Abstract
Purpose of Review
Human African Trypanosomiasis (HAT), also known as sleeping sickness, is a vector-borne parasitic neglected tropical disease (NTD) endemic in sub-Saharan Africa. This review aims to enhance our understanding of HAT and provide valuable insights to combat this significant public health issue by synthesizing the latest research and evidence.
Recent Findings
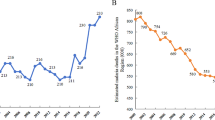
HAT has reached a historical < 1000 cases in 2018. In patients without neurologic symptoms and signs, the likelihood of a severe meningoencephalitic stage is deemed low, obviating the need for a lumbar puncture to guide treatment decisions using fexinidazole.
Summary
Both forms of the disease, gambiense HAT (gHAT) and rhodesiense HAT (rHAT), have specific epidemiology, risk factors, diagnosis, and treatment. Disease management still requires a high index of suspicion, infectious disease expertise, and specialized medical care. Essential stakeholders in health policy are critical to accomplishing the elimination goals of the NTD roadmap for 2021–2030.





Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Trypanosomiasis, human African (sleeping sickness). https://www.who.int/news-room/fact-sheets/detail/trypanosomiasis-human-african-(sleeping-sickness). Accessed 28 Aug 2023
Papagni R, Novara R, Minardi ML, et al. Human African trypanosomiasis (sleeping sickness): current knowledge and future challenges. Front Trop Dis. 2023:4. https://www.frontiersin.org/articles/10.3389/fitd.2023.1087003. Accessed 28 Aug 2023
Simarro PP, Cecchi G, Paone M, et al. The Atlas of human African trypanosomiasis: a contribution to global mapping of neglected tropical diseases. Int J Health Geogr. 2010;9:57. https://doi.org/10.1186/1476-072X-9-57.
Büscher P, Cecchi G, Jamonneau V, Priotto G. Human African trypanosomiasis. Lancet. 2017;390(10110):2397–409. https://doi.org/10.1016/S0140-6736(17)31510-6.
Bodimeade C, Marks M, Mabey D. Neglected tropical diseases: elimination and eradication. Clin Med. 2019;19(2):157–60. https://doi.org/10.7861/clinmedicine.19-2-157.
Simarro PP, Cecchi G, Franco JR, et al. Monitoring the progress towards the elimination of gambiense human African trypanosomiasis. PLoS Negl Trop Dis. 2015;9(6):e0003785. https://doi.org/10.1371/journal.pntd.0003785.
Steverding D. The history of African trypanosomiasis. Parasit Vectors. 2008;1:3. https://doi.org/10.1186/1756-3305-1-3.
The elimination of human African trypanosomiasis: achievements in relation to WHO road map targets for 2020 | LOS Neglected Tropical Diseases. https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0010047. Accessed 28 Aug 2023
Franco JR, Cecchi G, Priotto G, et al. Monitoring the elimination of human African trypanosomiasis at continental and country level: update to 2018. PLoS Negl Trop Dis. 2020;14(5):e0008261. https://doi.org/10.1371/journal.pntd.0008261.
Franco JR, Simarro PP, Diarra A, Jannin JG. Epidemiology of human African trypanosomiasis. Clin Epidemiol. 2014;6:257–75. https://doi.org/10.2147/CLEP.S39728.
Cecchi G, Courtin F, Paone M, et al. Mapping sleeping sickness in Western Africa in a context of demographic transition and climate change. Parasite Paris Fr. 2009;16(2):99–106. https://doi.org/10.1051/parasite/2009162099.
Hasker E, Hope A, Bottieau E. Gambiense human African trypanosomiasis: the bumpy road to elimination. Curr Opin Infect Dis. 2022;35(5):384–9. https://doi.org/10.1097/QCO.0000000000000860.
• Elenga VA, Lissom A, Elion DOA, et al. Risk factors and prevalence of human African trypanosomiasis in individuals living in remote areas of the Republic of Congo. BMC Public Health. 2022;22(1):2322. https://doi.org/10.1186/s12889-022-14577-9. Study described contemporary socioeconomic risk factors for HAT in The Republic of Congo.
