality and meteorological parameters and evaluated possible associations between these variables and pathogen concentrations in stormwater runoff in the micropool. Observation diaries were used to understand exposure to SUDS water. We estimated the risk of infection in children who play with stormwater stored in the micropool through Quantitative Microbial Risk Assessment (QMRA). We evaluated three exposure routes, including hand-to-mouth, water droplet, and total ingestion (hand-to-mouth and water droplet ingestion added).
2 Materials and Methods
2.1 Study Site
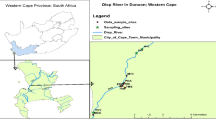
There is a SUDS treatment train located at San Cristobal’s Metropolitan Park in Bogotá, Colombia. This drainage system treatment train consists of a grassed swale and an extended dry detention basin. Grassed swales are open and shallow channels with circular, trapezoidal, or parabolic shapes, whose vegetation cover withstands the erosion caused by runoff flow and reduces runoff speed; they are useful to maximize hydraulic residence time (Xie et al. 2017; Mohammed and Weljker 2020). Extended dry detention basins are designed to temporarily store runoff water and release it slowly through overland flow by means of a hydraulic structure (Perales-Momparler et al. 2017; Xie et al. 2017). The San Cristobal Park train treatment is composed of a 70 m long and 4 m wide grassed swale that manages runoff water and leads the flow to an extended dry basin with a volume of 195 m3 and a depth of 0.25 m, where runoff water is temporarily stored while the water infiltrates into the soil (Ramirez Cely 2018). The system ends in a 3 m diameter micropool, that retains water prior to discharge, to regulate flow, maintaining a water depth of 15 cm (Ramirez Cely 2018; Giraldo et al. 2019). A benefit out of having a micropool is the enhanced control of resuspended solids (Guo et al. 2012). The drainage areas of this SUDS treatment train are composed of vehicular roads at the outer east part of the park, paved playground basketball courts, large landscape areas, and pedestrian paths (Giraldo et al. 2019; Ramirez Cely 2018) (Fig. 1).
2.2 Physicochemical and Meteorological Parameters
Water samples from the micropool were collected once a week for 25 weeks (wet and dry seasons), starting in September 2019. For microbial analyses, 100 mL samples were collected from the micropool and preserved according to EPA-600 guide for surface water sampling (Wright et al. 1987). Simultaneously, 1-L samples were collected for physicochemical analyses, which included Biological Oxygen Demand (BOD), Total Kjeldahl Nitrogen (TKN), Phosphates (PO4 − 3), and Total Suspended Solids (TSS). Samples for TKN and BOD were preserved at low pH (< 2), and samples for PO4 − 3 and TSS analyses were preserved at 4 oC. All physicochemical analyses were conducted by the local water utility laboratory (Empresa de Acueducto y Alcantarillado de Bogotá), which is certified by the Hydrology and Meteorology National Institute (IDEAM for its acronym in Spanish) and the Colombian accreditation institute (ONAC for its acronym in Spanish). This laboratory uses the Standard Methods for the Examination of Water and Wastewater. TSS was estimated by gravimetry and drying at 103 to 105 °C. BOD was estimated by luminescence after 5-day incubation and measurement of dissolved oxygen concentration. Both TKN and PO4 − 3 were analyzed through spectrophotometry.
We obtained precipitation and solar radiation data from a weather station, which is part of the Air Quality Monitoring System of the city (operated by the local environmental agency, Secretaría Distrital de Ambiente in Spanish), located at San Cristobal Park. At this station, precipitation is measured every minute, and radiation every hour. Additionally, a tipping bucket rain gauge (Davis model 7585 M) that measures precipitation at 0.2 mm accuracy was placed at a kindergarten inside the park to validate the information provided by the Air Quality Monitoring System weather station (Fig. 1).
2.3 Determination of Microorganism Concentrations
2.3.1 Determination of Sampling Location
Prior to the 25-week sampling period, five different points (four radial sections and the center of the micropool (Fig. 1)) were selected as sampling points; three water samples per point were collected twice a week for three weeks, and microorganism concentrations were measured in those samples. After obtaining plate counts for all samples, we conducted a Shapiro-Wilk test to investigate if the data fitted the normal distribution. To test whether the average concentrations from the different sampling points were statistically different, we conducted an ANOVA statistical test. As the ANOVA test did not show a significant statistical differences (Gaber et al. 2020), we determined that pathogen concentrations in the middle of the micropool adequately represented microorganism concentrations throughout the system, Thus, the middle point was selected as the sampling point for further analyses.
2.3.2 Pathogen Quantification
E. Coli O157 and Salmonella spp. containing the invA gene (Henao-Herreño et al. 2017) were used as positive controls. Salmonella spp. containing the invA gene was selected as a pathogen of interest, as this gene is associated with infection in mammals (Skovgaard 2004).
