Abstract
Background
Dissecting cellulitis of the scalp (DCS) is a chronic inflammatory skin condition characterized by abscesses, nodules, fistulas, and scarring alopecia. Management of this oftentimes debilitating dermatosis can be challenging due to its recalcitrant nature. There is limited data regarding the efficacy of treatment options for DCS.
Objective
The aim of this study was to conduct a systematic review of the literature to explore the efficacy and safety of reported DCS treatments.
Methods
In October 2022, MEDLINE and EMBASE databases were searched for articles on treatments for DCS. Studies that contained outcome efficacy data for DCS treatments were included. Reviews, conference abstracts, meta-analyses, commentaries, non-relevant articles, and articles with no full-text available were excluded. Data extraction was performed by two independent reviewers.
Results
A total of 110 relevant articles with 417 patients were identified. A majority of studies (86.4%) were case reports or series. Treatment options included systemic antibiotics, oral retinoids, biologics, procedural treatments, combination agents, and topical treatments. Oral retinoids and photodynamic therapy were the most extensively studied medical and procedural interventions, respectively.
Conclusion
Overall, randomized controlled trials are needed to evaluate various treatment regimens for DCS and provide patients with a robust, evidence-based approach to therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Although several medical and procedural treatments for dissecting cellulitis of the scalp (DCS) have been reported in the literature, the majority of data has been based on case reports or series. |
In terms of systemic therapies, antibiotics and oral retinoids have the most data to support their use in DCS, but in recent years there has been an increasing number of reports to support use of various biologic therapies. |
Procedural therapies for DCS including surgical excisions, photodynamic therapy, laser treatments, and radiation therapy overall had low rates of reported adverse events. |
Introduction
Dissecting cellulitis of the scalp (DCS) is a chronic, inflammatory skin disease that disproportionately affects African American males [1]. It is not a rare entity, with a reported prevalence of around 0.7% [2]. The condition is part of the follicular occlusion tetrad, which also consists of hidradenitis suppurativa (HS), acne conglobata, and pilonidal disease [1, 3]. DCS is characterized by abscesses and nodules that can eventually progress to form interconnected sinus tracts and cause scarring alopecia [4]. The disease is oftentimes refractory to treatment, making management challenging for clinicians. Although various treatment modalities have been reported, there is a lack of an updated review on their efficacy and safety. Herein, we aim to systematically evaluate existing literature on the efficacy and safety of medical and procedural treatments for DCS.
Methods
Search Strategy
This study was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and was preregistered on PROSPERO (CRD42022364109). This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. On 1 October 2022, two independent reviewers (E.M. and C.J.) searched MEDLINE and EMBASE databases from inception to search date with the following terms: (“dissecting cellulitis” OR “perifolliculitis capitis abscedens” OR “dissecting folliculitis” OR “Hoffman disease”).
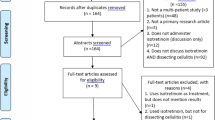
A total of 773 articles were identified and filtered to remove non-English language and non-human studies. Duplicate articles were excluded. Titles and abstracts were screened for relevance. Full text review was then manually performed on the remaining 218 articles by the two independent reviewers (E.M. and C.J.). Studies that contained outcome efficacy data for DCS treatments were considered eligible for inclusion. Reviews, conference abstracts, meta-analyses, commentaries, non-relevant articles, and articles with no full-text available were excluded. Any discrepancies were discussed to consensus with a third reviewer (J.L.H.). Reference lists of articles that met inclusion criteria were screened for additional relevant articles and 5 additional articles were identified.
Data Extraction
Two reviewers (E.M. and C.J.) independently completed data extraction. Any discrepancies were discussed to consensus with a third reviewer (J.L.H.). For each article, the study design, country of study, patient characteristics, study intervention, efficacy outcomes, and safety outcomes were recorded. Articles were assessed for quality utilizing Cochrane risk of bias for prospective trials [5] and Newcastle–Ottawa Scale (NOS) for cohort studies [6].
