Abstract
Purpose of Review
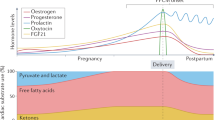
Peripartum cardiomyopathy (PPCM) is an important condition with high morbidity and mortality worldwide. Patients with PPCM are at risk of developing lifelong cardiac disease, requiring regular management and medical intervention. This article conducts a review of recent literature and gives insight into this disease.
Recent Findings
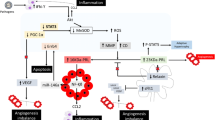
There is promising research in the fields of vascular, hormonal, and genetics. A number of genetic markers are being analyzed, including TTNC1, TTN, and STAT3. Mutations to these genes have been found to be prevalent in PPCM. These combined with the secretion of placental angiogenic factors potentially create imbalance in angiogenesis as the primary etiology.
Summary
Current biomarkers do not differentiate between PPCM and other variants of heart failure. Women with PPCM are more likely to have a cesarean section, to have hypertensive disease, at greater risk of major adverse cardiac events, and to have lifelong morbidity.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Cunningham FG, Byrne JJ, Nelson DB. Peripartum cardiomyopathy. Obstet Gynecol. 2019;133(1):167–79.
Bozkurt B, Colvin M, Cook J, Cooper LT, Deswal A, Fonarow GC, et al. American Heart Association Committee on Heart Failure and Transplantation of the Council on Clinical Cardiology; Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; Council on Epidemiology and Prevention; and Council on Quality of Care and Outcomes Research. Current diagnostic and treatment strategies for specific dilated cardiomyopathies: a scientific statement from the American Heart Association. Circulation. 2016;134:e579–e646. Most recent guidelines for PPCM in the USA.
Westhoff-Bleck M, Hilfiker-Kleiner D, Pankuweit S, Schieffer B. Cardiomyopathies and congenital heart disease in pregnancy. Geburtshilfe Frauenheilkd. 2018;78(12):1256–61.
Honigberg MC, Givertz MM. Peripartum cardiomyopathy. BMJ. 2019;364:k5287.
Arany Z, Elkayam U. Peripartum cardiomyopathy. Circulation. 2016;133:1397–409.
Haghikia A, Podewski E, Libhaber E, Labidi S, Fischer D, Roentgen P, et al. Phenotyping and outcome on contemporary management in a German cohort of patients with peripartum cardiomyopathy. Basic Res Cardiol. 2013;108(4):366.
Mielniczuk LM, Williams K, Davis DR, Tang ASL, Lemery R, Green MS, et al. Frequency of peripartum cardiomyopathy. Am J Cardiol. 2006;97(12):1765–8.
Hilfiker-Kleiner D, Haghikia A, Berliner D, Vogel-Claussen J, Schwab J, Franke A, et al. Bromocriptine for the treatment of peripartum cardiomyopathy: a multicentre randomized study. Eur Heart J. 2017;38(35):2671–9.
Afana M, Brinjikji W, Kao D, Jackson E, Maddox TM, Childers D, et al. Characteristics and in-hospital outcomes of peripartum cardiomyopathy diagnosed during delivery in the United States from the Nationwide Inpatient Sample (NIS) database. J Card Fail. 2016;22(7):512–9.
Pfeffer TJ, Hilfiker-Kleiner D. Pregnancy and heart disease: pregnancy-associated hypertension and peripartum cardiomyopathy. Curr Probl Cardiol. 2018;43(9):364–88.
Behrens I, Basit S, Lykke JA, Ranthe MF, Wohlfahrt J, Bundgaard H, et al. Hypertensive disorders of pregnancy and peripartum cardiomyopathy: a nationwide cohort study. PLoS One. 2019;14(2):e0211857.
Bello N, Rendon ISH, Arany Z. The relationship between pre-eclampsia and peripartum cardiomyopathy: a systematic review and meta-analysis. J Am Coll Cardiol. 2013;62(18):1715–23.
Kerpen K, Koutrolou-Sotiropoulou P, Zhu C, Yang J, Lyon J-A, Lima FV, et al. Disparities in death rates in women with peripartum cardiomyopathy between advanced and developing countries: a systematic review and meta-analysis. Arch Cardiovasc Dis. 2018;112(3):187–98.
• McNamara DM, Elkayam U, Alharethi R, Damp J, Hsich E, Ewald G, et al. Clinical outcomes for peripartum cardiomyopathy in North America: results of the IPAC Study (investigations of pregnancy-associated cardiomyopathy). J Am Coll Cardiol. 2015;66(8):905–14. A prospective cohort of 100 women with PPCM demonstrating an association between the level of ventricular dysfunction at enrollment and poorer long-term outcomes.
Goland S, Modi K, Hatamizadeh P, Elkayam U. Differences in clinical profile of African-American women with peripartum cardiomyopathy in the United States. J Card Fail. 2013;19(4):214–8.