Salmonella spp. was quantified by quantitative polymerase chain reaction (qPCR), by quantifying the invA gene in DNA extracts from the contaminated water, as previously used in risk analysis (Henao-Herreño et al. 2017). A 25 mL water sample was filtered using a 0.45 μm filter and the pellet remaining on the filter was used for DNA isolation using the MoBio PowerSoil DNA Isolation Kit (Henao-Herreño et al. 2017). Real-time qPCR of the extracted sample was carried out using Brilliant II SYBR Green Master Mix, as previously described (Nam et al. 2005; Henao-Herreño et al. 2017). A no-template control and a negative control containing deoxyribonucleic acid (DNA) from E. Coli O157 were used for quality control. The calibration curve for qPCR was constructed using DNA from the positive control of Salmonella spp. (Henao-Herreño et al. 2017) and doing plate counts on selective Salmonella-Shigella agar (SS Agar) (Busse 1995).
E. Coli O157 concentrations were determined by plate counts using chromogenic O157 agar (Chromatic, Liofilchem); serial dilutions (10− 2 and 10− 3) were plated on chromogenic O157 agar plates and incubated at 35 °C for 18 h under aerobic conditions (Akata et al. 2015). To evaluate the specificity of the chromogenic method, we conducted polymerase chain reaction (PCR) assays of selected individual colonies with the primers: stx1_F; stx2_F, stx2_R, eae_F, and eae_R as previously described (Koev et al. 2019).
2.4 Statistical Analysis
2.4.1 Common Trends Among Pathogen Concentrations
The Pearson correlation coefficient (r) was estimated between the concentrations of the two pathogens to evaluate if there were behavioral similarities among the concentrations of both pathogens, specifically, to test if there were any correlations between their concentrations (Dagnino 2014).
2.4.2 Correlation of Physicochemical and Meteorological Variables and Microorganism Concentrations
To determine if there is an association between the concentrations of pathogen concentration and any of the physicochemical and/or meteorological variables, two multivariate regression models were used. With weekly data over a period of 25 weeks, each multivariate model had as the response variable the concentration of either E. Coli O157 or Salmonella spp., and as independent variables weekly values of physicochemical parameters and meteorological variables (i.e., precipitation, solar radiation, TSS, BOD, TKN, and phosphate). Normality of the response variable was verified with a Q-Q Plot, multicollinearity was evaluated with a Variance Inflation Factor (VIF), homoscedasticity was verified with the White test, and the normality of the statistical errors were determined with the Shapiro-Wilk test (Sauerbrei et al. 2006; Liu et al. 2019; Jenkins and Quintana-Ascencio 2020). Statistical analysis was conducted using STATA version 14 (StataCorp, 4905 Lakeway Dr College Station, TX 77,845).
2.5 Quantitative Microbial Risk Assessment (QMRA)
2.5.1 Exposure Assessment
Children represent 45% of park users and most children that visit the SUDS at San Cristobal Park are students at Manuelita Saenz School, which is located close to the park. Other children that frequently visit the park are members of several sports teams that train there (Grisales Espinosa and Toro Medina 2014). As a result, most park users interacting with contaminated water at the micropool are children. To identify the exposure routes of children to runoff water stored in the micropool, we used different approaches. First, we filled daily activity diaries during the 25-week period; here, we registered each observed exposure of children to micropool water. We also registered other aspects, including the time that the child was inside the micropool, type of interaction (just feet, feet and hands, and washed face), and other characteristics that could be relevant for each event. We also conducted surveys with parents and teachers to determine the frequency of children visits to the micropool. This information was used to determine the type of exposure and exposure frequency, which are needed to estimate the daily exposure doses. Hand-to-mouth, water droplet, and total ingestion (addition of the two previous exposure routes) were selected as exposure routes.
We calculated the ingested water volume that resulted from hand-to-mouth exposure, \({Q}_{HM }\ ({\text{m}\text{m}}^{3}/\text{m}\text{i}\text{n}\ \text{o}\text{r}\ {\upmu }\text{L}/\text{m}\text{i}\text{n}),\) by multiplying the exposed hand area, A (mm2), by the thickness of water film remaining on hands, h (mm), and fHM (n/min) which is the frequency of hand-to-mouth contact (Man et al. 2014), as follows:
To calculate the volume of ingested water droplets, \({Q}_{D}\ ({\upmu }\text{L}/\text{m}\text{i}\text{n}),\) we multiplied the volume of water droplets, \({V}_{D}\ \left({\upmu }\text{L}\right)\), by the frequency of splashing fD (n/min) (Man et al. 2014), as follows:
Finally, we calculated the total ingested volume, by adding the volume of water ingested through hand-to-mouth exposure and from water droplet splashing (Man et al. 2014), as follows:
To estimate the exposure time (min), we established the distribution that most closely matched the observations reported on the activity diaries from the observations that indicated how much time children played at the micropool during each interaction. A better description of the parameters used for the analysis is shown in Table 1.
2.5.2 Dose-response
The exposure dose, d, was calculated by multiplying the concentration of each microorganism in micropool water (UFC/µL) by the ingestion volume calculated for each exposure route (µL/min) and the exposure time (min). Estimation of pathogen concentrations was previously described (Sect. 2.2); to estimate the risk of infection, pathogen concentrations were modeled as log-normal distributions using as input data the mean and standard deviation obtained from sample analysis.
The risk of infection with Salmonella spp. was estimated using the Beta-Poisson dose-response model \({P}_{inf}=1-{\left(1+\frac{d}{\beta }\right)}^{-\alpha }\) (Skovgaard 2004). The risk of infection with E. Coli was also estimated using a Beta-Poisson dose-response model that was specifically developed for children (Teunis et al. 2004). The model parameters used for the dose-response models for each pathogen are shown in Table 2.