Results
A total of 110 articles published between 1951 to 2022 fit the aforementioned search criteria and were included in this review (Fig. 1) [7]. These articles comprised 417 patients across 6 prospective trials, 8 retrospective cohort studies, 1 prospective cohort study, and 95 case reports/series. Study design, patient demographics, interventions, previous treatments, concomitant treatments, response, and adverse effects of the included studies are reported in Tables 1, 2, 3, and 4.
Data on patients’ sexes were available for 375 patients and 94.9% (356/375) of patients were male. Age at the time of study ranged from 14 months to 86 years. Top reported comorbidities included acne (17.5%, 73/417) and hidradenitis suppurativa (8.6%, 36/417). Study locations included the USA (n = 35), China (n = 11), UK (n = 7), Japan (n = 6), Spain (n = 5), Germany and Brazil (n = 4 each), Italy, India, Canada, Croatia and France (n = 3 each), Greece, Denmark, Turkey and Sweden (n = 2 each), Colombia, the Netherlands, Lebanon, Tunisia, Taiwan, Poland, Israel, Bulgaria, Saudi Arabia, Australia, Puerto Rico, Switzerland, Romania, Scotland and Mexico (n = 1 each). In terms of study quality, all cohort studies were of poor quality and all prospective trials carried a high risk of bias.
There were 17 articles on systemic antibiotic treatments, 25 on oral retinoids, 21 describing biologic treatments, and 39 on procedural treatments. Other interventions studied include combination (n = 10), topical (n = 3), and a few miscellaneous (n = 7) treatments. Response to treatment was defined as partial or complete improvement in a patient’s condition as determined by each individual study. Measures for improvement used by studies were variable and included reduction in number and size of inflammatory nodules and abscesses; flattening of sinus tracts; improvement in pain, drainage, itch, and swelling; and hair regrowth. The outcome measure for each individual study has been described in Tables 1–4, where available. Overall response rate to a specific DCS intervention was calculated on the basis of aggregate response across studies.
Topical Treatments
Limited data were available for topical DCS treatments (Table 1). Gamissans et al.’s retrospective cohort study reported a partial response in 3 out of 11 patients treated with topical antibiotics. Pruritus or erythema was reported to occur in 4 out of 11 treated patients [8]. One case report noted improvement with a isotretinoin gel and clindamycin gel combination treatment in a 20-year-old male [9]. Topical resorcinol at 15% strength applied twice daily induced clinical improvement in a 14-year-old male [10].
Systemic Antibiotics
The antibacterial and antiinflammatory effects of oral antibiotics may play a role in mitigating DCS symptoms (Table 2) [11]. The overall response rate to systemic antibiotic treatments across studies was 97.8% (91/93). Antibiotics that were utilized include doxycycline (n = 18), dapsone (n = 4), azithromycin (n = 3), ciprofloxacin and trimethoprim (n = 2 each), and lymecycline, oxytetracycline, rifampicin, clindamycin, terramycin, chloramphenicol, cephalexin and gentamicin (n = 1 each). Antibiotic combinations studied included clindamycin and rifampin (n = 7). Time to response to oral antibiotics, when reported, ranged from 6 days to 18 months. Seven patients who received doxycycline in a prospective trial had a good response to the intervention after 3 months, but none were cured of their disease [12]. A retrospective cohort study by Badaoui et al. described moderate improvement in 40 patients who received various systemic antibiotics; however, patients relapsed frequently after discontinuation of treatment [4]. Gamissans et al.’s retrospective cohort study described 14 patients, 6 of whom received doxycycline for 3 months, 4 patients on a regimen of rifampin and clindamycin for 3 months, and 4 patients on dapsone. Overall, complete recovery of alopecia was seen in 3 patients, and 11 exhibited a partial response. Four of the patients had gastrointestinal (GI) distress [8]. Melo et al.’s retrospective cohort study demonstrated improvement in nine out of ten patients on lymecycline 300 mg (mg) per day for 3 months [13].