Bültmann BD, Klingel K, Näbauer M, Wallwiener D, Kandolf R. High prevalence of viral genomes and inflammation in peripartum cardiomyopathy. Am J Obstet Gynecol. 2005;193(2):363–5.
Schelbert EB, Elkayam U, Cooper LT, Givertz MM, Alexis JD, Briller J, et al. Myocardial damage detected by late gadolinium enhancement cardiac magnetic resonance is uncommon in peripartum cardiomyopathy 2017;6(4):e005472.
Arany Z. Understanding peripartum cardiomyopathy 2018;69(1):165–76.
Vieira Borba V, Shoenfeld Y. Prolactin, autoimmunity, and motherhood: when should women avoid breastfeeding? Clin Rheumatol. 2019;38(5):1263–70.
Haghikia A, Kaya Z, Schwab J, Westenfeld R, Ehlermann P, Bachelier K, et al. Evidence of autoantibodies against cardiac troponin I and sarcomeric myosin in peripartum cardiomyopathy. Basic Res Cardiol. 2015;110(6):60.
Ware JS, Li J, Mazaika E, Yasso CM, DeSouza T, Cappola TP, et al. Shared genetic predisposition in peripartum and dilated cardiomyopathies. NEJM. 2016;374(3):233–41.
Yang Y, Rodriguez JE, Kitsis RN. A microRNA links prolactin to peripartum cardiomyopathy. J Clin Invest. 2013;123(5):1925–7.
• Hilfiker-Kleiner D, Kaminski K, Podewski E, Bonda T, Schaefer A, Sliwa K, et al. A cathepsin D-cleaved 16 kDa form of prolactin mediates postpartum cardiomyopathy. Cell. 2007;128(3):589–600. The authors posited a novel mechanism for the therapeutic use of bromocriptine in murine models with PPCM.
Bajou K, Herkenne S, Thijssen VL, D’Amico S, Nguyen NQ, Bouché A, et al. PAI-1 mediates the antiangiogenic and profibrinolytic effects of 16K prolactin. Nat Med. 2014;20:741–7.
Halkein J, Tabruyn SP, Ricke-Hoch M, Haghikia A, Nguyen N-Q-N, Scherr M, et al. MicroRNA-146a is a therapeutic target and biomarker for peripartum cardiomyopathy. J Clin Invest. 2013;123(5):2143–54.
Patten IS, Rana S, Shahul S, Rowe GC, Jang C, Liu L, et al. Cardiac angiogenic imbalance leads to peripartum cardiomyopathy. Nature. 2012;485(7398):333–8.
Mebazaa A, Seronde M-F, Gayat E, Tibazarwa K, Anumba DOC, Akrout N, et al. Imbalanced angiogenesis in peripartum cardiomyopathy ― diagnostic value of placenta growth factor ―. Circ J. 2017;81(11):1654–61.
Naftali-Shani N, Molotski N, Nevo-Caspi Y, Arad M, Kuperstein R, Amit U, et al. Modeling peripartum cardiomyopathy with human induced pluripotent stem cells reveals distinctive abnormal function of cardiomyocytes. Circulation. 2018;138(23):2721–3.
Sovio U, Gaccioli F, Cook E, Hund M, Charnock-Jones DS, Smith GCS. Prediction of preeclampsia using the soluble fms-like tyrosine kinase 1 to placental growth factor ratio: a prospective cohort study of unselected nulliparous women. Hypertension (Dallas, Tex : 1979). 2017;69(4):731–8.
Zeisler H, Llurba E, Chantraine F, Vatish M, Staff AC, Sennström M, et al. Predictive value of the sFlt-1:PlGF ratio in women with suspected preeclampsia. N Engl J Med. 2016;374(1):13–22.
Touyz RM, Herrmann J. Cardiotoxicity with vascular endothelial growth factor inhibitor therapy. JCO Precis Oncol. 2018;2(1):13.
Stack JP, Moslehi J, Sayed N, Wu JC. Cancer therapy-induced cardiomyopathy: can human induced pluripotent stem cell modelling help prevent it? Eur Heart J. 2018;0:1–7.
Sliwa K, Mebazaa A, Hilfiker-Kleiner D, Petrie MC, Maggioni AP, Laroche C, et al. Clinical characteristics of patients from the worldwide registry on peripartum cardiomyopathy (PPCM). Eur J Heart Fail. 2017;19(9):1131–41.
Masoomi R, Shah Z, Arany Z, Gupta K. Peripartum cardiomyopathy: an epidemiologic study of early and late presentations. Pregnancy Hypertens. 2018;13:273–8.
Stergiopoulos K, Lima FV. Peripartum cardiomyopathy-diagnosis, management, and long term implications. Trends Cardiovasc Med. 2019;29(3):164–73.
