Abstract
Purpose of Review
Drug addiction starts from recreational drug use, gradually becomes habitual or even compulsive. However, after exposure to addictive drugs, only 20% of individuals finally fall into a vicious cycle of addiction. As the number of addicts worldwide increases year by year, the need for understanding the neural basis underlying addiction is in high demand.
Recent Findings
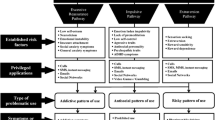
The intrastriatal functional shifts have been widely recognized to contribute to addiction development, where the nucleus accumbens permits the devolution of its control over behaviour to the dorsal striatum. In addition, compulsive drug seeking and taking have been taken as maladaptive habits, coupling with impaired goal-directed behaviour, thus habitual system dominant behaviour in spite of punishment. However, this hypothesis has not fully been proven at the level of neural mechanisms.
Summary
Here we elaborated the function of the striatum at the different stages of addiction within the associative learning theory framework to highlight the potentially vulnerable targets of addictive drugs. Furthermore, based on current findings, we proposed new possibilities to explain compulsive drug-seeking behaviour.


Similar content being viewed by others
Data Availability
N/A
Code Availability
N/A
References
Cartoni E, Balleine B, Baldassarre G. Appetitive Pavlovian-instrumental Transfer: a review. Neurosci Biobehav Rev. 2016.https://doi.org/10.1016/j.neubiorev.2016.09.020.
Morse AK, Leung BK, Heath E, Bertran-Gonzalez J, Pepin E, Chieng BC, et al. Basolateral amygdala drives a GPCR-mediated striatal memory necessary for predictive learning to influence choice. Neuron. 2020.https://doi.org/10.1016/j.neuron.2020.03.007.
Garbusow M, Schad DJ, Sebold M, Friedel E, Bernhardt N, Koch SP, et al. Pavlovian-to-instrumental transfer effects in the nucleus accumbens relate to relapse in alcohol dependence. Addict Biol. 2016.https://doi.org/10.1111/adb.12243.
Mendelsohn A, Pine A, Schiller D. Between thoughts and actions: motivationally salient cues invigorate mental action in the human brain. Neuron. 2014.https://doi.org/10.1016/j.neuron.2013.10.019.
Lüscher C, Robbins TW, Everitt BJ. The transition to compulsion in addiction. Nat Rev Neurosci. 2020.https://doi.org/10.1038/s41583-020-0289-z.
Dalley JW, Everitt BJ, Robbins TW. Impulsivity, compulsivity, and top-down cognitive control. Neuron. 2011.https://doi.org/10.1016/j.neuron.2011.01.020.
Everitt BJ, Robbins TW. Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol. 2016.https://doi.org/10.1146/annurev-psych-122414-033457.
Wolf ME. Synaptic mechanisms underlying persistent cocaine craving. Nat Rev Neurosci. 2016.https://doi.org/10.1038/nrn.2016.39.
Everitt BJ, Robbins TW. Neural systems of reinforcement for drug addiction: from actions to habits to compulsion. Nat Neurosci. 2005.https://doi.org/10.1038/nn1579.
Rescorla RA, Solomon RL. Two-process learning theory: relationships between Pavlovian conditioning and instrumental learning. Psychol Rev. 1967. https://doi.org/10.1037/h0024475.
Yin HH, Ostlund SB, Balleine BW. Reward-guided learning beyond dopamine in the nucleus accumbens: the integrative functions of cortico-basal ganglia networks. Eur J Neurosci. 2008.https://doi.org/10.1111/j.1460-9568.2008.06422.x.
Corbit LH, Balleine BW. Learning and motivational processes contributing to Pavlovian-instrumental transfer and their neural bases: dopamine and beyond. Curr Top Behav Neurosci. 2016. https://doi.org/10.1007/7854_2015_388.
Di Chiara G, Imperato A. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Natl Acad Sci U S A. 1988. https://doi.org/10.1073/pnas.85.14.5274.
Nestler EJ. Is there a common molecular pathway for addiction? Nat Neurosci. 2005. https://doi.org/10.1038/nn1578.
Maskos U, Molles BE, Pons S, Besson M, Guiard BP, Guilloux JP, et al. Nicotine reinforcement and cognition restored by targeted expression of nicotinic receptors. Nature. 2005. https://doi.org/10.1038/nature03694.