Oral Retinoids
Retinoids have antiinflammatory and antiproliferative effects, which may normalize the epithelium and decrease follicular occlusion [14]. Isotretinoin has been the most studied oral retinoid for DCS with an overall response rate of 91% (142/156); 25.4% (29/114) of patients treated with isotretinoin had concomitant acne. Isotretinoin dosing ranged from 0.3–1 mg/kg/day and reported duration of treatment ranged from 1–12 months. Isotretinoin led to a significant reduction of inflammatory activity in 90.3% of 72 adult patients in a large retrospective multicenter study [15]. Badaoui et al.’s retrospective cohort study described improvement with isotretinoin in 33 of 35 patients; however, patients relapsed frequently after discontinuation [4]. Lee et al.’s retrospective study also reported complete or partial response in 12 out of 16 patients who received isotretinoin for their DCS. Although there was no significant correlation between the isotretinoin dose and degree of response, the two patients who had recurrences had received lower cumulative doses [16]. Another retrospective cohort study described a partial or complete response in three out of four patients who received isotretinoin. One out of three patients with a reported response had a recurrence [8]. Adverse effects reported with isotretinoin across all studies included xerosis, dry eyes, erosive skin lesions, pruritus, epistaxis, and hyperlipidemia. There was one report of alitretinoin use in the literature. Significant improvement was noted in a 15-year-old male who received a dose of 10–20 mg/day for 5 months; however, erosive skin lesions developed when the dose was increased to 20 mg/day [17]. Finally, a patient who was prescribed acitretin 30 mg/day was lost to follow-up [4].
Biologics
It has been posited that biologics may downregulate key inflammatory markers such as tumor necrosis factor (TNF)-alpha and interleukin (IL)-12/23, which are associated with follicular occlusion disorders such as DCS [18, 19]. Biologics studied for treatment of DCS include adalimumab, infliximab, secukinumab, guselkumab, risankizumab, and tildrakizumab. Most (70%, 14/20) studies on biologics for DCS have been published in the last 5 years. HS was reported as a comorbidity in 41.2% (14/34) of patients. The majority of patients had previously failed treatment with oral antibiotics (82.4%, 28/34) and oral retinoids (58.8%, 20/34).
Adalimumab (ADA) was the most studied, and overall 94.1% (16/17) of patients exhibited a response to the immunomodulator. Most (83.3%, 10/12) of the studies were case reports/series. Dosing of ADA ranged from 40–80 mg every 1–2 weeks. A response rate of 85.7% (12/14) was observed for infliximab (IFX) treatment across two retrospective cohort studies and five case reports/series. The IFX dosing interval was 5 mg/kg every 4–8 weeks. Adverse events reported with IFX treatment include an infusion reaction, psoriasiform spongiotic dermatitis, and retrobulbar optic neuritis.
IL-23 inhibitors have a few reported cases in the literature for DCS. A reduction in pustules, decreased tenderness, and hair regrowth was observed in a 28-year-old male after receiving two doses of tildrakizumab [20]. Babalola et al. described improvement after 7 months in a 65-year-old male’s DCS symptoms with risankizumab dosed at 150 mg every 12 weeks [21]. Finally, guselkumab treatment at a maintenance dose of 100 mg every 8 weeks for 6 months was effective in inducing near complete resolution of symptoms in a case report of a male patient who had failed treatment with ADA [22].
Treatment with secukinumab, an IL-17 inhibitor, was described in a 63-year-old male patient. He had complete remission for 1 year after taking secukinumab 150 mg weekly for 6 weeks (patient mistakenly took an extra loading dose) then monthly for 2 months. Although the patient developed an eczematous reaction as an adverse event, this was successfully treated with topical therapy [23].
Miscellaneous Systemic Therapies
Long-term prednisone therapy was reported in one patient. Signs of active inflammation subsided within 4 months and full regrowth of scalp hair was observed after 1 year of treatment. The patient was maintained on 5 mg of prednisone every other day after 1 year of multiple tapered courses of treatment [24]. Finasteride 1 mg/day was also shown to be effective in two out of three treated patients [25].
Zinc is known to have antiinflammatory and antioxidant properties via inhibition of inflammatory mediators such as TNF-alpha [26]. A retrospective cohort study which included eight patients receiving zinc found that none of them exhibited a response [4]. The use of zinc for DCS was also described in two case reports, both of which reported positive results [27, 28]. One of the patients was in remission for 5 years and the other had well-controlled lesions for 1 year. Oral saireito, a Japanese herb, improved DCS disease course in two patients [29].
