Abstract
Purpose
Aim of this study is focusing on bone metabolism in AN patients with amenorrhoea and related estrogen deficiency effects.
Methods
AN patients were compared both with healthy females and with postmenopausal women (reference model for estrogen deficiency). The study sample included 81 females with AN. Laboratory tests [25-OH vitamin D, bone turnover markers, intact parathyroid hormone, sclerostin (SOST) and dickkopf-related protein (DKK1)] and dual energy X-ray absorptiometry (DXA) were taken into account.
Results
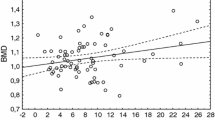
AN patients had higher levels of C-terminal telopeptide of type I collagen (CTX) than both control groups. AN adolescents had CTX higher than AN young adults. In postmenopausal women, intact N-propeptide of type I collagen was higher if compared with each other group. In AN groups, Dickkopf-related protein 1 was significantly lower than the two control groups. No differences were found in sclerostin except in adolescents. In AN adolescents, DXA values at femoral sites were higher than in AN young adults and a positive correlation was found with body weight (p < 0.01) and with fat mass evaluated using DXA (p < 0.01).
Conclusions
AN women with amenorrhoea have an increased bone resorption like postmenopausal women but bone formation is depressed. The consequent remodeling uncoupling is considerably more severe than that occurring after menopause.



Similar content being viewed by others
Change history
29 November 2019
Unfortunately, the sixth author name was incorrectly spelled as ���S. Fassio��� instead of ���A. Fassio��� in the original publication.
References
Favaro A, Caregaro L, Tenconi E et al (2009) Time trends in age at onset of anorexia nervosa and bulimia nervosa. J Clin Psychiatry 70:1715–1721. doi:10.4088/JCP.09m05176blu
Misra M, Klibanski A (2014) Endocrine consequences of anorexia nervosa. Lancet Diabetes Endocrinol 2:581–592. doi:10.1016/S2213-8587(13)70180-3
Zuckerman-Levin N, Hochberg Z, Latzer Y (2014) Bone health in eating disorders. Obes Rev Off J Int Assoc Study Obes 15:215–223. doi:10.1111/obr.12117
Greco EA, Lenzi A, Migliaccio S (2016) The pathophysiological basis of bone tissue alterations associated with eating disorders. Horm Mol Biol Clin Investig. doi:10.1515/hmbci-2016-0006
Gordon CM, Goodman E, Emans SJ et al (2002) Physiologic regulators of bone turnover in young women with anorexia nervosa. J Pediatr 141:64–70. doi:10.1067/mpd.2002.125003
Stefanis N, Mackintosh C, Abraha HD et al (1998) Dissociation of bone turnover in anorexia nervosa. Ann Clin Biochem 35(Pt 6):709–716
Soyka LA, Grinspoon S, Levitsky LL et al (1999) The effects of anorexia nervosa on bone metabolism in female adolescents. J Clin Endocrinol Metab 84:4489–4496. doi:10.1210/jcem.84.12.6207
Faje AT, Fazeli PK, Katzman DK et al (2012) Sclerostin levels and bone turnover markers in adolescents with anorexia nervosa and healthy adolescent girls. Bone 51:474–479. doi:10.1016/j.bone.2012.06.006
Misra M, Aggarwal A, Miller KK et al (2004) Effects of anorexia nervosa on clinical, hematologic, biochemical, and bone density parameters in community-dwelling adolescent girls. Pediatrics 114:1574–1583. doi:10.1542/peds.2004-0540
Soyka LA, Misra M, Frenchman A et al (2002) Abnormal bone mineral accrual in adolescent girls with anorexia nervosa. J Clin Endocrinol Metab 87:4177–4185. doi:10.1210/jc.2001-011889
Misra M, Katzman DK, Cord J et al (2008) Bone metabolism in adolescent boys with anorexia nervosa. J Clin Endocrinol Metab 93:3029–3036. doi:10.1210/jc.2008-0170
