Abstract
Objectives
The aim of this study is to define the maximal safe radiation dose to guide further study of the GliaSite balloon brachytherapy (GSBT) system in untreated newly diagnosed glioblastoma (NEW-GBM) and recurrent high-grade glioma (REC-HGG). GBST is a balloon placed in the resection cavity and later filled through a subcutaneous port with liquid I-125 Iotrex, providing radiation doses that diminish uniformly with distance from the balloon surface.
Methods
The Adult Brain Tumor Consortium initiated prospective dose-finding studies to determine maximum tolerated dose in NEW-GBM treated before standard RT or after surgery for REC-HGG. Patients were inevaluable if there was progression before the 90-day posttreatment toxicity evaluation point.
Results
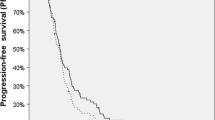
Ten NEW-GBM patients had the balloon placed, and 2/10 reached the 90 day timepoint. Five REC-HGG enrolled and two were assessable at the 90-day evaluation endpoint. Imaging progression occurred before 90-day evaluation in 7/12 treated patients. The trials were closed as too few patients were assessable to allow dose escalation, although no dose-limiting toxicities (DLTs) were observed. Median survival from treatment was 15.3 months (95 % CI 7.1–23.6) for NEW-GBM and 12.8 months (95 % CI 4.2–20.9) for REC-HGG.
Conclusion
These trials failed to determine a maximum tolerated dose (MTD) for further testing as early imaging changes, presumed to be progression, were common and interfered with the assessment of treatment-related toxicity. The survival outcomes in these and other related studies, although based on small populations, suggest that GSBT may be worthy of further study using clinical and survival endpoints, rather than standard imaging results. The implications for local therapy development are discussed.



Similar content being viewed by others
References
Stubbs JB, Frankel RH, Schultz K, Crocker I, Dillehay D, Olson JJ (2002) Preclinical evaluation of a novel device for delivering brachytherapy to the margins of resected brain tumor cavities. J Neurosurg 96(2):335–343
Stubbs JB, Strickland AD, Frank RK, Simon J, McMillan K, Williams JA (2000) Biodistribution and dosimetry of an aqueous solution containing sodium 3-(125I)iodo-4-hydroxybenzenesulfonate (iotrex) for brachytherapy of resected malignant brain tumors. Cancer Biother Radiopharm 15(6):645–656
Dempsey JF, Williams JA, Stubbs JB, Patrick TJ, Williamson JF (1998) Dosimetric properties of a novel brachytherapy balloon applicator for the treatment of malignant brain-tumor resection-cavity margins. Int J Radiat Oncol Biol Phys 42(2):421–429
Tatter SB, Shaw EG, Rosenblum ML et al (2003) An inflatable balloon catheter and liquid 125I radiation source (GliaSite radiation therapy system) for treatment of recurrent malignant glioma: multicenter safety and feasibility trial. J Neurosurg 99(2):297–303
Wallner KE, Galicich JH, Krol G, Arbit E, Malkin MG (1989) Patterns of failure following treatment for glioblastoma multiforme and anaplastic astrocytoma. Int J Radiat Oncol Biol Phys 16(6):1405–1409
Chan JL, Lee SW, Fraass BA et al (2002) Survival and failure patterns of high-grade gliomas after three-dimensional conformal radiotherapy. J Clin Oncol 20(6):1635–1642
Lee SW, Fraass BA, Marsh LH et al (1999) Patterns of failure following high-dose 3-D conformal radiotherapy for high-grade astrocytomas: a quantitative dosimetric study. Int J Radiat Oncol Biol Phys 43(1):79–88
Chamberlain MC (2011) Radiographic patterns of relapse in glioblastoma. J Neurooncol 101(2):319–323
Patel TR, McHugh BJ, Bi WL, Minja FJ, Knisely JP, Chiang VL (2011) A comprehensive review of MR imaging changes following radiosurgery to 500 brain metastases. AJNR Am J Neuroradiol 32(10):1885–1892
Kleinberg L, Yoon G, Weingart JD, et al. Imaging after GliaSite brachytherapy: prognostic MRI indicators of disease control and recurrence. Int J Radiat Oncol Biol Phys. 2009.