Procedural Treatments
Surgical Interventions
Surgical excisions with and without skin grafts were reported to induce improvement in 21 patients across one retrospective cohort study and 13 case reports/series; long-term remission was reported across multiple studies. Many of the patients undergoing surgery had failed medical management with oral antibiotics and/or oral retinoids (Table 3). Split-thickness skin grafts were most commonly used for reconstruction. Complications were uncommon in the literature. Baneu et al. reported a seroma at the skin graft donor site [30], and Dellon et al.’s case series described a skin graft infection in one patient [31].
Photodynamic Therapy
Photodynamic therapy (PDT) inhibits follicular secretions, mitigates hyperkeratinization, reduces inflammation, and ablates tissues. PDT may also destroy persistent microbial biofilms, which can contribute to DCS symptoms [32]. The overall response rate to PDT with aminolaevulinic acid (ALA) across three prospective trials, one retrospective cohort study, and six case reports/series was 88.3% (83/94).
Wu et al.’s 2021 prospective trial demonstrated a novel technique in which a heated microneedle was used to puncture lesions. Thereafter, 5% ALA was applied followed by red-light PDT treatment. After four sessions, a total of 34 out of 42 patients reported improvement. Ten of the patients had relapsed within 1 year. Itching, burning, swelling, and thin crusts were noted as transient side effects [33]. Su et al. and He et al. described similar procedures in 3 and 6 patients, respectively [34, 35]. All patients had good results, but notably, Su et al. used isotretinoin as a concomitant treatment in all patients [34]. Adverse events reported with PDT include erythema, pain, itching, and swelling.
Laser Treatments
Lasers may selectively or non-selectively destroy hair follicles, thereby counteracting the progression of events that lead to follicular occlusion and rupture [36].
Long-pulsed neodymium-doped yttrium–aluminum–garnet (Nd:YAG) laser treatment sessions were effective in reducing drainage and tenderness in four males aged 25–40. A total of three to seven treatment sessions were conducted [37]. A long-pulsed ruby laser was used to perform laser hair removal in two males and one female. All patients had beneficial results, although one reported crusting and erosion with persistent hypopigmentation [38]. A case series reported improvement in two males after treatment with the erbium-doped (Er):YAG laser. One of the patients had good results after two treatment sessions and the other improved after 4 sessions [39].
Improvement was observed in a single case report of a 35-year-old male who received four treatments with 800-nm pulsed diode laser. Significant epilation was reported after 1 month and the disease was quiescent at the 6 month follow-up [40]. In Glass et al.’s 1989 case report, excision with a carbon dioxide laser led to complete clearance of disease after 6 weeks. No recurrence was observed after 4 months [41].
Radiation Treatment
A total of four patients received radiation therapy consisting of electron beam radiation or combination electron and photon radiation; all patients had decreased drainage and nodule sizes after the culmination of treatment sessions. Mild scalp irritation, erythema, xeroderma, and pruritus were noted [42]. A 2016 study by Paul et al. reported substantial improvement in a 46-year-old male with brachytherapy to the occiput [43]. Of note, x-ray irradiation, no longer used in the present day for cutaneous conditions given its side effect profile, was performed in nine patients across three studies conducted in the 1950s and 1960s and resulted in disease improvement in all patients [44,45,46].
Intralesional Steroid Injections
Gamissans et al.’s retrospective cohort study included one patient who received intralesional steroid injections that resulted in partial recovery of disease, although discrete skin atrophy was noted as a side effect [8]. A 30-year-old female experienced improvement after two treatments with hydrocortisone injections into abscesses every 4–5 days. However, the patient relapsed after 6 months of no treatments [45].
Intracavitary Foam Sclerotherapy
Fibrosis and flattening of DCS lesions were observed in three patients who underwent intracavitary foam sclerotherapy. No complications were noted throughout the course of treatment [47].