Kagbadouno MS, Camara M, Rouamba J, et al. Epidemiology of sleeping sickness in Boffa (Guinea): where are the trypanosomes? PLoS Negl Trop Dis. 2012;6(12):e1949. https://doi.org/10.1371/journal.pntd.0001949.
Simarro PP, Cecchi G, Franco JR, et al. Risk for human African trypanosomiasis, Central Africa, 2000–2009. Emerg Infect Dis. 2011;17(12):2322–4. https://doi.org/10.3201/eid1712.110921.
Selby R, Wamboga C, Erphas O, et al. Gambian human African trypanosomiasis in North West Uganda. Are we on course for the 2020 target? PLoS Negl Trop Dis. 2019;13(8):e0007550. https://doi.org/10.1371/journal.pntd.0007550.
Ruiz-Postigo JA, Franco JR, Lado M, Simarro PP. Human African trypanosomiasis in South Sudan: how can we prevent a new epidemic? PLoS Negl Trop Dis. 2012;6(5):e1541. https://doi.org/10.1371/journal.pntd.0001541.
Bemba I, Bamou R, Lenga A, Okoko A, Awono-Ambene P, Antonio-Nkondjio C. Review of the situation of human African trypanosomiasis in the Republic of Congo from the 1950s to 2020. J Med Entomol. 2022;59(2):421–9. https://doi.org/10.1093/jme/tjab225.
Lumbala C, Simarro PP, Cecchi G, et al. Human African trypanosomiasis in the Democratic Republic of the Congo: disease distribution and risk. Int J Health Geogr. 2015;14:20. https://doi.org/10.1186/s12942-015-0013-9.
Atlas of HAT. https://www.who.int/teams/control-of-neglected-tropical-diseases/human-african-trypanosomiasis/atlas-of-hat. Accessed 28 Aug 2023
Lindner AK, Priotto G. The unknown risk of vertical transmission in sleeping sickness--a literature review. PLoS Negl Trop Dis. 2010;4(12):e783. https://doi.org/10.1371/journal.pntd.0000783.
Malvy D, Chappuis F. Sleeping sickness. Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis. 2011;17(7):986–95. https://doi.org/10.1111/j.1469-0691.2011.03536.x.
Pays E, Radwanska M, Magez S. The pathogenesis of African trypanosomiasis. Annu Rev Pathol. 2023;18:19–45. https://doi.org/10.1146/annurev-pathmechdis-031621-025153.
Blum JA, Neumayr AL, Hatz CF. Human African trypanosomiasis in endemic populations and travellers. Eur J Clin Microbiol Infect Dis Off Publ Eur Soc Clin Microbiol. 2012;31(6):905–13. https://doi.org/10.1007/s10096-011-1403-y.
Bottieau E, Clerinx J. Human African trypanosomiasis: progress and stagnation. Infect Dis Clin North Am. 2019;33(1):61–77. https://doi.org/10.1016/j.idc.2018.10.003.
Sudarshi D, Brown M. Human African trypanosomiasis in non-endemic countries. Clin Med Lond Engl. 2015;15(1):70–3. https://doi.org/10.7861/clinmedicine.15-1-70.
Migchelsen SJ, Büscher P, Hoepelman AIM, Schallig HDFH, Adams ER. Human African trypanosomiasis: a review of non-endemic cases in the past 20 years. Int J Infect Dis IJID Off Publ Int Soc Infect Dis. 2011;15(8):e517–24. https://doi.org/10.1016/j.ijid.2011.03.018.
Gao JM, Qian ZY, Hide G, Lai DH, Lun ZR, Wu ZD. Human African trypanosomiasis: the current situation in endemic regions and the risks for non-endemic regions from imported cases. Parasitology. 147(9):922–31. https://doi.org/10.1017/S0031182020000645.
Gobbi F, Bisoffi Z. Human African trypanosomiasis in travellers to Kenya. Euro Surveill Bull Eur Sur Mal Transm Eur Commun Dis Bull. 2012;17(10):20109.