Lima FV, Yang J, Xu J, Stergiopoulos K. National trends and in-hospital outcomes in pregnant women with heart disease in the United States. Am J Cardiol. 2017;119(10):1694–700.
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH, et al. ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J am Coll Cardiol. 2013;62:e147–e239.
Bauersachs J, Arrigo M, Hilfiker-Kleiner D, Veltmann C, Coats AJS, Crespo-Leiro MG, et al. Current management of patients with severe acute peripartum cardiomyopathy: practical guidance from the Heart Failure Association of the European Society of Cardiology Study Group on peripartum cardiomyopathy. 2016;18(9):1096–105.
Iung B, Blomström-Lundqvist C, Warnes CA, Rosano GMC, Lang IM, Morais J, et al. 2018 ESC guidelines for the management of cardiovascular diseases during pregnancy. Eur Heart J. 2018;39(34):3165–241.
Bachelier-Walenta K, Hilfiker-Kleiner D, Sliwa K. Peripartum cardiomyopathy: update 2012. Curr Opin Crit Care. 2013;19(5):397–403.
Sliwa K, Hilfiker-Kleiner D, Mebazaa A, Petrie MC, Maggioni AP, Regitz-Zagrosek V, et al. EURObservational Research Programme: a worldwide registry on peripartum cardiomyopathy (PPCM) in conjunction with the Heart Failure Association of the European Society of Cardiology Working Group on PPCM. 2014;16(5):583–91.
Hilfiker-Kleiner D, Haghikia A, Masuko D, Nonhoff J, Held D, Libhaber E, et al. Outcome of subsequent pregnancies in patients with a history of peripartum cardiomyopathy. Eur J Heart Fail. 2017;19(12):1723–8.
Koenig T, Bauersachs J, Hilfiker-Kleiner D. Bromocriptine for the treatment of peripartum cardiomyopathy. Card Fail Rev. 2018;4(1):46–9.
Arrigo M, Blet A, Mebazaa A. Bromocriptine for the treatment of peripartum cardiomyopathy: welcome on BOARD. Eur Heart J. 2017;38(35):2680–2.
Harper MA, Meyer RE, Berg CJ. Peripartum cardiomyopathy: population-based birth prevalence and 7-year mortality. Obstet Gynecol. 2012;120(5):1013–9.
Dayoub EJ, Datwani H, Lewey J, Groeneveld PW. One-year cardiovascular outcomes in patients with peripartum cardiomyopathy. J Card Fail. 2018;24(10):711–5.
Lima FV, Parikh PB, Zhu J, Yang J, Stergiopoulos K. Association of cardiomyopathy with adverse cardiac events in pregnant women at the time of delivery. JACC: Heart Fail. 2015;3(3):257–66.
Chhabra N, Gupta A, Chibber R, Minhaj M, Hofer J, Mueller A, et al. Outcomes and mortality in parturient and non-parturient patients with peripartum cardiomyopathy: a national readmission database study. Pregnancy Hypertens. 2017;10:143–8.
Shah M, Ram P, Lo KB, Patnaik S, Patel B, Tripathi B, et al. Etiologies, predictors, and economic impact of 30-day readmissions among patients with peripartum cardiomyopathy. Am J Cardiol. 2018;122(1):156–65.
Lameijer H, Aalberts JJJ, van Veldhuisen DJ, Meijer K, Pieper PG. Efficacy and safety of direct oral anticoagulants during pregnancy; a systematic literature review. Thromb Res. 2018;169:123–7.
Kolte D, Khera S, Aronow WS, Palaniswamy C, Mujib M, Ahn C, et al. Temporal trends in incidence and outcomes of peripartum cardiomyopathy in the United States: a nationwide population based study 2014;3(3):e001056.
Elkayam U, Tummala PP, Rao K, Akhter MW, Karaalp IS, Wani OR, et al. Maternal and fetal outcomes of subsequent pregnancies in women with peripartum cardiomyopathy. 2001;344(21):1567–71.
Koutrolou-Sotiropoulou P, Lima FV, Stergiopoulos K. Quality of life in survivors of peripartum cardiomyopathy. Am J Cardiol. 2016;118(2):258–63.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Lindsay Ballard and Adrian Cois declare no conflict of interest.
Bory Kea is a site investigator for Ortho Clinical Diagnostics; no funds were directly received. Dr. Kea also received a grant through the NHLBI K08 Career Development Award.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Heart Failure
Rights and permissions
About this article
Cite this article
Ballard, L.C., Cois, A. & Kea, B. Peripartum Cardiomyopathy: a Review. Curr Emerg Hosp Med Rep 7, 127–134 (2019). https://doi.org/10.1007/s40138-019-00192-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40138-019-00192-3