Abstract
Background
Hydroxychloroquine is an oral drug prescribed to pregnant women with rheumatic disease to reduce disease activity and prevent flares. Physiologic changes during pregnancy may substantially alter drug pharmacokinetics. However, the effect of pregnancy on hydroxychloroquine disposition and the potential need for dose adjustment remains virtually unknown.
Methods
We performed a population-pharmacokinetic analysis using samples from the Duke Autoimmunity in Pregnancy Registry from 2013 to 2016. We measured hydroxychloroquine concentration using high-performance liquid chromatography/tandem mass spectrometry and analyzed data using non-linear mixed-effect modeling. We calculated differences between pregnancy and postpartum empirical Bayesian estimates using paired t tests. We computed steady-state concentration profiles for hydroxychloroquine during pregnancy and postpartum using individual clinical data and empirical Bayesian estimates developed from the final pharmacokinetic model.
Results
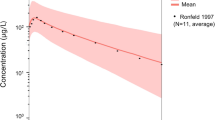
We obtained 145 serum samples from 50 patients, 25 of whom had paired pregnancy and postpartum specimens. Five subjects had average concentrations (pregnancy and postpartum) < 100 ng/mL, consistent with medication non-adherence, and were excluded. The population estimated apparent volume of distribution was 1850 L/70 kg and estimated apparent clearance was 51 L/h. Compared with postpartum, median apparent volume of distribution increased significantly during pregnancy (p < 0.001), whereas apparent clearance and 24-h area under the curve did not change.
Conclusions
We developed a one-compartment population-pharmacokinetic model for hydroxychloroquine in pregnant women with rheumatic disease. Estimates for serum CL were within the expected range for plasma in non-pregnant adults. Because CL and 24-h area under the curve did not change during pregnancy compared with postpartum, our modeling in this small cohort does not support adjusting hydroxychloroquine dose during pregnancy.

Similar content being viewed by others
References
Clowse ME. Lupus activity in pregnancy. Rheum Dis Clin N Am. 2007;33:237–52.
Gotestam Skorpen C, Hoeltzenbein M, Tincani A, Fischer-Betz R, Elefant E, Chambers C, et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann Rheum Dis. 2016;75:795–810.
Clowse ME, Magder L, Witter F, Petri M. Hydroxychloroquine in lupus pregnancy. Arthritis Rheum. 2006;54:3640–7.
Eudy AM, Siega-Riz AM, Engel SM, Franceschini N, Howard AG, Clowse MEB, et al. Effect of pregnancy on disease flares in patients with systemic lupus erythematosus. Ann Rheum Dis. 2018;77:855–60.
Fischer-Betz R, Specker C, Brinks R, Aringer M, Schneider M. Low risk of renal flares and negative outcomes in women with lupus nephritis conceiving after switching from mycophenolate mofetil to azathioprine. Rheumatology (Oxford). 2013;52:1070–6.
Ke AB, Rostami-Hodjegan A, Zhao P, Unadkat JD. Pharmacometrics in pregnancy: an unmet need. Annu Rev Pharmacol Toxicol. 2014;54:53–69.
Abduljalil K, Furness P, Johnson TN, Rostami-Hodjegan A, Soltani H. Anatomical, physiological and metabolic changes with gestational age during normal pregnancy: a database for parameters required in physiologically based pharmacokinetic modelling. Clin Pharmacokinet. 2012;51:365–96.
Rainsford KD, Parke AL, Clifford-Rashotte M, Kean WF. Therapy and pharmacological properties of hydroxychloroquine and chloroquine in treatment of systemic lupus erythematosus, rheumatoid arthritis and related diseases. Inflammopharmacology. 2015;23:231–69.
Browning DJ. Pharmacology of chloroquine and hydroxychloroquine. In: Hydroxychloroquine and chloroquine retinopathy. New York: Springer New York; 2014. pp. 35–63.
NMS Labs. Hydroxychloroquine test summary sheet. http://www.nmslabs.com/tests/Hydroxychloroquine–Serum-Plasma/2362SP. Accessed 6 Feb 2018.
Lim HS, Im JS, Cho JY, Bae KS, Klein TA, Yeom JS, et al. Pharmacokinetics of hydroxychloroquine and its clinical implications in chemoprophylaxis against malaria caused by Plasmodium vivax. Antimicrob Agents Chemother. 2009;53:1468–75.
Morita S, Takahashi T, Yoshida Y, Yokota N. Population pharmacokinetics of hydroxychloroquine in Japanese patients with cutaneous or systemic lupus erythematosus. Ther Drug Monit. 2016;38:259–67.
Costedoat-Chalumeau N, Amoura Z, Hulot JS, Aymard G, Leroux G, Marra D, et al. Very low blood hydroxychloroquine concentration as an objective marker of poor adherence to treatment of systemic lupus erythematosus. Ann Rheum Dis. 2007;66:821–4.
Costedoat-Chalumeau N, Le Guern V, Piette JC. Routine hydroxychloroquine blood concentration measurement in systemic lupus erythematosus reaches adulthood. J Rheumatol. 2015;42:1997–9.
Iudici M, Pantano I, Fasano S, Pierro L, Charlier B, Pingeon M, et al. Health status and concomitant prescription of immunosuppressants are risk factors for hydroxychloroquine non-adherence in systemic lupus patients with prolonged inactive disease. Lupus. 2018;27:265–72.