• Franco JR, Cecchi G, Priotto G, et al. Human African trypanosomiasis cases diagnosed in non-endemic countries (2011-2020). PLoS Negl Trop Dis. 2022;16(11):e0010885. https://doi.org/10.1371/journal.pntd.0010885. Study revised the clinical characteristics of HAT cases diagnosed in non-endemic countries.
Gautret P, Clerinx J, Caumes E, et al. Imported human African trypanosomiasis in Europe, 2005-2009. Euro Surveill Bull Eur Sur Mal Transm Eur Commun Dis Bull. 2009;14(36):19327.
Simarro PP, Franco JR, Cecchi G, et al. Human African trypanosomiasis in non-endemic countries (2000-2010). J Travel Med. 2012;19(1):44–53. https://doi.org/10.1111/j.1708-8305.2011.00576.x.
Kennedy PGE. Human African trypanosomiasis-neurological aspects. J Neurol. 2006;253(4):411–6. https://doi.org/10.1007/s00415-006-0093-3.
Kennedy PGE, Rodgers J. Clinical and neuropathogenetic aspects of human African trypanosomiasis. Front Immunol. 2019;10:39. https://doi.org/10.3389/fimmu.2019.00039.
Naessens J, Mwangi DM, Buza J, Moloo SK. Local skin reaction (chancre) induced following inoculation of metacyclic trypanosomes in cattle by tsetse flies is dependent on CD4 T lymphocytes. Parasite Immunol. 2003;25(8-9):413–9. https://doi.org/10.1111/j.1365-3024.2003.00649.x.
Duggan AJ, Hutchinson MP. Sleeping sickness in Europeans: a review of 109 cases. J Trop Med Hyg. 1966;69(6):124–31.
Malvy D, Djossou F, Weill FX, Chapuis P, Longy-Boursier M, Le Bras M. Guess what! Human West African trypanosomiasis with chancre presentation. Eur J Dermatol EJD. 2000;10(7):561–2.
Lejon V, Bentivoglio M, Franco JR. Human African trypanosomiasis. Handb Clin Neurol. 2013;114:169–81. https://doi.org/10.1016/B978-0-444-53490-3.00011-X.
Reincke M, Arlt W, Heppner C, Petzke F, Chrousos GP, Allolio B. Neuroendocrine dysfunction in African trypanosomiasis. The role of cytokines. Ann N Y Acad Sci. 1998;840:809–21. https://doi.org/10.1111/j.1749-6632.1998.tb09619.x.
Ortiz HIA, Farina JM, Saldarriaga C, et al. Human African trypanosomiasis & heart. Expert Rev Cardiovasc Ther. 2020;18(12):859–65. https://doi.org/10.1080/14779072.2020.1828066.
Blum JA, Zellweger MJ, Burri C, Hatz C. Cardiac involvement in African and American trypanosomiasis. Lancet Infect Dis. 2008;8(10):631–41. https://doi.org/10.1016/S1473-3099(08)70230-5.
Stephan C, Just-Nuebling G, Fichtlscherer S, Kriener S, Brodt HR. Winterbottom’s sign and hypertrophic cardiomyopathy. Scand J Infect Dis. 2002;34(7):544–5. https://doi.org/10.1080/003655402320208848.
Urech K, Neumayr A, Blum J. Sleeping sickness in travelers - do they really sleep? PLoS Negl Trop Dis. 2011;5(11):e1358. https://doi.org/10.1371/journal.pntd.0001358.
Kennedy PG. Clinical features, diagnosis, and treatment of human African trypanosomiasis (sleeping sickness). Lancet Neurol. 2013;12(2):186–94. https://doi.org/10.1016/S1474-4422(12)70296-X.
Blum J, Schmid C, Burri C. Clinical aspects of 2541 patients with second stage human African trypanosomiasis. Acta Trop. 2006;97(1):55–64. https://doi.org/10.1016/j.actatropica.2005.08.001.
Kennedy PGE. The continuing problem of human African trypanosomiasis (sleeping sickness). Ann Neurol. 2008;64(2):116–26. https://doi.org/10.1002/ana.21429.
