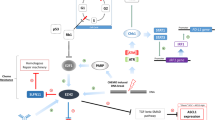
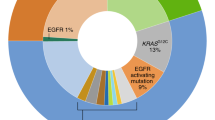
Abstract
Small-cell lung cancer has defied our scientific community for decades. Chemotherapy has been the mainstay treatment for small-cell lung cancer (SCLC) and unlike its counterpart, non-small cell lung cancer, no significant therapeutic breakthroughs have been made since the 1970s. Among the reasons for this slow-paced therapeutic development, one that stands out is the distinctive and almost universal loss of function of the tumour suppressor genes TP53 and RB1 in this disease, for which pharmacological activation has yet to be achieved, despite having been highly sought after. Although no molecularly targeted approach has been approved for clinical practice thus far, several strategies are currently exploring the potential to drug the tumour’s “Achilles heel” that stems from essential pathways regulating DNA-damage response. Most recently, we have witnessed newfound reasons to hope, as the combination of immunotherapy and systemic chemotherapy has improved survival outcomes, representing the first landmark achievement in decades and a new standard of care for patients with extensive disease SCLC. However, continuous efforts are still needed towards a better understanding of the molecular pathways that singularise this tumour to eventually identify the predictive biomarkers that might result in the development of a more rational therapeutic approach, including the use of immunotherapy combinations. In this review we aim to uncover critical aspects of the immune microenvironment and biology of SCLC and provide an overview of the current and future landscape of promising therapeutic opportunities. The challenge still stands, but regardless, we are living in exciting times to finally check SCLC off the ”bucket list” of our scientific community.


Similar content being viewed by others
References
Dayen C, Debieuvre D, Molinier O, Raffy O, Paganin F, Virally J, et al. New insights into stage and prognosis in small cell lung cancer: an analysis of 968 cases. J Thorac Dis. 2017;9(12):5101–11.
American Cancer Society. Lung Cancer Survival Rates. https://www.cancer.org/cancer/lung-cancer/detection-diagnosis-staging/survival-rates.html. Accessed 16 Nov 2019.
Travis WD, Brambilla E, Nicholson AG, Yatabe Y, Austin JHM, Beasley MB, et al. The 2015 World Health Organization classification of lung tumours: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol. 2015;10(9):1243–60.
Thunnissen E, Borczuk AC, Flieder DB, Witte B, Beasley MB, Chung JH, et al. The use of immunohistochemistry improves the diagnosis of small cell lung cancer and its differential diagnosis. An international reproducibility study in a demanding set of cases. J Thorac Oncol. 2017;12(2):334–46.
Govindan R, Page N, Morgensztern D, Read W, Tierney R, Vlahiotis A, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol. 2006;24(28):4539–44.
Rossi A, Di Maio M, Chiodini P, Rudd RM, Okamoto H, Skarlos DV, et al. Carboplatin- or cisplatin-based chemotherapy in first-line treatment of small-cell lung cancer: the COCIS meta-analysis of individual patient data. J Clin Oncol. 2012;30(14):1692–8.
von Pawel J, Schiller JH, Shepherd FA, Fields SZ, Kleisbauer JP, Chrysson NG, et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol. 1999;17(2):658–67.
O’Brien MER, Ciuleanu T-E, Tsekov H, Shparyk Y, Čučeviá B, Juhasz G, et al. Phase III trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer. J Clin Oncol. 2006;24(34):5441–7.
Baize N, Monnet I, Greillier L, Quere G, Kerjouan M, Janicot H, et al. Second-line treatments of small-cell lung cancers. Expert Rev Anticancer Ther. 2017;17(11):1033–43.
von Pawel J, Jotte R, Spigel DR, O’Brien MER, Socinski MA, Mezger J, et al. Randomized phase III trial of amrubicin versus topotecan as second-line treatment for patients with small-cell lung cancer. J Clin Oncol. 2014;32(35):4012–9.
Fruh M, De Ruysscher D, Popat S, Crino L, Peters S, Felip E, et al. Small-cell lung cancer (SCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24(Suppl 6):vi99–105.
Slotman BJ, van Tinteren H, Praag JO, Knegjens JL, El Sharouni SY, Hatton M, et al. Use of thoracic radiotherapy for extensive stage small-cell lung cancer: a phase 3 randomised controlled trial. Lancet. 2015;385(9962):36–42.
Slotman B, Faivre-Finn C, Kramer G, Rankin E, Snee M, Hatton M, et al. Prophylactic cranial irradiation in extensive small-cell lung cancer. N Engl J Med. 2007;357(7):664–72.
Takahashi T, Yamanaka T, Seto T, Harada H, Nokihara H, Saka H, et al. Prophylactic cranial irradiation versus observation in patients with extensive-disease small-cell lung cancer: a multicentre, randomised, open-label, Phase 3 trial. Lancet Oncol. 2017;18(5):663–71.
Mamdani H, Induru R, Jalal SI. Novel therapies in small cell lung cancer. Transl Lung Cancer Res. 2015;4(5):533–44.
Peifer M, Fernández-Cuesta L, Sos ML, George J, Seidel D, Kasper LH, et al. Integrative genome analyses identify key somatic driver mutations of small-cell lung cancer. Nat Genet. 2012;44(10):1104–10.
George J, Lim JS, Jang SJ, Cun Y, Ozretić L, Kong G, et al. Comprehensive genomic profiles of small cell lung cancer. Nature. 2015;524(7563):47–53.
Graus F, Dalmou J, Reñé R, Tora M, Malats N, Verschuuren JJ, et al. Anti-Hu antibodies in patients with small-cell lung cancer: association with complete response to therapy and improved survival. J Clin Oncol. 1997;15(8):2866–72.