2.5.3 Risk Characterization
Monte Carlo simulations using Crystal Ball (Oracle Crystal Ball, Redwood Shores, CA, USA) (Jaidi et al. 2009; USEPA 2009) with five thousand testing simulations were used to estimate the daily probability of infection. We did not estimate annual infection risks, because exposures were sporadic. As reported in activity diaries, most children interacted just once with the structure during the 25-week period.
3 Results and Discussion
3.1 Meteorological and Physicochemical Variables
Accumulated precipitation was measured for 25 weeks and fluctuated between 0 and 62 mm with a standard deviation of 19.29 mm, and average precipitation of 28.76 mm. The accumulated weekly radiation ranged between 24,873 and 53,344 W/m2, with a standard deviation of 9012.78 W/m2 and average radiation of 36520.24 W/m2, being highly variable during the sampling period (Fig. 2). It is also important to mention that the average temperature during the sampling period was 13.5 °C with a standard deviation of 1.03 °C.
TKN concentrations ranged between 1 and 9 mg-N/L with a standard deviation of 1.85 mg-N/L and an average concentration of 3.64 mg-N/L; phosphate concentrations ranged between 0.07 and 2.2 mg-PO4/L with a standard deviation of 0.44 mg-PO4/L and an average concentration of 0.49 mg-PO4/L; TSS concentrations ranged between 77 and 333 mg/L with a standard deviation of 74.59 mg/L and an average concentration of 210.2 mg/L; and BOD5 concentrations ranged between 3 and 21 mg-O2/L with a standard deviation of 4.12 mg-O2/L and an average concentration of 10.2 mg-O2/L (Fig. 2). A recent study that evaluated stormwater quality at the inlet of this SUDS treatment train found that nitrogen and organic matter likely entered the train through irregular wastewater discharges, while total phosphorus, mainly in the particulate form, was associated with total suspended solids from diffuse sources, such as soil erosion in the catchment and solid transport in this urban drainage (Silva et al. 2022). Increased nutrient and organic matter concentrations improve the conditions for pathogen growth in the micropool.
3.2 Microorganism Concentrations
E Coli O157 concentrations ranged between 0 and 38,000 (± 2259) CFU/mL and Salmonella spp. concentrations ranged between 0 and 3079 (± 140) copy #/mL during the 25 weeks of sampling (Fig. 2). E. Coli O157 concentrations in this study were, in general, much higher than those reported in recreational waters in Brazil, e.g., Rodrigues et al. (2016), as well as concentrations reported for E. coli in stormwater in developed countries, including Australia, UK, and United States (Lundy et al. 2018). Salmonella spp. concentrations were, on average, much higher than those reported for stormwater in the United States (Ahmed et al. 2019). The high pathogen concentrations in runoff water could be a result of contamination from malfunctioning CSO structure (Silva et al. 2022). These structures collect water from rain and sewage combined, and after heavy rains, wastewater mixed with stormwater runoff is discharged into the environment. It has been well documented that combined sewer overflow structures can be a source of environmental pathogens in developing countries (Huang et al. 2017). This could also partly explain why pathogen concentrations were much higher during weeks with high precipitation (4–7, 9–14, 18–22). However, other experiments, including the use of tracers and biomarkers, are needed to further explore this hypothesis.
To evaluate if the concentrations of the two pathogens were correlated, the Pearson correlation coefficient (r) was determined (Dagnino 2014). The concentrations of E. Coli O157 and Salmonella spp. were statistically similar, with an r of 0.34 and a p-value of 0.048. Figure 2 shows that during weeks 4–8, 10–14, and 18–22, both E. Coli O157 and Salmonella spp. concentrations increased.
3.3 Comparative Evaluation of Meteorological Variables, Physicochemical Parameters, and Microorganism Concentrations
During rainy periods, microbial concentrations increased, suggesting that the concentrations of pathogens are affected by precipitation (Fig. 2); this can be observed in the periods between weeks 4 and 7, week 10 and week 14, and weeks 18 and 22. This phenomenon could be the result of the resuspension of pathogens and the first flush phenomenon, which refers to the existence of high loads of pollutants in runoff during the first minutes of precipitation events (Mamun et al. 2020). Solar radiation appeared to create the opposite effect and reduce pathogen concentrations; it can be observed during weeks 8, 15, and 16 when radiation increased that microbial concentrations decreased.
Multivariate regression models were used to analyze potential associations between pathogen concentrations in the micropool and the physicochemical and meteorological parameters measured (Table 3). The results indicate that E. Coli O157 concentrations were significantly associated with precipitation (p = 0.007) and TSS (p = 0.023) (Table 3). A significant association was also observed between Salmonella ssp.. Concentrations and both precipitation (p = 0.002) and TSS (p = 0.025), and a marginal association was observed with PO4 − 3 (p = 0.064) (Table 3).
It has been previously observed that intervals of high precipitation may lead to an increase in microbial populations, resulting from the decreased amount of solar radiation (Williamson et al. 2017). Even though solar radiation was not correlated with pathogen concentrations in this study, it is widely known that UV radiation is a common disinfection method for both E. Coli O157 and Salmonella ssp..