Fink J, Born D, Chamberlain MC (2011) Pseudoprogression: Relevance with respect to treatment of high-grade gliomas. Curr Treat Options Oncol 12(3):240–252
Souhami L, Seiferheld W, Brachman D et al (2004) Randomized comparison of stereotactic radiosurgery followed by conventional radiotherapy with carmustine to conventional radiotherapy with carmustine for patients with glioblastoma multiforme: report of radiation therapy oncology group 93–05 protocol. Int J Radiat Oncol Biol Phys 60(3):853–860
Laperriere NJ, Leung PM, McKenzie S et al (1998) Randomized study of brachytherapy in the initial management of patients with malignant astrocytoma. Int J Radiat Oncol Biol Phys 41(5):1005–1011
Cardinale R, Won M, Choucair A et al (2006) A phase II trial of accelerated radiotherapy using weekly stereotactic conformal boost for supratentorial glioblastoma multiforme: RTOG 0023. Int J Radiat Oncol Biol Phys 65(5):1422–1428
Schupak K, Malkin M, Anderson L, Arbit E, Lindsley K, Leibel S (1995) The relationship between the technical accuracy of stereotactic interstitial implantation for high grade gliomas and the pattern of tumor recurrence. Int J Radiat Oncol Biol Phys 32(4):1167–1176
Kleinberg L, Yoon G, Weingart JD et al (2009) Imaging after GliaSite brachytherapy: prognostic MRI indicators of disease control and recurrence. Int J Radiat Oncol Biol Phys 75(5):1385–1391
Chan TA, Weingart JD, Parisi M et al (2005) Treatment of recurrent glioblastoma multiforme with GliaSite brachytherapy. Int J Radiat Oncol Biol Phys 62(4):1133–1139
Gabayan AJ, Green SB, Sanan A et al (2006) GliaSite brachytherapy for treatment of recurrent malignant gliomas: a retrospective multi-institutional analysis. Neurosurgery 58(4):701–9, discussion 701–9
Gobitti C, Borsatti E, Arcicasa M et al (2011) Treatment of recurrent high-grade gliomas with GliaSite brachytherapy: a prospective mono-institutional italian experience. Tumori 97(5):614–619
Westphal M, Ram Z, Riddle V, Hilt D, Bortey E, Executive Committee of the Gliadel Study Group (2006) Gliadel wafer in initial surgery for malignant glioma: long-term follow-up of a multicenter controlled trial. Acta Neurochir (Wien) 148(3):269–75, discussion 275
Westphal M, Hilt DC, Bortey E et al (2003) A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (gliadel wafers) in patients with primary malignant glioma. Neuro Oncol 5(2):79–88
Brem H, Piantadosi S, Burger PC et al (1995) Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The polymer-brain tumor treatment group. Lancet 345(8956):1008–1012
Matheus MG, Castillo M, Ewend M et al (2004) CT and MR imaging after placement of the GliaSite radiation therapy system to treat brain tumor: initial experience. AJNR Am J Neuroradiol 25(7):1211–1217
Brandes AA, Tosoni A, Spagnolli F et al (2008) Disease progression or pseudoprogression after concomitant radiochemotherapy treatment: pitfalls in neurooncology. Neuro Oncol 10(3):361–367
Brandsma D, Stalpers L, Taal W, Sminia P, van den Bent MJ (2008) Clinical features, mechanisms, and management of pseudoprogression in malignant gliomas. Lancet Oncol 9(5):453–461
Walker AJ, Ruzevick J, Malayeri AA et al (2014) Postradiation imaging changes in the CNS: how can we differentiate between treatment effect and disease progression? Future Oncol 10(7):1277–1297
Brem H, Mahaley MS Jr, Vick NA et al (1991) Interstitial chemotherapy with drug polymer implants for the treatment of recurrent gliomas. J Neurosurg 74(3):441–446
Brem H, Ewend MG, Piantadosi S, Greenhoot J, Burger PC, Sisti M (1995) The safety of interstitial chemotherapy with BCNU-loaded polymer followed by radiation therapy in the treatment of newly diagnosed malignant gliomas: phase I trial. J Neurooncol 26(2):111–123
Olivi A, Grossman SA, Tatter S et al (2003) Dose escalation of carmustine in surgically implanted polymers in patients with recurrent malignant glioma: a new approaches to brain tumor therapy CNS consortium trial. J Clin Oncol 21(9):1845–1849
Kleinberg LR, Weingart J, Burger P et al (2004) Clinical course and pathologic findings after Gliadel and radiotherapy for newly diagnosed malignant glioma: implications for patient management. Cancer Invest 22(1):1–9
Welsh J, Sanan A, Gabayan AJ et al (2007) GliaSite brachytherapy boost as part of initial treatment of glioblastoma multiforme: a retrospective multi-institutional pilot study. Int J Radiat Oncol Biol Phys 68(1):159–165
Funding
This study was funded by the National Cancer Institute Grants CA62-475 and CA137443 and by Proxima Therapeutics, Alpharetta, GA.
Ethical standards statement
All human and animal studies have been approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All persons gave their informed consent prior to their inclusion in the study.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kleinberg, L.R., Stieber, V., Mikkelsen, T. et al. Outcome of Adult Brain Tumor Consortium (ABTC) prospective dose-finding trials of I-125 balloon brachytherapy in high-grade gliomas: challenges in clinical trial design and technology development when MRI treatment effect and recurrence appear similar. J Radiat Oncol 4, 235–241 (2015). https://doi.org/10.1007/s13566-015-0210-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13566-015-0210-y