Arriola E, Wheater M, Galea I, Cross N, Maishman T, Hamid D, et al. Outcome and biomarker analysis from a multicenter phase 2 study of ipilimumab in combination with carboplatin and etoposide as first-line therapy for extensive-stage SCLC. J Thorac Oncol. 2016;11(9):1511–21.
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Small Cell Lung Cancer. Version 2.2020. https://www.nccn.org. Accessed 16 Nov 2019.
Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SAJR, Behjati S, Biankin AV, et al. Signatures of mutational processes in human cancer. Nature. 2013;500:415.
Lee JK, Lee J, Kim S, Kim S, Youk J, Park S, et al. Clonal history and genetic predictors of transformation into small-cell carcinomas from lung adenocarcinomas. J Clin Oncol. 2017;35(26):3065–74.
Rudin CM, Durinck S, Stawiski EW, Poirier JT, Modrusan Z, Shames DS, et al. Comprehensive genomic analysis identifies SOX2 as a frequently amplified gene in small-cell lung cancer. Nat Genet. 2012;44(10):1111–6.
Meuwissen R, Linn SC, Linnoila RI, Zevenhoven J, Mooi WJ, Berns A. Induction of small cell lung cancer by somatic inactivation of both Trp53 and Rb1 in a conditional mouse model. Cancer Cell. 2003;4(3):181–9.
Park JW, Lee JK, Sheu KM, Wang L, Balanis NG, Nguyen K, et al. Reprogramming normal human epithelial tissues to a common, lethal neuroendocrine cancer lineage. Science. 2018;362(6410):91–5.
Chen HJ, Poran A, Unni AM, Huang SX, Elemento O, Snoeck HW, et al. Generation of pulmonary neuroendocrine cells and SCLC-like tumours from human embryonic stem cells. J Exp Med. 2019;216(3):674–87.
Jia D, Augert A, Kim DW, Eastwood E, Wu N, Ibrahim AH, et al. Crebbp loss drives small cell lung cancer and increases sensitivity to HDAC inhibition. Cancer Discov. 2018;8(11):1422–37.
Zehir A, Benayed R, Shah RH, Syed A, Middha S, Kim HR, et al. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat Med. 2017;23(6):703–13.
Schultheis AM, Bos M, Schmitz K, Wilsberg L, Binot E, Wolf J, et al. Fibroblast growth factor receptor 1 (FGFR1) amplification is a potential therapeutic target in small-cell lung cancer. Mod Pathol. 2014;27(2):214–21.
Thomas A, Lee JH, Abdullaev Z, Park KS, Pineda M, Saidkhodjaeva L, et al. Characterization of fibroblast growth factor receptor 1 in small-cell lung cancer. J Thorac Oncol. 2014;9(4):567–71.
Borges M, Linnoila RI, van de Velde HJ, Chen H, Nelkin BD, Mabry M, et al. An achaete-scute homologue essential for neuroendocrine differentiation in the lung. Nature. 1997;386(6627):852–5.
Ito T, Udaka N, Yazawa T, Okudela K, Hayashi H, Sudo T, et al. Basic helix-loop-helix transcription factors regulate the neuroendocrine differentiation of fetal mouse pulmonary epithelium. Development. 2000;127(18):3913–21.
Fujino K, Motooka Y, Hassan WA, Ali Abdalla MO, Sato Y, Kudoh S, et al. Insulinoma-associated protein 1 is a crucial regulator of neuroendocrine differentiation in lung cancer. Am J Pathol. 2015;185(12):3164–77.
Rudin CM, Pietanza MC, Bauer TM, Ready N, Morgensztern D, Glisson BS, et al. Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: a first-in-human, first-in-class, open-label, Phase 1 study. Lancet Oncol. 2017;18(1):42–51.
Tanaka K, Isse K, Fujihira T, Takenoyama M, Saunders L, Bheddah S, et al. Prevalence of Delta-like protein 3 expression in patients with small cell lung cancer. Lung Cancer. 2018;115:116–20.
Furuta M, Sakakibara-Konishi J, Kikuchi H, Yokouchi H, Nishihara H, Minemura H, et al. Analysis of DLL3 and ASCL1 in surgically resected small cell lung cancer (HOT1702). Oncologist. 2019. https://doi.org/10.1634/theoncologist.2018-0676.
Giroux-Leprieur E, Costantini A, Ding VW, He B. Hedgehog signaling in lung cancer: from oncogenesis to cancer treatment resistance. Int J Mol Sci. 2018;19(9):E2835.
Byers LA, Wang J, Nilsson MB, Fujimoto J, Saintigny P, Yordy J, et al. Proteomic profiling identifies dysregulated pathways in small cell lung cancer and novel therapeutic targets including PARP1. Cancer Discov. 2012;2(9):798–811.
Poirier JT, Gardner EE, Connis N, Moreira AL, de Stanchina E, Hann CL, et al. DNA methylation in small cell lung cancer defines distinct disease subtypes and correlates with high expression of EZH2. Oncogene. 2015;34(48):5869–78.
Sabari JK, Lok BH, Laird JH, Poirier JT, Rudin CM. Unravelling the biology of SCLC: implications for therapy. Nat Rev Clin Oncol. 2017;14(9):549–61.
Kalari S, Jung M, Kernstine KH, Takahashi T, Pfeifer GP. The DNA methylation landscape of small cell lung cancer suggests a differentiation defect of neuroendocrine cells. Oncogene. 2013;32(30):3559–68.
Carney DN, Gazdar AF, Bepler G, Guccion JG, Marangos PJ, Moody TW, et al. Establishment and identification of small cell lung cancer cell lines having classic and variant features. Cancer Res. 1985;45(6):2913–23.