Stich A, Abel PM, Krishna S. Human African trypanosomiasis. BMJ. 2002;325(7357):203–6.
WHO interim guidelines for the treatment of gambiense human African trypanosomiasis. https://www.who.int/publications-detail-redirect/9789241550567. Accessed 28 Aug 2023
Human African trypanosomiasis: epidemiology, clinical manifestations, and diagnosis - UpToDate. https://www.uptodate.com/contents/human-african-trypanosomiasis-epidemiology-clinical-manifestations-and-diagnosis?search=hat&source=search_result&selectedTitle=1~64&usage_type=default&display_rank=1. Accessed 28 Aug 2023
Chappuis F, Loutan L, Simarro P, Lejon V, Büscher P. Options for field diagnosis of human african trypanosomiasis. Clin Microbiol Rev. 2005;18(1):133–46. https://doi.org/10.1128/CMR.18.1.133-146.2005.
Inojosa WO, Augusto I, Bisoffi Z, et al. Diagnosing human African trypanosomiasis in Angola using a card agglutination test: observational study of active and passive case finding strategies. BMJ. 2006;332(7556):1479. https://doi.org/10.1136/bmj.38859.531354.7C.
Magnus E, Vervoort T, Van Meirvenne N. A card-agglutination test with stained trypanosomes (C.A.T.T.) for the serological diagnosis of T. B. gambiense trypanosomiasis. Ann Soc Belg Med Trop. 1978;58(3):169–76.
Van Meirvenne N, Magnus E, Buscher P. Evaluation of variant specific trypanolysis tests for serodiagnosis of human infections with Trypanosoma brucei gambiense. Acta Trop. 1995;60(3):189–99. https://doi.org/10.1016/0001-706x(95)00127-z.
Mumba Ngoyi D, Ali Ekangu R, Mumvemba Kodi MF, et al. Performance of parasitological and molecular techniques for the diagnosis and surveillance of gambiense sleeping sickness. PLoS Negl Trop Dis. 2014;8(6):e2954. https://doi.org/10.1371/journal.pntd.0002954.
Miézan TW, Meda HA, Doua F, Djè NN, Lejon V, Büscher P. Single centrifugation of cerebrospinal fluid in a sealed pasteur pipette for simple, rapid and sensitive detection of trypanosomes. Trans R Soc Trop Med Hyg. 2000;94(3):293. https://doi.org/10.1016/s0035-9203(00)90327-4.
Wellde BT, Chumo DA, Reardon MJ, et al. Diagnosis of Rhodesian sleeping sickness in the Lambwe Valley (1980-1984). Ann Trop Med Parasitol. 1989;83(Suppl 1):63–71. https://doi.org/10.1080/00034983.1989.11812410.
MacLean LM, Odiit M, Chisi JE, Kennedy PGE, Sternberg JM. Focus–specific clinical profiles in human African trypanosomiasis caused by Trypanosoma brucei rhodesiense. PLoS Negl Trop Dis. 2010;4(12):e906. https://doi.org/10.1371/journal.pntd.0000906.
Fairlamb AH. Fexinidazole for the treatment of human African trypanosomiasis. Drugs Today Barc Spain 1998. 2019;55(11):705–12. https://doi.org/10.1358/dot.2019.55.11.3068795.
Keating J, Yukich JO, Sutherland CS, Woods G, Tediosi F. Human African trypanosomiasis prevention, treatment and control costs: a systematic review. Acta Trop. 2015;150:4–13. https://doi.org/10.1016/j.actatropica.2015.06.003.
• VKBK M, Kalonji WM, Bardonneau C, et al. Oral fexinidazole for late-stage African Trypanosoma brucei gambiense trypanosomiasis: a pivotal multicentre, randomised, non-inferiority trial. Lancet Lond Engl. 2018;391(10116):144–54. https://doi.org/10.1016/S0140-6736(17)32758-7. Clinical trial demonstrating oral fexinidazole is effective and safe for the treatment of gHAT infection compared with nifurtimox eflornithine combination therapy in late stage.