Johnson SW, North RA. Opioids excite dopamine neurons by hyperpolarization of local interneurons. J Neurosci. 1992. https://doi.org/10.1523/jneurosci.12-02-00483.1992.
Montague PR, Dayan P, Sejnowski TJ. A framework for mesencephalic dopamine systems based on predictive Hebbian learning. J Neurosci. 1996. https://doi.org/10.1523/jneurosci.16-05-01936.1996.
Schultz W. Predictive reward signal of dopamine neurons. J Neurophysiol. 1998. https://doi.org/10.1152/jn.1998.80.1.1.
Mameli M, Lüscher C. Synaptic plasticity and addiction: learning mechanisms gone awry. Neuropharmacology. 2011. https://doi.org/10.1016/j.neuropharm.2011.01.036.
Heymann G, Jo YS, Reichard KL, McFarland N, Chavkin C, Palmiter RD, et al. Synergy of distinct dopamine projection populations in behavioral reinforcement. Neuron. 2020. https://doi.org/10.1016/j.neuron.2019.11.024.
Stuber GD, Sparta DR, Stamatakis AM, van Leeuwen WA, Hardjoprajitno JE, Cho S, et al. Excitatory transmission from the amygdala to nucleus accumbens facilitates reward seeking. Nature. 2011. https://doi.org/10.1038/nature10194.
Rocha A, Kalivas PW. Role of the prefrontal cortex and nucleus accumbens in reinstating methamphetamine seeking. Eur J Neurosci. 2010. https://doi.org/10.1111/j.1460-9568.2010.07134.x.
Zhou Y, Zhu H, Liu Z, Chen X, Su X, Ma C, et al. A ventral CA1 to nucleus accumbens core engram circuit mediates conditioned place preference for cocaine. Nat Neurosci. 2019. https://doi.org/10.1038/s41593-019-0524-y.
de Jong JW, Afjei SA, PollakDorocic I, Peck JR, Liu C, Kim CK, et al. A neural circuit mechanism for encoding aversive stimuli in the mesolimbic dopamine system. Neuron. 2019. https://doi.org/10.1016/j.neuron.2018.11.005.
Puaud M, Higuera-Matas A, Brunault P, Everitt BJ, Belin D. The basolateral amygdala to nucleus accumbens core circuit mediates the conditioned reinforcing effects of cocaine-paired cues on cocaine seeking. Biol Psychiat. 2021. https://doi.org/10.1016/j.biopsych.2020.07.022.
Alaghband Y, Kramár E, Kwapis JL, Kim ES, Hemstedt TJ, López AJ, et al. CREST in the nucleus accumbens core regulates cocaine conditioned place preference, cocaine-seeking behavior, and synaptic plasticity. J Neurosci. 2018. https://doi.org/10.1523/jneurosci.2911-17.2018.
Ito R, Dalley JW, Howes SR, Robbins TW, Everitt BJ. Dissociation in conditioned dopamine release in the nucleus accumbens core and shell in response to cocaine cues and during cocaine-seeking behavior in rats. J Neurosci. 2000. https://doi.org/10.1523/jneurosci.20-19-07489.2000.
Zinsmaier AK, Dong Y, Huang YH. Cocaine-induced projection-specific and cell type-specific adaptations in the nucleus accumbens. Mol Psychiatry. 2021. https://doi.org/10.1038/s41380-021-01112-2.
Corre J, van Zessen R, Loureiro M, Patriarchi T, Tian L, Pascoli V, et al. Dopamine neurons projecting to medial shell of the nucleus accumbens drive heroin reinforcement. eLife. 2018; https://doi.org/10.7554/eLife.39945.
Pontieri FE, Tanda G, Di Chiara G. Intravenous cocaine, morphine, and amphetamine preferentially increase extracellular dopamine in the “shell” as compared with the “core” of the rat nucleus accumbens. Proc Natl Acad Sci U S A. 1995. https://doi.org/10.1073/pnas.92.26.12304.
Jedynak J, Hearing M, Ingebretson A, Ebner SR, Kelly M, Fischer RA, et al. Cocaine and amphetamine induce overlapping but distinct patterns of AMPAR plasticity in nucleus accumbens medium spiny neurons. Neuropsychopharmacol. 2016. https://doi.org/10.1038/npp.2015.168.