Total suspended solids concentrations measured in the micropool are typical for runoff water. TSS concentration is one of the most common parameters used for evaluating runoff stormwater quality (Goyal et al. 1977; Yazdi et al. 2019), and it is known that microorganisms represent an important fraction of TSS. This could partially explain why pathogen concentrations were significantly associated with TSS concentrations in this study.
3.4 Quantitative Microbial Risk Assessment (QMRA)
It has been previously documented that QMRA can facilitate risk management efforts as well as decision-making in terms of stormwater management (Ahmed et al. 2019; Ishaq et al. 2020, 2022). Here, QMRA was conducted to estimate the risk of infection resulting from exposure to runoff water stored in the micropool. Salmonella spp. concentration was modeled as a log-normal distribution with \(\stackrel{-}{X}\) = 1562.615 copy #/mL and σ2 = 1296.598 copy #/mL and E. Coli O157 concentration was modelled as a log-normal distribution with \(\stackrel{-}{X}\) = 9159.75 CFU/mL and σ2 = 9704.009 CFU/mL. Distribution parameters were determined from previous results (Sect. 3.2). Non-detects were substituted as one-half of the reporting limit (USEPA 2002).
Here we estimated the risk of infection with Salmonella spp. and E. Coli O157 from exposure to micropool stormwater runoff, as the two microorganisms were found at high concentrations in micropool water; exposure routes were selected to evaluate indirect ingestion (Sales-Ortells and Medema 2015). From observations, we established that only one-third of the children were exposed to hand-to-mouth and water droplets simultaneously, with an average exposure time of 6.33 min. This exposure time is short but was the result of exposure times reported in the activities diaries during the 25-week period.
During the 25-week observation period, 78 children had direct contact with water in the micropool. The estimated mean ingested volume per minute resulting from hand-to-mouth exposure was 56.57 µL/min (95% CI-range 5.14-183.24 µL/min); ingestion of water droplets was estimated as 624.15 µL/min (95% CI-range 19.86-2196.35 µL/min) and total ingestion was 680.724 µL/min (95% CI-range 61.774-2152.334 µL/min).
Based on these ingestion volumes, the estimated daily risk of infection (per event) with E. Coli 0157 was 0.574 (95% CI-range 0.437–0.629) for total ingestion, 0.571 (95% CI-range 0.402–0.626) for ingestion of water droplets, and 0.476 (95% CI-range 0.294–0.546) for hand-to-mouth ingestion, meaning that on average 57% of the children who interacted with the micropool could have been infected with this pathogen.
E. Coli O157 showed the highest risk of infection, being total ingestion and water droplet ingestion the routes with the highest risk. These observed high risks are consistent with the high E. Coli O157 concentrations reported in this study, which are much higher than those previously reported in stormwater (Lundy et al. 2018). The estimated daily risk of infection (per event) with Salmonella ssp. was slightly lower than that predicted for E. Coli O157, but was still high, with 0.481 (95% CI-range 0.213–0.579) for total ingestion, 0.475 (95% CI-range 0.158–0.584) for ingestion of water droplets, and 0.283 (95% CI-range 0.047–0.416) for hand-to-mouth ingestion of Salmonella spp. As expected, hand-to-mouth exposure resulted in lower infection risk as compared to ingestion of water droplets (Man et al. 2014).
The disease burden obtained in this study using the methodology previously described (Ishaq et al. 2020) was high for E. coli (10.9 DALYs/1000 persons (CI 95% 8.3–11.9)) and for Salmonella (2.86 DALYs/1000 persons (CI 95% 1.2–3.4)), higher than that observed in SUDS for children playing at a water plaza (Ishaq et al. 2020), and exceeding the guidelines set by the USEPA for recreational use of 5.75 DALYs/1000 bathers.
The daily risks of infection from exposure to Salmonella spp. and E. Coli O157 were very high, as compared to the risks reported from other sources (McBride et al. 2013; Van Abel et al. 2017; Allafchi et al. 2019; Taguchi et al. 2020). It is essential to mitigate this impact by restricting the interaction of children with contaminated runoff stormwater.
Our findings differ from the risks predicted by other stormwater runoff studies. Differences could be explained by the fact that previous QMRA studies investigate different variables from those considered in this study, including different pathogens and different routes of exposure (Yeah-King 2009; McBride et al. 2013). These differences could also be partially explained by the fact that these studies were conducted in developed countries (Ishaq et al. 2020), and so far, we have found differences regarding pathogen concentrations in runoff water between developed and developing countries (up to 2–3 orders of magnitude), likely resulting from inadequate sewer system maintenance.
Although infection does not necessarily result in illness, the reported daily infection risks are high, highlighting the dangers for children who play with the structure in the park. One of the limitations of this study is that due to privacy issues, we did not follow up with the families of the children that interacted with the pool and are not aware of whether any of the interactions resulted in illness. Also, in this study we did not estimate the annual risk of infection, as exposure to the contaminated micropool was sporadic (most children only interacted once with micropool water during the 25-week sampling period); some studies use the daily risk of infection when the risk can be overestimated in annual terms (Park et al. 2004; Oh et al. 2018). Other limitations of this study include the fact that we used some exposure data from the literature to estimate risk; it is unknown if these exposure values adequately represent the exposure observed at the micropool.