Gazdar AF, Carney DN, Nau MM, Minna JD. Characterization of variant subclasses of cell lines derived from small cell lung cancer having distinctive biochemical, morphological, and growth properties. Cancer Res. 1985;45(6):2924–30.
Poirier JT, Dobromilskaya I, Moriarty WF, Peacock CD, Hann CL, Rudin CM. Selective tropism of Seneca Valley virus for variant subtype small cell lung cancer. J Natl Cancer Inst. 2013;105(14):1059–65.
Borromeo MD, Savage TK, Kollipara RK, He M, Augustyn A, Osborne JK, et al. ASCL1 and NEUROD1 reveal heterogeneity in pulmonary neuroendocrine tumours and regulate distinct genetic programs. Cell Rep. 2016;16(5):1259–72.
Mollaoglu G, Guthrie MR, Böhm S, Brägelmann J, Can I, Ballieu PM, et al. MYC drives progression of small cell lung cancer to a variant neuroendocrine subtype with vulnerability to aurora kinase inhibition. Cancer Cell. 2017;31(2):270–85.
Huang YH, Klingbeil O, He XY, Wu XS, Arun G, Lu B, et al. POU2F3 is a master regulator of a tuft cell-like variant of small cell lung cancer. Genes Dev. 2018;32(13–14):915–28.
McColl K, Wildey G, Sakre N, Lipka MB, Behtaj M, Kresak A, et al. Reciprocal expression of INSM1 and YAP1 defines subgroups in small cell lung cancer. Oncotarget. 2017;8(43):73745–56.
Rudin CM, Poirier JT, Byers LA, Dive C, Dowlati A, George J, et al. Molecular subtypes of small cell lung cancer: a synthesis of human and mouse model data. Nat Rev Cancer. 2019;19(5):289–97.
Cardnell RJ, Li L, Sen T, Bara R, Tong P, Fujimoto J, et al. Protein expression of TTF1 and cMYC define distinct molecular subgroups of small cell lung cancer with unique vulnerabilities to aurora kinase inhibition, DLL3 targeting, and other targeted therapies. Oncotarget. 2017;8(43):73419–32.
Schalper KA, Carvajal-Hausdorf DE, McLaughlin JF, Altan M, Chiang AC, Velcheti V, et al. Objective measurement and significance of PD-L1, B7-H3, B7-H4 and TILs in small cell lung cancer (SCLC). J Clin Oncol. 2016;34(15 suppl):8566.
Busch SE, Hanke ML, Kargl J, Metz HE, MacPherson D, Houghton AM. Lung cancer subtypes generate unique immune responses. J Immunol. 2016;197(11):4493–503.
Doyle A, Martin WJ, Funa K, Gazdar A, Carney D, Martin SE, et al. Markedly decreased expression of class I histocompatibility antigens, protein, and mRNA in human small-cell lung cancer. J Exp Med. 1985;161(5):1135–51.
He Y, Rozeboom L, Rivard CJ, Ellison K, Dziadziuszko R, Yu H, et al. MHC class II expression in lung cancer. Lung Cancer. 2017;112:75–80.
Olugbile SO, Bao R, Sweis RF, Hensing TA, Nakamura Y, Gajewski T, et al. Molecular characterization of immune exclusion in small-cell lung cancer. J Clin Oncol. 2016;34(15 suppl):8565.
Willingham SB, Volkmer J-P, Gentles AJ, Sahoo D, Dalerba P, Mitra SS, et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumours. Proc Natl Acad Sci USA. 2012;109(17):6662–7.
Weiskopf K, Jahchan NS, Schnorr PJ, Cristea S, Ring AM, Maute RL, et al. CD47-blocking immunotherapies stimulate macrophage-mediated destruction of small-cell lung cancer. J Clin Investig. 2016;126(7):2610–20.
Rivalland G, Walkiewicz M, Wright GM, John T. Small cell lung cancer: the immune microenvironment and prognostic impact of checkpoint expression. J Clin Oncol. 2017;35(15 suppl):8569.
Koyama K, Kagamu H, Miura S, Hiura T, Miyabayashi T, Itoh R, et al. Reciprocal CD4+ T-cell balance of effector CD62Llow CD4+ and CD62LhighCD25+ CD4+ regulatory T cells in small cell lung cancer reflects disease stage. Clin Cancer Res. 2008;14(21):6770–9.
Wang W, Hodkinson P, McLaren F, Mackean MJ, Williams L, Howie SEM, et al. Histologic assessment of tumour-associated CD45(+) cell numbers is an independent predictor of prognosis in small cell lung cancer. Chest. 2013;143(1):146–51.
Ishii H, Azuma K, Kawahara A, Yamada K, Imamura Y, Tokito T, et al. Significance of programmed cell death-ligand 1 expression and its association with survival in patients with small cell lung cancer. J Thorac Oncol. 2015;10(3):426–30.
Kim HS, Lee JH, Nam SJ, Ock C-Y, Moon J-W, Yoo CW, et al. Association of PD-L1 expression with tumour-infiltrating immune cells and mutation burden in high-grade neuroendocrine carcinoma of the lung. J Thorac Oncol. 2018;13(5):636–48.
Calles A, Aguado G, Sandoval C, Alvarez R. The role of immunotherapy in small cell lung cancer. Clin Transl Oncol. 2019;21(8):961–76.
Reck M, Bondarenko I, Luft A, Serwatowski P, Barlesi F, Chacko R, et al. Ipilimumab in combination with paclitaxel and carboplatin as first-line therapy in extensive-disease-small-cell lung cancer: results from a randomized, double-blind, multicenter Phase 2 trial. Ann Oncol. 2013;24(1):75–83.