Lejon V, Büscher P. Review Article: cerebrospinal fluid in human African trypanosomiasis: a key to diagnosis, therapeutic decision and post-treatment follow-up. Trop Med Int Health TM IH. 2005;10(5):395–403. https://doi.org/10.1111/j.1365-3156.2005.01403.x.
Nok AJ. Arsenicals (melarsoprol), pentamidine and suramin in the treatment of human African trypanosomiasis. Parasitol Res. 2003;90(1):71–9. https://doi.org/10.1007/s00436-002-0799-9.
Seixas J, Atouguia J, Josenando T, et al. Clinical study on the melarsoprol-related encephalopathic syndrome: risk factors and HLA association. Trop Med Infect Dis. 2020;5(1):5. https://doi.org/10.3390/tropicalmed5010005.
Robays J, Nyamowala G, Sese C, et al. High failure rates of melarsoprol for sleeping sickness, Democratic Republic of Congo. Emerg Infect Dis. 2008;14(6):966–7. https://doi.org/10.3201/eid1406.071266.
New drugs for human African trypanosomiasis: a twenty first century success story. https://pubmed.ncbi.nlm.nih.gov/32092897/. Accessed 6 Sept 2023
• Betu Kumeso VK, Kalonji WM, Rembry S, et al. Efficacy and safety of acoziborole in patients with human African trypanosomiasis caused by Trypanosoma brucei gambiense: a multicentre, open-label, single-arm, phase 2/3 trial. Lancet Infect Dis. 2023;23(4):463–70. https://doi.org/10.1016/S1473-3099(22)00660-0. Given the high efficacy and favourable safety profile, acoziborole hold promise as a new HAT therapy.
Kaiser M, Bray MA, Cal M, Bourdin Trunz B, Torreele E, Brun R. Antitrypanosomal activity of fexinidazole, a new oral nitroimidazole drug candidate for treatment of sleeping sickness. Antimicrob Agents Chemother. 2011;55(12):5602–8. https://doi.org/10.1128/AAC.00246-11.
Study Record | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT03974178?tab=results. Accessed 6 Sept 2023
Lejon V, Jacobs J, Simarro PP. Elimination of sleeping sickness hindered by difficult diagnosis. Bull World Health Organ. 2013;91(10):718. https://doi.org/10.2471/BLT.13.126474.
Koffi M, N’Djetchi M, Ilboudo H, et al. A targeted door-to-door strategy for sleeping sickness detection in low-prevalence settings in Côte d’Ivoire. Parasite Paris Fr. 2016;23:51. https://doi.org/10.1051/parasite/2016059.
Snijders R, Fukinsia A, Claeys Y, et al. Cost of a new method of active screening for human African trypanosomiasis in the Democratic Republic of the Congo. PLoS Negl Trop Dis. 2020;14(12):e0008832. https://doi.org/10.1371/journal.pntd.0008832.
Álvarez-Rodríguez A, Jin BK, Radwanska M, Magez S. Recent progress in diagnosis and treatment of Human African Trypanosomiasis has made the elimination of this disease a realistic target by 2030. Front Med. 2022;9:1037094. https://doi.org/10.3389/fmed.2022.1037094.
Is vector control needed to eliminate gambiense human African trypanosomiasis?. https://pubmed.ncbi.nlm.nih.gov/23914350/. Accessed 7 Sept 2023
Franco JR, Simarro PP, Diarra A, Ruiz-Postigo JA, Jannin JG. The journey towards elimination of gambiense human African trypanosomiasis: not far, nor easy. Parasitology. 2014;141(6):748–60. https://doi.org/10.1017/S0031182013002102.
Author information
Authors and Affiliations
Contributions
YOM wrote the main manuscript. AAHM edited the manuscript and provided supervision. MGK, FB, and SL provided critical feedback on the manuscript draft.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ortiz-Martínez, Y., Kouamé, M.G., Bongomin, F. et al. Human African Trypanosomiasis (Sleeping Sickness)—Epidemiology, Clinical Manifestations, Diagnosis, Treatment, and Prevention. Curr Trop Med Rep 10, 222–234 (2023). https://doi.org/10.1007/s40475-023-00304-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40475-023-00304-w