Results from our study indicate that even though SUDS are a suitable and adequate solution to manage stormwater, the concentrations of pathogens in runoff water, especially in developing countries, may be higher than in developed countries. Pathogens in water from SUDS pose a risk for potential recreational users of the SUDS, especially children while playing. This fact should be considered during the SUDS design process and its operation.
Specifically, in the case of the San Cristobal SUDS, we recommended evaluating alternative management strategies to minimize exposure of children to contaminated water as pathogen concentrations in water are very high resulting in high infection risks. Alternative management strategies included frequent cleaning and maintenance of the micropool. In fact, prior to starting this study the micropool was cleaned; right after cleaning, microorganism concentrations were below the detection limit (first three weeks). Based on this observation we concluded that cleaning the micropool periodically could help reduce the risk of infection. It was also recommended to evaluate the possibility of control access to micropool water by using vertical barriers (i.e., providing upland, riparian, or wetland plant massing, see (Echols and Pennypacker 2008)) and using informative advertising by the micropool area to help reduce children exposure.
4 Conclusion
E. Coli and Salmonella spp concentrations measured in this research were much higher than those reported for stormwater in developed countries. These concentrations were proved to be associated with precipitation and TSS concentrations in stormwater runoff. Those high concentrations lead to a disease burden and daily risks of infection that is higher than that observed in other SUDS. Our results highlight the importance of a better stormwater quality understanding prior to SUDS implementation and the need for this kind of studies in developing countries, as SUDS have become more interesting alternatives for flooding control. There are still many unknowns with respect to SUDS proper operation and management in low-income settings. In the specific case of the San Cristobal SUDS, after assessing the risk, we were able to make recommendations with respect to its management and operation to reduce children’s exposure to contaminated water.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Abia ALK, Ubomba-Jaswa E, Schmidt C, Dippenaar MA (2018) Where did they come from - multi-drug resistant pathogenic Escherichia coli in a cemetery. environment? Antibiot 7(3):73. https://doi.org/10.3390/antibiotics7030073
Ahmed W, Hamilton K, Toze S, Cook S, Declan P (2019) A review on microbial contaminants in stormwater runoff and outfalls: potential health risks and mitigation strategies. Sci Total Environ 692:1304–1321. https://doi.org/10.1016/j.scitotenv.2019.07.055
Ahmed W, Zhang Q, Lobos A, Senkbeil J, Sadowsky MJ, Harwood VJ, Saeidi N, Marinoni O, Ishii S (2018) Precipitation influences pathogenic bacteria and antibiotic resistance gene abundance in storm drain outfalls in coastal sub-tropical waters. Environ Int 116:308–318. https://doi.org/10.1016/j.envint.2018.04.005
Akata I, Torlak E, Erci F (2015) Efficacy of gaseous ozone for reducing microflora and foodborne pathogens on button mushroom. Postharvest Biol Technol 109:40–44. https://doi.org/10.1016/j.postharvbio.2015.06.008
Allafchi F, Valeo C, He J, Neumann NF (2019) An integrated hydrological-CFD model for estimating bacterial levels in stormwater ponds. Water (Switzerland) 11(15):1016. https://doi.org/10.3390/w11051016
Ariza SLJ, Martínez JA, Muñoz AF, Quijano JP, Rodríguez JP, Camacho LA, Díaz-Granados M (2019) A multicriteria planning framework to locate and select sustainable urban drainage systems (SUDS) in consolidated urban areas. Sustainability 11(8):2312. https://doi.org/10.3390/su11082312
Bohman A, Glaas E, Karlson M (2020) Integrating sustainable stormwater management in urban planning: Ways forward towards institutional change and collaborative action. Water (Switzerland) 12(1):203. https://doi.org/10.3390/w12010203
Busse M (1995) Chap. 11 Media for salmonella. 187–201. Editor(s): Janet E.L. Corry, G.D.W. Curtis, Rosamund M. Baird, Progress in Industrial Microbiology, Volume 34. ISSN 0079-6352, ISBN 9780444814982, https://doi.org/10.1016/s0079-6352(05)80013-x
Cardonha AMS, Cardonha AMS, Vieira RHS, Rodrigues DP, Macrae A, Peirano G, Teophilo GND (2004) Fecal pollution in water from storm sewers and adjacent seashores in Natal, Rio Grande do Norte, Brazil. Int Microbiol 7(3):213–218
Chen SS, Tsang DCW, He M, Sun Y, Lau LSY, Leung RWM, Lau ESC, Hou D, Liu A, Mohanty S (2021) Designing sustainable drainage systems in subtropical cities: Challenges and opportunities. J Clean Prod 280:124418. https://doi.org/10.1016/j.jclepro.2020.124418
Dagnino J (2014) Correlacion. Rev Chil Anest 150–153