Reck M, Luft A, Szczesna A, Havel L, Kim SW, Akerley W, et al. Phase III randomized trial of ipilimumab plus etoposide and platinum versus placebo plus etoposide and platinum in extensive-stage small-cell lung cancer. J Clin Oncol. 2016;34(31):3740–8.
Horn L, Mansfield AS, Szczesna A, Havel L, Krzakowski M, Hochmair MJ, et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N Engl J Med. 2018;379(23):2220–9.
Reck M, Liu SV, Mansfield AS, Mok TSK, Scherpereel A, Reinmuth N, et al. 1736OIMpower133: updated overall survival (OS) analysis of first-line (1L) atezolizumab (atezo) + carboplatin + etoposide in extensive-stage SCLC (ES-SCLC). Ann Oncol. 2019;30(suppl 5).
Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a Phase 3, open-label, multicentre randomised controlled trial. Lancet. 2017;389(10066):255–65.
FDA approves atezolizumab for extensive-stage small cell lung cancer. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-atezolizumab-extensive-stage-small-cell-lung-cancer. Accessed 16 Nov 2019.
Tecentriq—European Medicines Agency—Europa EU. https://www.ema.europa.eu/en/medicines/human/summaries-opinion/tecentriq-1. Accessed 16 Nov 2019.
Paz-Ares L, Dvorkin M, Chen Y, Reinmuth N, Hotta K, Trukhin D, et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, Phase 3 trial. Lancet. 2019. https://doi.org/10.1016/s0140-6736(19)32222-6.
Owonikoko TK, Kim HR, Govindan R, Ready N, Reck M, Peters S, et al. LBA1_PRNivolumab (nivo) plus ipilimumab (ipi), nivo, or placebo (pbo) as maintenance therapy in patients (pts) with extensive disease small cell lung cancer (ED-SCLC) after first-line (1L) platinum-based chemotherapy (chemo): Results from the double-blind, randomized Phase III CheckMate 451 study. Ann Oncol. 2019;30(suppl 2).
Rudin CM, Shen L, Pietanza MC. 1535TiP KEYNOTE-604: Phase 3 trial of pembrolizumab plus etoposide/platinum (EP) for first-line treatment of extensive stage small-cell lung cancer (ES-SCLC). Ann Oncol. 2017;28(suppl 5).
Gadgeel SM, Pennell NA, Fidler MJ, Halmos B, Bonomi P, Stevenson J, et al. Phase II study of maintenance pembrolizumab in patients with extensive-stage small cell lung cancer (SCLC). J Thorac Oncol. 2018;13(9):1393–9.
Thomas M, Ponce-Aix S, Navarro A, Riera-Knorrenschild J, Schmidt M, Wiegert E, et al. Immunotherapeutic maintenance treatment with toll-like receptor 9 agonist lefitolimod in patients with extensive-stage small-cell lung cancer: results from the exploratory, controlled, randomized, international Phase II IMPULSE study. Ann Oncol. 2018;29(10):2076–84.
Gandhi L, Rodriguez-Abreu D, Gadgeel S, Esteban E, Felip E, De Angelis F, et al. Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med. 2018;378(22):2078–92.
Paz-Ares L, Luft A, Vicente D, Tafreshi A, Gumus M, Mazieres J, et al. Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med. 2018;379(21):2040–51.
De Ruysscher D, Pujol JL, Popat S, Reck M, Le Pechoux C, Liston A, et al. STIMULI: A randomised open-label Phase II trial of consolidation with nivolumab and ipilimumab in limited-stage SCLC after standard of care chemo-radiotherapy conducted by ETOP and IFCT. Ann Oncol. 2016;27(suppl 6).
Senan S, Shire N, Mak G, Yao W, Jiang H. 67TiP ADRIATIC: A Phase III trial of durvalumab ± tremelimumab after concurrent chemoradiation for patients with limited stage small cell lung cancer. Ann Oncol. 2019;30(suppl 2).
Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379(24):2342–50.
Antonia SJ, Lopez-Martin JA, Bendell J, Ott PA, Taylor M, Eder JP, et al. Nivolumab alone and nivolumab plus ipilimumab in recurrent small-cell lung cancer (CheckMate 032): a multicentre, open-label, Phase 1/2 trial. Lancet Oncol. 2016;17(7):883–95.
Hellmann MD, Ott PA, Zugazagoitia J, Ready NE, Hann CL, Braud FGD, et al. Nivolumab (nivo) ± ipilimumab (ipi) in advanced small-cell lung cancer (SCLC): First report of a randomized expansion cohort from CheckMate 032. J Clin Oncol. 2017;35(15_suppl):8503.
Ready N, Farago AF, de Braud F, Atmaca A, Hellmann MD, Schneider JG, et al. Third-line nivolumab monotherapy in recurrent SCLC: CheckMate 032. J Thorac Oncol. 2019;14(2):237–44.
FDA grants nivolumab accelerated approval for third-line treatment of metastatic small cell lung cancer. https://www.fdagov/drugs/resources-information-approved-drugs/fda-grants-nivolumab-accelerated-approval-third-line-treatment-metastatic-small-cell-lung-cancer. Accessed 16 Nov 2019.
Reck M, Vicente D, Ciuleanu T, Gettinger S, Peters S, Horn L, et al. LBA5 Efficacy and safety of nivolumab (nivo) monotherapy versus chemotherapy (chemo) in recurrent small cell lung cancer (SCLC): Results from CheckMate 331. Ann Oncol. 2018;29(suppl 10).