Grinspoon S, Miller K, Coyle C et al (1999) Severity of osteopenia in estrogen-deficient women with anorexia nervosa and hypothalamic amenorrhea. J Clin Endocrinol Metab 84:2049–2055. doi:10.1210/jcem.84.6.5792
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Association, Arlington
Institute of Medicine (US) Committee to review dietary reference intakes for vitamin D and calcium (2011) Dietary reference intakes for calcium and vitamin D. National Academies Press, Washington (DC)
Radetti G, Franceschi R, Adami S et al (2014) Higher circulating parathormone is associated with smaller and weaker bones in obese children. Calcif Tissue Int 95:1–7. doi:10.1007/s00223-014-9853-8
Kelly TL, Berger N, Richardson TL (1998) DXA body composition: theory and practice. Appl Radiat Isot 49:511–513
Adami S, Viapiana O, Gatti D et al (2008) Relationship between serum parathyroid hormone, vitamin D sufficiency, age, and calcium intake. Bone 42:267–270. doi:10.1016/j.bone.2007.10.003
Nomura Y, Yoshizaki A, Yoshikata H et al (2013) Study of the distribution by age group of serum cross-linked C-terminal telopeptide of type I collagen and procollagen type I N-propeptide in healthy Japanese women to establish reference values. J Bone Miner Metab 31:644–651. doi:10.1007/s00774-013-0460-y
Jenkins N, Black M, Paul E et al (2013) Age-related reference intervals for bone turnover markers from an Australian reference population. Bone 55:271–276. doi:10.1016/j.bone.2013.04.003
Swenne I, Stridsberg M (2012) Bone metabolism markers in adolescent girls with eating disorders and weight loss: effects of growth, weight trend, developmental and menstrual status. Arch Osteoporos 7:125–133. doi:10.1007/s11657-012-0090-3
Tubić B, Pettersson C, Svedlund A et al (2016) Increased bone mineral content during rapid weight gain therapy in anorexia nervosa. Horm Metab Res 48:664–672. doi:10.1055/s-0042-115304
Adami S, Bianchi G, Brandi ML et al (2008) Determinants of bone turnover markers in healthy premenopausal women. Calcif Tissue Int 82:341–347. doi:10.1007/s00223-008-9126-5
Baron R, Kneissel M (2013) WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat Med 19:179–192. doi:10.1038/nm.3074
Gong Y, Slee RB, Fukai N et al (2001) LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell 107:513–523
Boyden LM, Mao J, Belsky J et al (2002) High bone density due to a mutation in LDL-receptor-related protein 5. N Engl J Med 346:1513–1521. doi:10.1056/NEJMoa013444
Baron R, Rawadi G (2007) Targeting the Wnt/beta-catenin pathway to regulate bone formation in the adult skeleton. Endocrinology 148:2635–2643. doi:10.1210/en.2007-0270
Ott SM (2005) Sclerostin and Wnt signaling–the pathway to bone strength. J Clin Endocrinol Metab 90:6741–6743. doi:10.1210/jc.2005-2370
Anastasilakis AD, Polyzos SA, Toulis KA (2011) Role of wingless tail signaling pathway in osteoporosis: an update of current knowledge. Curr Opin Endocrinol Diabetes Obes 18:383–388. doi:10.1097/MED.0b013e32834afff2
Drake MT, Srinivasan B, Mödder UI et al (2010) Effects of parathyroid hormone treatment on circulating sclerostin levels in postmenopausal women. J Clin Endocrinol Metab 95:5056–5062. doi:10.1210/jc.2010-0720
Diarra D, Stolina M, Polzer K et al (2007) Dickkopf-1 is a master regulator of joint remodeling. Nat Med 13:156–163. doi:10.1038/nm1538
Szulc P, Schoppet M, Rachner TD et al (2014) Severe abdominal aortic calcification in older men is negatively associated with DKK1 serum levels: the STRAMBO study. J Clin Endocrinol Metab 99:617–624. doi:10.1210/jc.2013-3201
Shen Q, Fan J, Yang X-R et al (2012) Serum DKK1 as a protein biomarker for the diagnosis of hepatocellular carcinoma: a large-scale, multicentre study. Lancet Oncol 13:817–826. doi:10.1016/S1470-2045(12)70233-4