Ito R, Dalley JW, Robbins TW, Everitt BJ. Dopamine release in the dorsal striatum during cocaine-seeking behavior under the control of a drug-associated cue. J Neurosci. 2002. https://doi.org/10.1523/jneurosci.22-14-06247.2002.
McGeorge AJ, Faull RL. The organization of the projection from the cerebral cortex to the striatum in the rat. Neuroscience. 1989. https://doi.org/10.1016/0306-4522(89)90128-0.
Hintiryan H, Foster NN, Bowman I, Bay M, Song MY, Gou L, et al. The mouse cortico-striatal projectome. Nat Neurosci. 2016. https://doi.org/10.1038/nn.4332.
Hilário MR, Costa RM. High on habits. Front Neurosci. 2008. https://doi.org/10.3389/neuro.01.030.2008.
Balleine BW, Liljeholm M, Ostlund SB. The integrative function of the basal ganglia in instrumental conditioning. Behav Brain Res. 2009. https://doi.org/10.1016/j.bbr.2008.10.034.
Gremel CM, Chancey JH, Atwood BK, Luo G, Neve R, Ramakrishnan C, et al. Endocannabinoid modulation of orbitostriatal circuits gates habit formation. Neuron. 2016. https://doi.org/10.1016/j.neuron.2016.04.043.
Vandaele Y, Mahajan NR, Ottenheimer DJ, Richard JM, Mysore SP, Janak PH. Distinct recruitment of dorsomedial and dorsolateral striatum erodes with extended training. eLife. 2019; https://doi.org/10.7554/eLife.49536.
Bergstrom HC, Lipkin AM, Lieberman AG, Pinard CR, Gunduz-Cinar O, Brockway ET, et al. Dorsolateral striatum engagement interferes with early discrimination learning. Cell Rep. 2018. https://doi.org/10.1016/j.celrep.2018.04.081.
Kreitzer AC, Malenka RC. Striatal plasticity and basal ganglia circuit function. Neuron. 2008. https://doi.org/10.1016/j.neuron.2008.11.005.
Li Y, He Y, Chen M, Pu Z, Chen L, Li P, et al. Optogenetic activation of adenosine A2A receptor signaling in the dorsomedial striatopallidal neurons suppresses goal-directed behavior. Neuropsychopharmacol. 2016. https://doi.org/10.1038/npp.2015.227.
Furlong TM, Supit AS, Corbit LH, Killcross S, Balleine BW. Pulling habits out of rats: adenosine 2A receptor antagonism in dorsomedial striatum rescues meth-amphetamine-induced deficits in goal-directed action. Addict Biol. 2017. https://doi.org/10.1111/adb.12316.
Vicente AM, Galvão-Ferreira P, Tecuapetla F, Costa RM. Direct and indirect dorsolateral striatum pathways reinforce different action strategies. Curr Biol. 2016. https://doi.org/10.1016/j.cub.2016.02.036.
O’Hare JK, Ade KK, Sukharnikova T, Van Hooser SD, Palmeri ML, Yin HH, et al. Pathway-specific striatal substrates for habitual behavior. Neuron. 2016. https://doi.org/10.1016/j.neuron.2015.12.032.
Schmidt R, Leventhal DK, Mallet N, Chen F, Berke JD. Canceling actions involves a race between basal ganglia pathways. Nat Neurosci. 2013. https://doi.org/10.1038/nn.3456.
Sjoerds Z, de Wit S, van den Brink W, Robbins TW, Beekman AT, Penninx BW, et al. Behavioral and neuroimaging evidence for overreliance on habit learning in alcohol-dependent patients. Transl Psychiatry. 2013. https://doi.org/10.1038/tp.2013.107.
Corbit LH, Nie H, Janak PH. Habitual alcohol seeking: time course and the contribution of subregions of the dorsal striatum. Biol Psychiat. 2012. https://doi.org/10.1016/j.biopsych.2012.02.024.
Rogers RD, Everitt BJ, Baldacchino A, Blackshaw AJ, Swainson R, Wynne K, et al. Dissociable deficits in the decision-making cognition of chronic amphetamine abusers, opiate abusers, patients with focal damage to prefrontal cortex, and tryptophan-depleted normal volunteers: evidence for monoaminergic mechanisms. Neuropsychopharmacol. 1999. https://doi.org/10.1016/s0893-133x(98)00091-8.