Ott PA, Elez E, Hiret S, Kim DW, Morosky A, Saraf S, et al. Pembrolizumab in patients with extensive-stage small-cell lung cancer: results from the phase Ib KEYNOTE-028 study. J Clin Oncol. 2017;35(34):3823–9.
Chung HC, Lopez-Martin JA, Kao SC-H, Miller WH, Ros W, Gao B, et al. Phase 2 study of pembrolizumab in advanced small-cell lung cancer (SCLC): KEYNOTE-158. J Clin Oncol. 2018;36(15 suppl):8506.
Chung HC, Piha-Paul SA, Lopez-Martin J, Schellens JHM, Kao S, Miller WH, et al. Pembrolizumab after two or more lines of prior therapy in patients with advanced small-cell lung cancer (SCLC): results from the KEYNOTE-028 and KEYNOTE-158 studies. Cancer Res. 2019;79(13 Suppl):CT073.
MERCK. FDA Grants Priority Review to Merck’s Supplemental Biologics License Application for KEYTRUDA® (pembrolizumab) Monotherapy for Third-Line Treatment of Patients with Advanced Small Cell Lung Cancer (SCLC). https://investors.merck.com/news/press-release-details/2019/FDA-Grants-Priority-Review-to-Mercks-Supplemental-Biologics-License-Application-for-KEYTRUDA-pembrolizumab-Monotherapy-for-Third-Line-Treatment-of-Patients-with-Advanced-Small-Cell-Lung-Cancer-SCLC/default.aspx . Accessed 16 July 2019.
Sequist LV, Chiang A, Gilbert J, Gordon M, Conkling PR, Thompson D, et al. Clinical activity, safety and predictive biomarkers results from a Phase Ia atezolizumab (atezo) trial in extensive-stage small cell lung cancer (ES-SCLC). Ann Oncol. 2016;27(suppl 6).
Pujol JL, Greillier L, Audigier-Valette C, Moro-Sibilot D, Uwer L, Hureaux J, et al. A randomized non-comparative phase II study of anti-programmed cell death-ligand 1 atezolizumab or chemotherapy as second-line therapy in patients with small cell lung cancer: results from the IFCT-1603 trial. J Thorac Oncol. 2019;14(5):903–13.
Goldman JW, Dowlati A, Antonia SJ, Nemunaitis JJ, Butler MO, Segal NH, et al. Safety and antitumour activity of durvalumab monotherapy in patients with pretreated extensive disease small-cell lung cancer (ED-SCLC). J Clin Oncol. 2018;36(15 suppl):8518.
Bondarenko I, Juan-Vidal O, Pajkos G, Kryzhanivska A, Pápai Székely Z, Vicente D, et al. 1665PD Preliminary efficacy of durvalumab plus tremelimumab in platinum-refractory/resistant ED-SCLC from arm A of the Phase II BALTIC study. Ann Oncol. 2018;29(suppl 8).
Pavan A, Attili I, Pasello G, Guarneri V, Conte PF, Bonanno L. Immunotherapy in small-cell lung cancer: from molecular promises to clinical challenges. J Immunother Cancer. 2019;7(1):205.
Schultheis AM, Scheel AH, Ozretić L, George J, Thomas RK, Hagemann T, et al. PD-L1 expression in small cell neuroendocrine carcinomas. Eur J Cancer. 2015;51(3):421–6.
Bonanno L, Pavan A, Dieci MV, Di Liso E, Schiavon M, Comacchio G, et al. The role of immune microenvironment in small-cell lung cancer: distribution of PD-L1 expression and prognostic role of FOXP3-positive tumour infiltrating lymphocytes. Eur J Cancer. 2018;101:191–200.
Paz-Ares L, Goldman JW, Garassino MC, Dvorkin M, Trukhin D, Statsenko G, et al. LBA89PD-L1 expression, patterns of progression and patient-reported outcomes (PROs) with durvalumab plus platinum-etoposide in ES-SCLC: results from CASPIAN. Ann Oncol. 2019;30(Suppl_5).
Lin H, Wei S, Hurt EM, Green MD, Zhao L, Vatan L, et al. Host expression of PD-L1 determines efficacy of PD-L1 pathway blockade-mediated tumour regression. J Clin Investig. 2018;128(2):805–15.
Tang H, Liang Y, Anders RA, Taube JM, Qiu X, Mulgaonkar A, et al. PD-L1 on host cells is essential for PD-L1 blockade-mediated tumour regression. J Clin Investig. 2018;128(2):580–8.
Paz-Ares LG, Jiang H, Huang Y, Dennis PA. A Phase 3, randomized study of first-line durvalumab (D) ± tremelimumab (T) + platinum-based chemotherapy (CT) vs CT alone in extensive disease small-cell lung cancer (ED-SCLC): Caspian. J Clin Oncol. 2017;35(15 suppl):TPS8586.
Goodman AM, Kato S, Bazhenova L, Patel SP, Frampton GM, Miller V, et al. Tumour mutational burden as an independent predictor of response to immunotherapy in diverse cancers. Mol Cancer Ther. 2017;16(11):2598–608.
Wagner AH, Devarakonda S, Skidmore ZL, Krysiak K, Ramu A, Trani L, et al. Recurrent WNT pathway alterations are frequent in relapsed small cell lung cancer. Nat Commun. 2018;9(1):3787.
Hellmann MD, Callahan MK, Awad MM, Calvo E, Ascierto PA, Atmaca A, et al. Tumour Mutational Burden and Efficacy of Nivolumab Monotherapy and in Combination with Ipilimumab in Small-Cell Lung Cancer. Cancer Cell. 2018;33(5):853–61 e4.