Brunkow ME, Gardner JC, Van Ness J et al (2001) Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein. Am J Hum Genet 68:577–589
Robling AG, Niziolek PJ, Baldridge LA et al (2008) Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/sclerostin. J Biol Chem 283:5866–5875. doi:10.1074/jbc.M705092200
Amrein K, Amrein S, Drexler C et al (2012) Sclerostin and its association with physical activity, age, gender, body composition, and bone mineral content in healthy adults. J Clin Endocrinol Metab 97:148–154. doi:10.1210/jc.2011-2152
Urano T, Shiraki M, Ouchi Y, Inoue S (2012) Association of circulating sclerostin levels with fat mass and metabolic disease–related markers in Japanese postmenopausal women. J Clin Endocrinol Metab 97:E1473–E1477. doi:10.1210/jc.2012-1218
Kuipers AL, Zhang Y, Yu S et al (2014) Relative influence of heritability, environment and genetics on serum sclerostin. Osteoporos Int 25:905–912. doi:10.1007/s00198-013-2517-0
Miller KK, Wexler TL, Zha AM et al (2007) Androgen deficiency: association with increased anxiety and depression symptom severity in anorexia nervosa. J Clin Psychiatry 68:959–965
Misra M, Katzman D, Miller KK et al (2011) Physiologic estrogen replacement increases bone density in adolescent girls with anorexia nervosa. J Bone Miner Res 26:2430–2438. doi:10.1002/jbmr.447
Miller KK, Grieco KA, Mulder J et al (2004) Effects of risedronate on bone density in anorexia nervosa. J Clin Endocrinol Metab 89:3903–3906. doi:10.1210/jc.2003-031885
Miller KK, Meenaghan E, Lawson EA et al (2011) Effects of risedronate and low-dose transdermal testosterone on bone mineral density in women with anorexia nervosa: a randomized, placebo-controlled study. J Clin Endocrinol Metab 96:2081–2088. doi:10.1210/jc.2011-0380
Golden NH, Iglesias EA, Jacobson MS et al (2005) Alendronate for the treatment of osteopenia in anorexia nervosa: a randomized, double-blind, placebo-controlled trial. J Clin Endocrinol Metab 90:3179–3185. doi:10.1210/jc.2004-1659
Fazeli PK, Wang IS, Miller KK et al (2014) Teriparatide increases bone formation and bone mineral density in adult women with anorexia nervosa. J Clin Endocrinol Metab 99:1322–1329. doi:10.1210/jc.2013-4105
Caprio M, Infante M, Calanchini M et al (2016) Vitamin D: not just the bone. Evidence for beneficial pleiotropic extraskeletal effects. Eat Weight Disord. doi:10.1007/s40519-016-0312-6
Maïmoun L, Guillaume S, Lefebvre P et al (2016) Evidence of a link between resting energy expenditure and bone remodelling, glucose homeostasis and adipokine variations in adolescent girls with anorexia nervosa. Osteoporos Int 27:135–146. doi:10.1007/s00198-015-3223-x
Acknowledgements
We thank Caterina Fraccarollo for the ELISA assays.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Viapiana O has received speaking fees from Abiogen, Amgen, Merck Sharp & Dohme Corp. Idolazzi L has received speaking fees from Eli Lilly, UCB, Abbvie, Novartis. Fassio A declares he has no conflict of interests. Caimmi C declares he has no conflict of interests. Rossini M has received speaking and consulting fees from Abiogen, Amgen, Eli-Lilly and Merck Sharp & Dohme Corp. Davide Gatti has received speaking fees from Abiogen, Amgen, Eli-Lilly and Neopharmed-Gentili. El Ghoch M, Dalle Grave R, Bazzani PV, Calugi S, Bertoldo F, Braga V declare they have no conflict of interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Idolazzi, L., El Ghoch, M., Dalle Grave, R. et al. Bone metabolism in patients with anorexia nervosa and amenorrhoea. Eat Weight Disord 23, 255–261 (2018). https://doi.org/10.1007/s40519-016-0337-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40519-016-0337-x