Carmichael SJ, Charles B, Tett SE. Population pharmacokinetics of hydroxychloroquine in patients with rheumatoid arthritis. Ther Drug Monit. 2003;25:671–81.
Tett SE, Cutler DJ, Day RO, Brown KF. A dose-ranging study of the pharmacokinetics of hydroxy-chloroquine following intravenous administration to healthy volunteers. Br J Clin Pharmacol. 1988;26:303–13.
Jallouli M, Galicier L, Zahr N, Aumaitre O, Frances C, Le Guern V, et al. Determinants of hydroxychloroquine blood concentration variations in systemic lupus erythematosus. Arthritis Rheumatol. 2015;67:2176–84.
Yeon Lee J, Lee J, Ki Kwok S, Hyeon JuJ, Su Park K, Park SH. Factors related to blood hydroxychloroquine concentration in patients with systemic lupus erythematosus. Arthritis Care Res (Hoboken). 2017;69:536–42.
Hebert MF, Zheng S, Hays K, Shen DD, Davis CL, Umans JG, et al. Interpreting tacrolimus concentrations during pregnancy and postpartum. Transplantation. 2013;95:908–15.
Costedoat-Chalumeau N, Houssiau F, Izmirly P, Le Guern V, Navarra S, Jolly M, et al. A prospective international study on adherence to treatment in 305 patients with flaring SLE: assessment by drug levels and self-administered questionnaires. Clin Pharmacol Ther. 2018;103(6):1074–82.
Durcan L, Clarke WA, Magder LS, Petri M. Hydroxychloroquine blood levels in systemic lupus erythematosus: clarifying dosing controversies and improving adherence. J Rheumatol. 2015;42:2092–7.
Costedoat-Chalumeau N, Amoura Z, Hulot JS, Hammoud HA, Aymard G, Cacoub P, et al. Low blood concentration of hydroxychloroquine is a marker for and predictor of disease exacerbations in patients with systemic lupus erythematosus. Arthritis Rheum. 2006;54:3284–90.
Tett SE, Cutler DJ, Beck C, Day RO. Concentration-effect relationship of hydroxychloroquine in patients with rheumatoid arthritis: a prospective, dose ranging study. J Rheumatol. 2000;27:1656–60.
Munster T, Gibbs JP, Shen D, Baethge BA, Botstein GR, Caldwell J, et al. Hydroxychloroquine concentration-response relationships in patients with rheumatoid arthritis. Arthritis Rheum. 2002;46:1460–9.
Lee JY, Vinayagamoorthy N, Han K, Kwok SK, Ju JH, Park KS, et al. Association of polymorphisms of cytochrome P450 2D6 with blood hydroxychloroquine levels in patients with systemic lupus erythematosus. Arthritis Rheumatol. 2016;68:184–90.
Shaw E, Kaczorowski J. Postpartum care: what’s new? Curr Opin Obstet Gynecol. 2007;19:561–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Salary and/or research support for this project was provided by the Rheumatology Research Foundation’s Scientist Development Award, the Thrasher Research Fund, the Derfner Foundation, NIGMS/NICHD (2T32GM086330-06), NICHD (5R01-HD076676-04, HHSN275201000003I), and a Duke Health/Private Diagnostic Clinic ENABLE grant. The National Institutes of Health Sponsored open access.
Conflict of interest
Stephen J. Balevic receives salary and research support from the National Institute of General Medical Sciences and the National Institute of Child Health and Human Development (2T32GM086330-06, 5R01-HD076676-04, HHSN275201000003I), the Rheumatology Research Foundation, and the Thrasher Research Fund. Michael Cohen-Wolkowiez receives support for research from the National Institutes of Health (5R01-HD076676, HHSN275201000003I), National Institute of Allergy and Infectious Diseases/National Institutes of Health (HHSN272201500006I), US Food and Drug Administration (1U18-FD006298), Biomedical Advanced Research and Development Authority (HHSO1201300009C), and from the industry for the drug development in adults and children (http://www.dcri.duke.edu/research/coi.jsp). Amanda M. Eudy is a consultant for GlaxoSmithKline and was previously a graduate research assistant for GlaxoSmithKline. Megan E. B. Clowse and Amanda M. Eudy have received independent medical education Grants from GlaxoSmithKline. Thomas P. Green has no conflicts of interest directly relevant to the contents of this article. Laura E. Schanberg receives research support from the National Institutes of Health (5R01-AR063890-02, 1U19AR069522-01, 1U34AR066294), Patient-Centered Outcomes Research Institute (CER-1408-20534, PPRN-1306-04601), Childhood Arthritis and Rheumatology Research Alliance, and Swedish Orphan Biovitrum AB. She participates in the Data Safety and Monitoring Board for UCB and Sanofi. Megan E. B. Clowse serves as a consultant for UCB and AstraZeneca.
Ethics approval
We conducted the study in compliance with the Declaration of Helsinki and the Duke Institutional Review Board approved the protocol.
Informed consent
All subjects included in the study provided informed consent.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Balevic, S.J., Green, T.P., Clowse, M.E.B. et al. Pharmacokinetics of Hydroxychloroquine in Pregnancies with Rheumatic Diseases. Clin Pharmacokinet 58, 525–533 (2019). https://doi.org/10.1007/s40262-018-0712-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-018-0712-z