Squibb B-M. Bristol-Myers Squibb Provides Update on the Ongoing Regulatory Review of Opdivo Plus Low-Dose Yervoy in First-Line Lung Cancer Patients with Tumour Mutational Burden ≥ 10 mut/Mb. https://news.bms.com/press-release/corporatefinancial-news/bristol-myers-squibb-provides-update-ongoing-regulatory-review . Accessed 16 July 2019.
Hardy-Werbin M, Arpi O, Taus A, Rocha P, Joseph-Pietras D, Nolan L, et al. Assessment of neuronal autoantibodies in patients with small cell lung cancer treated with chemotherapy with or without ipilimumab. Oncoimmunology. 2018;7(2):e1395125.
Krug LM, Ragupathi G, Hood C, George C, Hong F, Shen R, et al. Immunization with N-propionyl polysialic acid-KLH conjugate in patients with small cell lung cancer is safe and induces IgM antibodies reactive with SCLC cells and bactericidal against group B meningococci. Cancer Immunol Immunother. 2012;61(1):9–18.
Brezicka FT, Olling S, Bergman B, Berggren H, Engström CP, Hammarström S, et al. Coexpression of ganglioside antigen Fuc-GM1, neural-cell adhesion molecule, carcinoembryonic antigen, and carbohydrate tumour-associated antigen CA 50 in lung cancer. Tumour Biol. 1992;13(5–6):308–15.
Ponath P, Menezes D, Pan C, Chen B, Oyasu M, Strachan D, et al. A novel, fully human anti-fucosyl-GM1 antibody demonstrates potent in vitro and in vivo antitumour activity in preclinical models of small cell lung cancer. Clin Cancer Res. 2018;24(20):5178–89.
Chu QSC, van Herpen C, Leighl NB, Markman B, Clarke S, Juergens RA, et al. 1528PD Initial results of BMS-986012, a first-in-class fucosyl-GM1 mAb, in combination with nivolumab, in pts with relapsed/refractory (rel/ref) small-cell lung cancer (SCLC). Ann Oncol. 2017;28(suppl 5).
Yoshida S, Fukumoto S, Kawaguchi H, Sato S, Ueda R, Furukawa K. Ganglioside G(D2) in small cell lung cancer cell lines: enhancement of cell proliferation and mediation of apoptosis. Cancer Res. 2001;61(10):4244–52.
Ahmed M, Cheung NKV. Engineering anti-GD2 monoclonal antibodies for cancer immunotherapy. FEBS Lett. 2014;588(2):288–97.
Mueller BM, Romerdahl CA, Gillies SD, Reisfeld RA. Enhancement of antibody-dependent cytotoxicity with a chimeric anti-GD2 antibody. J Immunol. 1990;144(4):1382–6.
Yaghoubi S, Harrison T, Messerschmidt G, Corritori S. Abstract 543: Dinutuximab binds specifically to disialoganglioside-2. Cancer Res. 2019;79(13 Supplement):543.
Edelman MJ, Juan O, Navarro A, Golden G, Borg E, Saunders AV. A two-part, open-label, randomized, Phase 2/3 study of dinutuximab and irinotecan versus irinotecan for second-line treatment of subjects with relapsed or refractory small cell lung cancer. J Clin Oncol. 2018;36(15 suppl):TPS8588.
Edelman MJ, Juan O, Navarro A, Golden G, Saunders A. 88P Feasibility of outpatient dinutuximab (D) and irinotecan (I) for second-line treatment of relapsed or refractory small cell lung cancer (RR SCLC): part 1 of an open-label, randomized, Phase 2/3 study. J Thorac Oncol. 2018;13(4):S48–9.
Straathof K, Flutter B, Wallace R, Thomas S, Cheung G, Collura A, et al. Abstract CT145: A Cancer Research UK Phase I trial of anti-GD2 chimeric antigen receptor (CAR) transduced T-cells (1RG-CART) in patients with relapsed or refractory neuroblastoma. Cancer Res. 2018;78(13 Suppl):CT145.
Mount CW, Majzner RG, Sundaresh S, Arnold EP, Kadapakkam M, Haile S, et al. Potent antitumour efficacy of anti-GD2 CAR T cells in H3-K27M + diffuse midline gliomas. Nat Med. 2018;24(5):572–9.
Farago A, Isse K, Drapkin B, Kamesan V, Kem M, Saunders L, et al. Dynamics of DLL3 and ASCL1 expression in SCLC over disease course. J Thorac Oncol. 2018;13(10):S970–1.
Gerratana B. Biosynthesis, synthesis, and biological activities of pyrrolobenzodiazepines. Med Res Rev. 2012;32(2):254–93.
Saunders LR, Bankovich AJ, Anderson WC, Aujay MA, Bheddah S, Black K, et al. A DLL3-targeted antibody-drug conjugate eradicates high-grade pulmonary neuroendocrine tumour-initiating cells in vivo. Sci Transl Med. 2015;7(302):302ra136.
Morgensztern D, Besse B, Greillier L, Santana-Davila R, Ready N, Hann CL, et al. Efficacy and safety of rovalpituzumab tesirine in third-line and beyond patients with DLL3-expressing, relapsed/refractory small-cell lung cancer: results from the phase II TRINITY study. Clin Cancer Res. 2019. https://doi.org/10.1158/1078-0432.ccr-19-1133.
AbbVie. Phase 3 Trial of Rova-T as Second-line Therapy for Advanced Small-Cell Lung Cancer (TAHOE Study) Halted. https://news.abbvie.com/news/Phase-3-trial-rova-t-as-second-line-therapy-for-advanced-small-cell-lung-cancer-tahoe-study-halted.htm. Accessed 16 July 2019.
AbbVie Discontinues Rovalpituzumab Tesirine (Rova-T) Research and Development Program. https://news.abbvie.com/news/press-releases/abbvie-discontinues-rovalpituzumab-tesirine-rova-t-research-and-development-program.htm. Accessed 16 Nov 2019.