Dierkes C, Lucke T, Helmreich B (2015) General technical approvals for decentralised sustainable urban drainage systems (SUDS)-the current situation in Germany. Sustain 7:3031–3051. https://doi.org/10.3390/su7033031
Echols S, Pennypacker E (2008) From stormwater management to artful rainwater design. Landsc J 27:268–290
Gaber GA, Mohamed LZ, Tash MM (2020) Experimental correlation using ANOVA and DOE studies on corrosion behavior of Fe and Ni-based alloy under different media. Mater Res Express 7(3):036521. https://doi.org/10.1088/2053-1591/ab7e6d
Gilarranz M, Pérez IA, García M, Sánchez ML, Pardo N, Fernández-Duque B (2022) Analysis of spatial distribution and evolution of terrestrial precipitation. J Atmos Solar-Terrestrial Phys 237:105930. https://doi.org/10.1016/j.jastp.2022.105930
Giraldo MM, Jiménez SL, Alejandro J, Ramírez PM, Rodríguez JP, Camacho LA, Díaz-granados M (2019) Quantifying the performance of a SUDS treatment train. A case-study of San Cristobal Park, Bogotá. NOVATECH Conference, 1–5 July, Lyon (France)
Goyal SM, Gerba CP, Melnick JL (1977) Occurrence and distribution of bacterial indicators and pathogens in canal communities along the Texas coast. Appl Environ Microbiol 34:139–149. https://doi.org/10.1128/aem.34.2.139-149.1977
Grisales Espinosa V, Toro Medina AM (2014) Metodología para la integración social en proyectos de sistemas de drenaje urbano sostenible: caso de estudio San Cristobal sur, Bogotá. Undergraduate Thesis. Universidad de los Andes
Gülbaz S, Kazezyılmaz-Alhan CM (2018) Impact of LID implementation on Water Quality in Alibeyköy Watershed in Istanbul, Turkey. Environ Process 5(1):201–212. https://doi.org/10.1007/s40710-018-0318-3
Guo JC, Shih H, MacKenzie M (2012) Storm-water Quality Control Basin with Micropool. J Irrigartion Drain Eng 138(5). https://doi.org/10.1061/(ASCE)IR.1943-4774.0000415
Harmel RD, Hathaway JM, Wagner KL, Wolfe JE, Karthikeyan R, Francesconi W, McCarthy DT (2016) Uncertainty in monitoring E. coli concentrations in streams and stormwater runoff. J Hydrol 534:524–533. https://doi.org/10.1016/j.jhydrol.2016.01.040
Henao-Herreño LX, López-Tamayo AM, Ramos-Bonilla JP, Haas CN, Husserl J (2017) Risk of illness with salmonella due to consumption of raw unwashed vegetables irrigated with water from the Bogotá River. Risk Anal 37:733–743. https://doi.org/10.1111/risa.12656
Huang C, Hu Y, Wang L, Wang Y, Li N, Guo Y, Feng Y, Xiao L (2017) Environmental transport of emerging human-pathogenic Cryptosporidium species and subtypes through combined sewer overflow and wastewater. Appl Environ Microbiol 83(16):e00682–e00617. doi: https://doi.org/10.1128/AEM.00682-17
Ishaq S, Sadiq R, Chhipi-Shrestha G, Farooq S, Hewage K (2022) Developing an integrated “Regression-QMRA method” to predict public health risks of low impact developments (LIDs) for improved planning. Environ Manage 70:633–649. https://doi.org/10.1007/s00267-022-01657-0
Ishaq S, Sadiq R, Farooq S, Chhipi-Shrestha G, Hewage K (2020) Investigating the public health risks of low impact developments at residential, neighbourhood, and municipal levels. Sci Total Environ 744(20):140778. https://doi.org/10.1016/j.scitotenv.2020.140778
Jaidi K, Barbeau B, Carrière A, Desjardins R, Prévost M (2009) Including operational data in QMRA model: development and impact of model inputs. J Water Health 7:77–95. https://doi.org/10.2166/wh.2009.133
Jenkins DG, Quintana-Ascencio PF (2020) A solution to minimum sample size for regressions. PLoS ONE 15:1–15. https://doi.org/10.1371/journal.pone.0229345
Jodra-Lopez R, Sordo-Ward A, Gabriel-Martin I, Garrote L (2018) Effects of key properties of rainfall series on hydrologic design of Sustainable Urban Drainage Systems (SUDS). Proceedings 7:17. https://doi.org/10.3390/ecws-3-05827
Katukiza AY, Ronteltap M, Van Der Steen P, Foppen JWA, Lens PNL (2014) Quantification of microbial risks to human health caused by waterborne viruses and bacteria in an urban slum. J Appl Microbiol 116(2):447–463. https://doi.org/10.1111/jam.12368
Koev K, Zhelev G, Marutsov P, Gospodinova K, Petrov V, Stoyanchev T (2019) Molecular screening and characterization of shiga toxin-producing Escherichia coli by multiplex PCR assays for stx 1, stx 2, eaea, h7 in raw milk. Kafkas Univ Vet Fak Derg 25:271–275. https://doi.org/10.9775/kvfd.2018.20373
Kourtis IM, Tsihrintzis VA, Baltas E (2018) Simulation of Low Impact Development (LID) Practices and Comparison with Conventional Drainage Solutions. Proceedings 2, 640. https://doi.org/10.3390/proceedings2110640
Kourtis IM, Tsihrintzis VA, Baltas E (2020) A robust approach ror comparing conventional and sustainable flood mitigation measures in urban basins. J Environ Manage 269:110822. https://doi.org/10.1016/j.jenvman.2020.110822
Kourtis IM, Bellos V, Kopsiaftis G, Psiloglou B, Tsihrintzis VA (2021) Methodology for holistic assessment of grey-green flood mitigation measures for climate change adaptation in urban basins. Journal of Hydrology 603(Part A):126885. https://doi.org/10.1016/j.jhydrol.2021.126885.
