Abstract
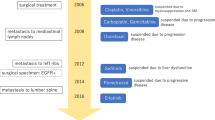
Afatinib is a second-generation, oral, epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI). One of the most common adverse effects of affatinib is diarrhea, which may lead to acute kidney injury (AKI) due to severe plasma volume loss; however, no case of glomerular injury directly induced by afatinib has been reported to date. Here, we describe the case of a 53-year-old Japanese male patient with advanced lung adenocarcinoma who twice developed AKI requiring dialysis, once after starting and once after increasing the dose of afatinib. Although serum anti-neutrophil cytoplasmic antibodies were negative, crescentic glomerulonephritis with no immune deposits was confirmed on kidney biopsy. No vasculitis-like signs were observed in other organs, such as lung, skin, or peripheral nerves. Afatinib was considered the cause of glomerular damage and was immediately discontinued; corticosteroids were administered. Renal function gradually recovered thereafter, with serum creatinine levels at ~ 2.3 mg/dL after second-line therapy with bevacizumab and atezolizumab. Several cases of cutaneous leukocytoclastic vasculitis have been reported in patients treated with other EGFR-TKIs; therefore, afatinib-induced vasculitis may lead to crescentic glomerulonephritis. Although afatinib-induced glomerular injury is extremely rare and has an unclear mechanism, renal function and urinary findings need to be closely monitored.



Similar content being viewed by others
References
Paez JG, Jänne PA, Lee JC, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304:1497–500.
Takano T, Fukui T, Ohe Y, et al. EGFR mutations predict survival benefit from gefitinib in patients with advanced lung adenocarcinoma: a historical comparison of patients treated before and after gefitinib approval in Japan. J Clin Oncol. 2008;26:5589–95.
Planchard D, Popat S, Kerr K, et al. Metastatic non-small cell lung cancer: ESMO clinical practice guidelines for diagnosis treatment and follow-up. Ann Oncol. 2018;29:iv192–237.
Wu YL, Planchard D, Lu S, et al. Pan-Asian adapted clinical practice guidelines for the management of patients with metastatic non-small-cell lung cancer: a CSCO-ESMO initiative endorsed by JSMO, KSMO, MOS SSO and TOS. Ann Oncol. 2019;30:171–210.
Park K, Tan EH, O’Byrne K, et al. Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol. 2016;17:577–89.
Wu YL, Zhou C, Hu CP, et al. Afatinib versus cisplatin plus gemcitabine for first-line treatment of Asian patients with advanced non-small-cell lung cancer harbouring EGFR mutations (LUX-Lung 6): an open-label, randomised phase 3 trial. Lancet Oncol. 2014;15:213–22.
Sequist LV, Yang JC, Yamamoto N, et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol. 2013;31:3327–34.
Soria JC, Felip E, Cobo M, et al. Afatinib versus erlotinib as second-line treatment of patients with advanced squamous cell carcinoma of the lung (LUX-Lung 8): an open-label randomised controlled phase 3 trial. Lancet Oncol. 2015;16:897–907.
Yang JC, Reguart N, Barinoff J, et al. Diarrhea associated with afatinib: an oral ErbB family blocker. Expert Rev Anticancer Ther. 2013;13:729–36.
Pharmaceutical interview form of Giotrif Tablets. Nippon Boehringer Ingelheim Co. L, editor. 2020. https://www.bij-kusuri.jp/products/attach/pdf/gio_t20_if.pdf
Guidelines for the treatment of rapidly progressive glomerulonephritis, second version. Nihon Jinzo Gakkai Shi. 2011; 53:509–55.
Gesualdo L, Di Paolo S, Calabró A, et al. Expression of epidermal growth factor and its receptor in normal and diseased human kidney: an immunohistochemical and in situ hybridization study. Kidney Int. 1996;49:656–65.
Bollée G, Flamant M, Schordan S, et al. Epidermal growth factor receptor promotes glomerular injury and renal failure in rapidly progressive crescentic glomerulonephritis. Nat Med. 2011;17:1242–50.
Benter IF, Benboubetra M, Hollins AJ, et al. Early inhibition of EGFR signaling prevents diabetes-induced up-regulation of multiple gene pathways in the mesenteric vasculature. Vasc Pharmacol. 2009;51:236–45.
François H, Placier S, Flamant M, et al. Prevention of renal vascular and glomerular fibrosis by epidermal growth factor receptor inhibition. Faseb J. 2004;18:926–8.
Hise MK, Salmanullah M, Liu L, et al. Control of the epidermal growth factor receptor and its ligands during renal injury. Nephron. 2001;88:71–9.
Gao Y, Zhao MH. Review article: drug-induced anti-neutrophil cytoplasmic antibody-associated vasculitis. Nephrology. 2009;14:33–41.
Oki R, Hirakawa Y, Oda Y, et al. Renal-limited ANCA-associated vasculitis during erlotinib treatment for lung carcinoma. CEN Case Rep. 2021. https://doi.org/10.1007/s13730-021-00632-8.
Kurita N, Mise N, Fujii A, et al. Crescentic glomerulonephritis in a patient with advanced lung cancer during erlotinib therapy. NDT Plus. 2009;2:512–3.
Nakopoulou L, Stefanaki K, Boletis J, et al. Immunohistochemical study of epidermal growth factor receptor (EGFR) in various types of renal injury. Nephrol Dial Transpl. 1994;9:764–9.
Faye E, Bondon-Guitton E, Olivier-Abbal P, et al. Spontaneous reporting of serious cutaneous reactions with protein kinase inhibitors. Eur J Clin Pharmacol. 2013;69:1819–26.
Kamo H, Shinozaki E, Sugase T, et al. Leukocytoclastic vasculitis with purpura and renal failure induced by the anti-epidermal growth factor receptor antibody panitumumab: a case report. J Med Case Rep. 2019;13:13.
Yang K, Zhang T, Chen J, et al. Immune thrombocytopenia as a paraneoplastic syndrome in patients with nasopharyngeal cancer. Head Neck. 2012;34:127–30.
Uchimiya H, Higashi Y, Kawai K, et al. Purpuric drug eruption with leukocytoclastic vasculitis due to gefitinib. J Dermatol. 2010;37:562–4.
Fekete GL, Fekete L. Cutaneous leukocytoclastic vasculitis associated with erlotinib treatment: a case report and review of the literature. Exp Ther Med. 2019;17:1128–31.
Acknowledgements
The authors would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interests exists.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Morita, D., Ito, K., Ikeuchi, N. et al. A case of crescentic glomerulonephritis induced by afatinib for lung adenocarcinoma. CEN Case Rep 12, 152–158 (2023). https://doi.org/10.1007/s13730-022-00737-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-022-00737-8