Mullard A. Cancer stem cell candidate Rova-T discontinued. Nat Rev Drug Discov. 2019;18(11):814.
Baeuerle PA, Reinhardt C. Bispecific T-cell engaging antibodies for cancer therapy. Cancer Res. 2009;69(12):4941–4.
Giffin M, Cooke K, Lobenhofer E, Friedrich M, Raum T, Coxon A. Targeting DLL3 with AMG 757, a BiTE® Antibody Construct, and AMG 119, a CAR-T, for the Treatment of SCLC. J Thorac Oncol. 2018;13(10):S971.
Giffin MJ, Lobenhofer EK, Cooke K, Raum T, Stevens J, Beltran PJ, et al. Abstract 3632: BiTE® antibody constructs for the treatment of SCLC. Cancer Res. 2017;77(13 Supplement):3632.
Smit M-AD, Borghaei H, Owonikoko TK, Hummel H-D, Johnson ML, Champiat S, et al. Phase 1 study of AMG 757, a half-life extended bispecific T cell engager (BiTE) antibody construct targeting DLL3, in patients with small cell lung cancer (SCLC). J Clin Oncol. 2019;37(15 suppl):TPS8577.
Byers LA, Chiappori A, Smit M-AD. Phase 1 study of AMG 119, a chimeric antigen receptor (CAR) T cell therapy targeting DLL3, in patients with relapsed/refractory small cell lung cancer (SCLC). J Clin Oncol. 2019;37(15 suppl):TPS8576.
European Medicines Agengy. EU/3/19/2143. https://www.ema.europa.eu/en/medicines/human/orphan-designations/eu3192143 . Accessed 16 July 2019.
Belgiovine C, Bello E, Liguori M, Craparotta I, Mannarino L, Paracchini L, et al. Lurbinectedin reduces tumour-associated macrophages and the inflammatory tumour microenvironment in preclinical models. Br J Cancer. 2017;117:628.
Harlow ML, Maloney N, Roland J, Guillen Navarro MJ, Easton MK, Kitchen-Goosen SM, et al. Lurbinectedin inactivates the ewing sarcoma oncoprotein EWS-FLI1 by redistributing it within the nucleus. Cancer Res. 2016;76(22):6657–68.
Santamaria Nunez G, Robles CM, Giraudon C, Martinez-Leal JF, Compe E, Coin F, et al. Lurbinectedin specifically triggers the degradation of phosphorylated RNA polymerase II and the formation of DNA breaks in cancer cells. Mol Cancer Ther. 2016;15(10):2399–412.
Elez ME, Tabernero J, Geary D, Macarulla T, Kang SP, Kahatt C, et al. First-in-human phase i study of lurbinectedin (PM01183) in patients with advanced solid tumours. Clin Cancer Res. 2014;20(8):2205–14.
Paz-Ares LG, Trigo Perez JM, Besse B, Moreno V, Lopez R, Sala MA, et al. Efficacy and safety profile of lurbinectedin in second-line SCLC patients: Results from a Phase II single-agent trial. J Clin Oncol. 2019;37(15 suppl):8506.
Calvo E, Moreno V, Flynn M, Holgado E, Olmedo ME, Lopez Criado MP, et al. Antitumour activity of lurbinectedin (PM01183) and doxorubicin in relapsed small-cell lung cancer: results from a Phase I study. Ann Oncol. 2017;28(10):2559–66.
Forster M, Moreno V, Calvo E, Olmedo ME, Lopez-Criado MP, Lopez-Vilariño J, et al. P1.12-20 Overall survival with lurbinectedin plus doxorubicin in relapsed SCLC. Results from an expansion cohort of a phase Ib trial. J Thorac Oncol. 2018;13(10):S581.
Farago AF, Paz-Ares LG, Ciuleanu T-E, Fülop A, Navarro A, Bonanno L, et al. ATLANTIS: global, randomized Phase III study of lurbinectedin (L) with doxorubicin (DOX) vs CAV or topotecan (T) in small-cell lung cancer after platinum therapy. J Clin Oncol. 2018;36(15 suppl):TPS8587.
de Bono J, Ramanathan RK, Mina L, Chugh R, Glaspy J, Rafii S, et al. Phase I, dose-escalation, two-part trial of the PARP inhibitor talazoparib in patients with advanced germline BRCA1/2 mutations and selected sporadic cancers. Cancer Discov. 2017;7(6):620–9.
Woll P, Gaunt P, Steele N, Ahmed S, Mulatero C, Shah R, et al. P1.07-015 STOMP: A UK National Cancer Research Network randomised, double blind, multicentre phase II trial of olaparib as maintenance therapy in SCLC: topic: drug treatment alone and in combination with radiotherapy. J Thorac Oncol. 2017;12(1):S704–5.
Lu S, Jiang L, Ai X, Li J, Dong X, Zhang D, et al. P1.12-04 A Ph3 study of niraparib as maintenance therapy in 1L platinum responsive extensive disease small cell lung cancer patients. J Thorac Oncol. 2018;13(10):S574.
Mikule K, Wang Y, Xiao Y, Rocono J, Broudy T, Wilcoxen K. Abstract B169: A mouse avatar tumour maintenance study identified a subset of SCLC patient-derived tumour xenograft models sensitive to the PARP inhibitor niraparib. Mol Cancer Ther. 2015;14(12 Suppl 2):B169.
Lok BH, Gardner EE, Schneeberger VE, Ni A, Desmeules P, Rekhtman N, et al. PARP inhibitor activity correlates with SLFN11 expression and demonstrates synergy with temozolomide in small cell lung cancer. Clin Cancer Res. 2017;23(2):523–35.