Lembrechts JJ, Van den Hoogen J, Aalto J, Ashcroft MB, De Frenne P, Kemppinen J, Kopecký M, Luoto M, Maclean IMD, Crowther TW (2022) Global maps of soil temperature. Glob Chang Biol 28(9):3110–3144. https://doi.org/10.1111/gcb.16060
Liu S, Lu M, Li H, Zuo Y (2019) Prediction of gene expression patterns with generalized linear regression model. Front Genet 10:1–11. https://doi.org/10.3389/fgene.2019.00120
Lundy L, Revitt M, Ellis B (2018) An impact assessment for urban stormwater use. Environ Sci Pollut Res 25:19259–19270. https://doi.org/10.1007/s11356-017-0547-4
Mamun A, Al, Shams S, Nuruzzaman M (2020) Review on uncertainty of the first-flush phenomenon in diffuse pollution control. Appl Water Sci 10:1–10. https://doi.org/10.1007/s13201-019-1127-1
Man H, De, Bouwknegt M, Heijnsbergen E, Van, Leenen EJTM, Van Knapen F, De Roda Husman AM (2014) Health risk assessment for splash parks that use rainwater as source water. Water Res 54:254–261. https://doi.org/10.1016/j.watres.2014.02.010
McBride GB, Stott R, Miller W, Bambic D, Wuertz S (2013) Discharge-based QMRA for estimation of public health risks from exposure to stormwater-borne pathogens in recreational waters in the United States. Water Res 47:5282–5297. https://doi.org/10.1016/j.watres.2013.06.001
McGinnis S, Spencer S, Firnstahl A, Stokdyk J, Borchardt M, McCarthy DT, Murphy HM (2018) Human bacteroides and total coliforms as indicators of recent combined sewer overflows and rain events in urban creeks. Sci Total Environ 630:967–976. https://doi.org/10.1016/j.scitotenv.2018.02.108
Mohammed M, Weljker A (2020) Impact of soil compaction on vegetated basin transition. Geo-Congress 2020 GSP 320:612–621. February 25–28, Minneapolis, Minesota. https://doi.org/10.1061/9780784482803.028
Nam HM, Srinivasan V, Gillespie BE, Murinda SE, Oliver SP (2005) Application of SYBR green real-time PCR assay for specific detection of Salmonella spp. in dairy farm environmental samples. Int J Food Microbiol 102:161–171. https://doi.org/10.1016/j.ijfoodmicro.2004.12.020
Oh J, Makar M, Fusco C, McCaffrey R, Rao K, Ryan EE, Washer L (2018) A generalizable, data-driven approach to predict daily risk of Clostridium difficile infection at two large academic health centers. Infect Control Hosp Epidemiol 39:425–433. https://doi.org/10.1017/ice.2018.16
Park P, Garton HJL, Kocan MJ, Thompson BG (2004) Risk of infection with prolonged ventricular catheterization. Neurosurgery 55:594–599. https://doi.org/10.1227/01.NEU.0000134289.04500.EE
Perales-Momparler S, Andrés-Doménech I, Hernández-Crespo C, Vallés-Morán F, Martín M, Escuder-Bueno I, Andreu J (2017) The role of monitoring sustainable drainage systems for promoting transition towards regenerative urban built environments: a case study in the valencian region, Spain. J Clean Prod 163:S113–S124. https://doi.org/10.1016/j.jclepro.2016.05.153
Ramirez Cely P (2018) Evaluación del desempeño de Sistemas Urbanos de Drenaje Sostenible: Caso de estudio Parque Metropolitano san Cristobal Sur. MSc Thesis. Universidad de los Andess
Rodrigues VFV, Rivera ING, Lim K-Y, Jiang SC (2016) Detection and risk assessment of diarrheagenic E. coli in recreational beaches of Brazil. Mar Pollut Bull 109:163–170. https://doi.org/10.1016/j.marpolbul.2016.06.007
Sales-Ortells H, Medema G (2014) Screening-level microbial risk assessment of urban water locations: a tool for prioritization. Environ Sci Technol 48:9780–9789. https://doi.org/10.1021/es5020407
Sales-Ortells H, Medema G (2015) Microbial health risks associated with exposure to stormwater in a water plaza. Water Res 74:34–46. https://doi.org/10.1016/j.watres.2015.01.044
Sauerbrei W, Meier-Hirmer C, Benner A, Royston P (2006) Multivariable regression model building by using fractional polynomials: description of SAS, STATA and R programs. Comput Stat Data Anal 50:3464–3485. https://doi.org/10.1016/j.csda.2005.07.015
Schoen ME, Ashbolt NJ, Jahne MA, Garland J (2017) Risk-based enteric pathogen reduction targets for non-potable and direct potable use of roof runoff, stormwater, and greywater. Microb Risk Anal 5:32–43. https://doi.org/10.1016/j.mran.2017.01.002
Sharma M, Handy ET, East CL, Kim S, Jiang C, Callahan MT, Allard SM (2020) Prevalence of Salmonella and Listeria monocytogenes in non-traditional irrigation waters in the Mid-Atlantic United States is affected by water type, season, and recovery method. PLoS ONE 15:1–15. https://doi.org/10.1371/journal.pone.0229365