Bechara A, Dolan S, Denburg N, Hindes A, Anderson SW, Nathan PE. Decision-making deficits, linked to a dysfunctional ventromedial prefrontal cortex, revealed in alcohol and stimulant abusers. Neuropsychologia. 2001. https://doi.org/10.1016/s0028-3932(00)00136-6.
Miller EK. The prefrontal cortex and cognitive control. Nat Rev Neurosci. 2000. https://doi.org/10.1038/35036228.
Loos M, Mueller T, Gouwenberg Y, Wijnands R, van der Loo RJ, Birchmeier C, et al. Neuregulin-3 in the mouse medial prefrontal cortex regulates impulsive action. Biol Psychiat. 2014. https://doi.org/10.1016/j.biopsych.2014.02.011.
Paine TA, O’Hara A, Plaut B, Lowes DC. Effects of disrupting medial prefrontal cortex GABA transmission on decision-making in a rodent gambling task. Psychopharmacology. 2015. https://doi.org/10.1007/s00213-014-3816-7.
Narayanan NS, Laubach M. Inhibitory control: mapping medial frontal cortex. Curr Biol. 2017. https://doi.org/10.1016/j.cub.2017.01.010.
Ersche KD, Williams GB, Robbins TW, Bullmore ET. Meta-analysis of structural brain abnormalities associated with stimulant drug dependence and neuroimaging of addiction vulnerability and resilience. Curr Opin Neurobiol. 2013. https://doi.org/10.1016/j.conb.2013.02.017.
Chen BT, Yau HJ, Hatch C, Kusumoto-Yoshida I, Cho SL, Hopf FW, et al. Rescuing cocaine-induced prefrontal cortex hypoactivity prevents compulsive cocaine seeking. Nature. 2013. https://doi.org/10.1038/nature12024.
Hu Y, Salmeron BJ, Krasnova IN, Gu H, Lu H, Bonci A, et al. Compulsive drug use is associated with imbalance of orbitofrontal- and prelimbic-striatal circuits in punishment-resistant individuals. Proc Natl Acad Sci U S A. 2019. https://doi.org/10.1073/pnas.1819978116.
Pascoli V, Hiver A, Van Zessen R, Loureiro M, Achargui R, Harada M, et al. Stochastic synaptic plasticity underlying compulsion in a model of addiction. Nature. 2018. https://doi.org/10.1038/s41586-018-0789-4.
Pascoli V, Terrier J, Hiver A, Lüscher C. Sufficiency of mesolimbic dopamine neuron stimulation for the progression to addiction. Neuron. 2015. https://doi.org/10.1016/j.neuron.2015.10.017.
Sul JH, Kim H, Huh N, Lee D, Jung MW. Distinct roles of rodent orbitofrontal and medial prefrontal cortex in decision making. Neuron. 2010. https://doi.org/10.1016/j.neuron.2010.03.033.
Hirokawa J, Vaughan A, Masset P, Ott T, Kepecs A. Frontal cortex neuron types categorically encode single decision variables. Nature. 2019. https://doi.org/10.1038/s41586-019-1816-9.
Mihindou C, Guillem K, Navailles S, Vouillac C, Ahmed SH. Discriminative inhibitory control of cocaine seeking involves the prelimbic prefrontal cortex. Biol Psychiat. 2013. https://doi.org/10.1016/j.biopsych.2012.08.011.
Capuzzo G, Floresco SB. Prelimbic and infralimbic prefrontal regulation of active and inhibitory avoidance and reward-seeking. J Neurosci. 2020. https://doi.org/10.1523/jneurosci.0414-20.2020.
Acknowledgements
We thank Prof. Nan Sui for guidance.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No.3197070674) and the National Natural Science Foundation of Beijing (Grant No. 7202128) to F.S., and CAS Key Laboratory of Mental Health, Institute of Psychology. The CAS-VPST Silk Road Science Fund 2021 to N.S.(Grant No. GJHZ202129).
Author information
Authors and Affiliations
Contributions
W.D. and F.S. wrote the paper. All authors discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
N/A
Consent to Participate
All authors consent to participate.
Consent for Publication
All authors consent to the publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuroscience & Addiction
Rights and permissions
About this article
Cite this article
Du, W., Liu, Y., Meng, Y. et al. A Translation from Goal-Directed to Habitual Control: the Striatum in Drug Addiction. Curr Addict Rep 8, 530–537 (2021). https://doi.org/10.1007/s40429-021-00392-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40429-021-00392-6