Pietanza MC, Waqar SN, Krug LM, Dowlati A, Hann CL, Chiappori A, et al. Randomized, double-blind, phase ii study of temozolomide in combination with either veliparib or placebo in patients with relapsed-sensitive or refractory small-cell lung cancer. J Clin Oncol. 2018;36(23):2386–94.
Farago AF, Drapkin BJ, Charles A, Yeap B, Heist RS, Azzoli CG, et al. Abstract CT048: Phase 1/2 study of olaparib tablets and temozolomide in patients with small cell lung cancer (SCLC) following failure of prior chemotherapy. Cancer Res. 2017;77(13 Suppl):CT048.
Owonikoko TK, Dahlberg SE, Sica GL, Wagner LI, Wade JL, Srkalovic G, et al. Randomized phase II trial of cisplatin and etoposide in combination with veliparib or placebo for extensive-stage small-cell lung cancer: ECOG-ACRIN 2511 study. J Clin Oncol. 2018;37(3):222–9.
Laird JH, Lok BH, Ma J, Bell A, de Stanchina E, Poirier JT, et al. Talazoparib is a potent radiosensitizer in small cell lung cancer cell lines and xenografts. Clin Cancer Res. 2018;24(20):5143–52.
Pantelidou C, Sonzogni O, De Oliveria Taveira M, Mehta AK, Kothari A, Wang D, et al. PARP inhibitor efficacy depends on CD8+ T-cell recruitment via intratumoural STING pathway activation in BRCA-deficient models of triple-negative breast cancer. Cancer Discov. 2019;9(6):722–37.
Sen T, Rodriguez BL, Chen L, Corte CMD, Morikawa N, Fujimoto J, et al. Targeting DNA damage response promotes antitumour immunity through STING-mediated T-cell activation in small cell lung cancer. Cancer Discov. 2019;9(5):646–61.
Wang Z, Sun K, Xiao Y, Feng B, Mikule K, Ma X, et al. Niraparib activates interferon signaling and potentiates anti-PD-1 antibody efficacy in tumour models. Sci Rep. 2019;9(1):1853.
Thomas A, Vilimas R, Trindade C, Erwin-Cohen R, Roper N, Xi L, et al. Durvalumab in combination with olaparib in patients with relapsed SCLC: results from a phase II study. J Thorac Oncol. 2019;14(8):1447–57.
Penson RT, Drew Y, de Jonge MJA, Hong SH, Park YH, Wolfer A, et al. 448TiP MEDIOLA: a phase I/II trial of olaparib (PARP inhibitor) in combination with durvalumab (anti-PD-L1 antibody) in pts with advanced solid tumours—new ovarian cancer cohorts. Ann Oncol. 2018;29(suppl 8).
Lu Y, Liu Y, Jiang J, Xi Z, Zhong N, Shi S, et al. Knocking down the expression of Aurora-A gene inhibits cell proliferation and induces G2/M Phase arrest in human small cell lung cancer cells. Oncol Rep. 2014;32(1):243–9.
Owonikoko TK, Nackaerts K, Csoszi T, Ostoros G, Baik C, Mark Z, et al. Randomized Phase 2 study of investigational aurora A kinase (AAK) inhibitor alisertib (MLN8237) + paclitaxel (P) vs placebo + P as second line therapy for small-cell lung cancer (SCLC). Ann Oncol. 2016;27(suppl 6).
Parker L, Piwnica-Worms H. Inactivation of the p34cdc2-cyclin B complex by the human WEE1 tyrosine kinase. Science. 1992;257(5078):1955–7.
Lallo A, Frese KK, Morrow CJ, Sloane R, Gulati S, Schenk MW, et al. The combination of the PARP inhibitor olaparib and the WEE1 inhibitor AZD1775 as a new therapeutic option for small cell lung cancer. Clin Cancer Res. 2018;24(20):5153–64.
Von Pawel J, Vynnychenko I, Jiang H, Huang Y, Dennis PA. A Phase II, open-label, multi-arm study of novel combinations of immunotherapies or DDR inhibitors in platinum-refractory, extensive disease small-cell lung cancer (ED-SCLC): BALTIC. J Clin Oncol. 2017;35(15 suppl):TPS8585.
Acknowledgements
We thank Kyla M. Juett for improving the use of English in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this manuscript.
Conflict of interest
Dr N. Reguart reports receiving honoraria from AstraZeneca, Novartis, Takeda, Eli Lilly, MSD, Boehringer Ingelheim and Pfizer for consulting, advisory role or speaker’s bureaus; travel and accommodation expenses from Roche, MSD; and research funding from Pfizer and Novartis. C. Teixido reports receiving honoraria from Bristol-Myers Squibb, Pfizer and MSD for advisory role or speaker’s bureaus; travel and accommodation expenses from Roche, MSD; and research funding from Novartis. Jordi Remon reports receiving honoraria from Boehringer Ingelheim, Pfizer, MSD, Bristol-Myers Squibb and AstraZeneca for consulting, advisory role or speaker’s bureaus; travel and accommodation expenses from Bristol-Myers Squibb, OSE-Immunotherapeutics, AstraZeneca and Roche. Elba Marin and Roxana Reyes declare that they have no conflicts of interest that might be relevant to the contents of this manuscript.
Rights and permissions
About this article
Cite this article
Reguart, N., Marin, E., Remon, J. et al. In Search of the Long-Desired ‘Copernican Therapeutic Revolution’ in Small-Cell Lung Cancer. Drugs 80, 241–262 (2020). https://doi.org/10.1007/s40265-019-01240-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-019-01240-8