Sidhu JPS, Ahmed W, Gernjak W, Aryal R, McCarthy D, PalmerA, Kolotelo P, Toze S (2013) Sewage pollution in urban stormwater runoff as evident from the widespread presence of multiple microbial and chemical source tracking markers. Sci Total Environ 463–464:488–496. https://doi.org/10.1016/j.scitotenv.2013.06.020
Silva TFdas, Beltrán G, de Oliveira Nascimento D, Rodríguez N, Mancipe-Muñoz JP N (2022) Assessing major drivers of runoff water quality using principal component analysis: a case study from a colombian and a brazilian catchments. Urban Water Journal. https://doi.org/10.1080/1573062X.2022.2029913
Skovgaard N (2004) Risk assessments of Salmonella in eggs and broiler chickens. Interpretative summary. Int J Food Microbiol 91:223. https://doi.org/10.1016/s0168-1605(03)00369-6
Soupir ML, Mostaghimi S, Dillaha T (2010) Attachment of Escherichia coli and Enterococci to particles in runoff. J Environ Qual 39:1019–1027. https://doi.org/10.2134/jeq2009.0296
Steele JA, Blackwood AD, Griffith JF, Noble RT, Schiff KC (2018) Quantification of pathogens and markers of fecal contamination during storm events along popular surfing beaches in San Diego, California. Water Res 136:137–149. https://doi.org/10.1016/j.watres.2018.01.056
Taguchi VJ, Olsen TA, Natarajan P, Janke BD, Gulliver JS, Finlay JC, Stefan HG (2020) Internal loading in stormwater ponds as a phosphorus source to downstream waters. Limnol Oceanogr Lett 5:322–330. https://doi.org/10.1002/lol2.10155
Teunis P, Takumi K, Shinagawa K (2004) Dose response for infection by Escherichia coli O157: H7 from outbreak data. Risk Anal An Int J 24(2):401–407. https://doi.org/10.1111/j.0272-4332.2004.00441.x
USEPA (2009) Child-Specific Exposure Factors Handbook (2008, Final Report). Washington, DC, EPA/600/R-06/096F, 2008. Last acceced 3 November 2022
USEPA (2002) Guidance for comparing background and chemical concentrations in Soil for CERCLA Sites - EPA 540-R-01-003, OSWER 9285.7–41
Van Abel N, Schoen ME, Kissel JC, Meschke JS (2017) Comparison of risk predicted by multiple norovirus dose–response models and implications for quantitative microbial risk assessment. Risk Anal 37:245–264. https://doi.org/10.1111/risa.12616
Vieira RHSF, Catter KM, Saker-Sampaio S, Rodrigues DP, Theophilo GND, Fonteles-Filho AA (2002) The stormwater drain system as a pollution vector of the seashore in Fortaleza (Ceará State, Brazil). Brazilian J Microbiol 33:294–298. https://doi.org/10.1590/S1517-83822002000400003
Williamson CE, Madronich S, Lal A, Zepp RG, Lucas RM, Overholt EP, Rose KC, Schladow SG, Lee-Taylor J (2017) Climate change-induced increases in precipitation are reducing the potential for solar ultraviolet radiation to inactivate pathogens in surface waters. Sci Rep 7:1–12. https://doi.org/10.1038/s41598-017-13392-2
Wright RS, Tew EL, Decker CE, Von Lehmden DJ, Barnard WF (1987) Performance audits of EPA protocol gases and inspection and maintenance calibration gases. J Air Pollut Control Assoc 37:384–385. https://doi.org/10.1080/08940630.1987.10466233
Xie J, Wu C, Li H, Chen G (2017) Study on storm-water management of grassed swales and permeable pavement based on SWMM. Water (Switzerland) 9(11):840. https://doi.org/10.3390/w9110840
Yazdi MN, Sample DJ, Scott D, Owen JS, Ketabchy M, Alamdari N (2019) Water quality characterization of storm and irrigation runoff from a container nursery. Sci Total Environ 667:166–178. https://doi.org/10.1016/j.scitotenv.2019.02.326
Yeah-King L(2009) Microbial risk assessment of sustainable urban stormwater management practices. PhD Thesis. University of California Irvine
Funding
This research was funded by the Department of Civil and Environmental Engineering at Universidad de los Andes. We would like to thank Empresa de Acueducto de Bogotá for their help with water quality parameter analyses and Juan David Chamat, Sebastián Jaramillo Toro, and Daniel Sarmiento for their support during sampling and sample analysis.
Open Access funding provided by Colombia Consortium
Author information
Authors and Affiliations
Contributions
Juan Pablo Rodríguez and Johana Husserl contributed to the study conception and design. Data collection was conducted by Daniela Salamanca and data analysis was conducted by all authors; statistical analysis was specifically guided by Juan Pablo Ramos-Bonilla. The first draft of the manuscript was written by Daniela Salamanca and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salamanca, D., Husserl, J., Ramos-Bonilla, J.P. et al. Pathogens in Runoff Water Treated by a Sustainable Urban Drainage System in a Developing Country. Environ. Process. 10, 3 (2023). https://doi.org/10.1007/s40710-022-00610-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40710-022-00610-x