Compression Bandage
A 44-year-old male self-devised a compression dressing consisting of gauze, an eye patch, and a headband worn under a cap. The affected lesion became smaller and flatter after 1 month and demonstrated terminal hair growth after 4 months [48].
Combination Therapies
A variety of combination treatment approaches have been reported for DCS (Table 4). All 17 patients across ten case reports/series improved with combination therapies for DCS. A total of 14 patients on combined medical therapies such as oral retinoids and oral antibiotics improved, with the exception of one who was lost to follow-up. Similarly, three studies described combined medical and procedural treatments in four patients. Of the three studies, two examined intralesional steroid injection use combined with various doses of isotretinoin [49, 50]. All three patients improved.
Discussion
Our systematic review of DCS management strategies highlights the paucity of evidence-based data for DCS. The majority of studies in the literature describing DCS treatments were case reports or series. Large randomized controlled trials and retrospective cohort studies were scarce, which limits our ability to draw definitive conclusions regarding the efficacy of the reported treatment modalities. Oral retinoids and PDT were the most extensively studied medical and procedural treatments, respectively.
The treatment of DCS remains a challenge for dermatologists due to the lack of standardized, evidence-based guidelines. Although the pathogenesis of DCS is poorly understood, it is believed that epithelial shedding is the initial inciting event that leads to hyperkeratosis and dilatation within the hair follicle. The distended follicle ruptures, which propagates a dysregulated inflammatory response and results in the formation of nodules and abscesses followed by fistulas and scarring [51]. DCS shares several commonalities with HS in terms of suspected pathogenesis and treatments, and some posit that DCS may be a variant of HS [52]. However, there are differences between the two conditions as well. DCS appears to be more responsive to oral retinoids compared with HS, which exhibits a mixed and inconsistent response to oral retinoids. Furthermore, at least in Western nations, HS has been found to predominantly affect women [53], whereas DCS affects men in a highly disproportionate manner.
Based on findings from this systematic review and the authors’ expert opinion, we developed a treatment algorithm for DCS to help guide clinicians (Fig. 2). We recommend using systemic antibiotics and intralesional steroid injections to help control symptoms based on the safety profile and fast onset of action of these therapies. Tetracyclines are the most studied antibiotic class for DCS; however, other antibiotics have also shown benefit in case of treatment failure and non-tolerance, including azithromycin, trimethoprim–sulfamethoxazole, and dapsone. Similar to HS, dual antibiotic combination therapy such as clindamycin and rifampin may also be utilized for more symptomatic cases of DCS.
Dissecting cellulitis of the scalp treatment algorithm.†BID, twice a day; CO2, carbon dioxide; IL, interleukin; ILTAC, intralesional triamcinolone; kg; kilogram; mg, milligram; ND:YAG, neodymium-doped yttrium–aluminum–garnet; q, every; QD, daily. †Algorithm developed on the basis of systematic review and authors’ expert opinion. *Other systemic antibiotic therapy to consider if patient fails or is intolerant to tetracyclines: dapsone, azithromycin, trimethoprim–sulfamethoxazole, cephalexin. **Consider oral prednisone 0.5–1 mg/kg for a 2–4 week taper as needed for severe flares and as a bridge to longer term therapy. Figure created with biorender.com
Based on available data, oral retinoids are a first-line therapeutic agent for DCS. Clinicians should note that systemic retinoids may require extended periods of treatment in some patients to demonstrate benefit. Optimal dosing regimen for isotretinoin for DCS still needs to be established. Oral retinoids could also potentially be combined with antibiotics to achieve a longer period of disease-free remission. Although isotretinoin was the most extensively studied oral retinoid for DCS in the literature, other options such as acitretin may merit further exploration, especially since acitretin has shown superior efficacy in HS compared with isotretinoin [54]. In addition, unlike HS, DCS predominantly affects men, which makes acitretin a viable treatment option for more patients with DCS, given its teratogenicity and long half-life. Acitretin also imparts a lower burden on prescribers in the USA, since it does not require enrollment in the iPLEDGE program [55]. Of note, tetracyclines and isotretinoin should not be used concomitantly due to the increased risk of pseudotumor cerebri.
For patients with DCS that is recalcitrant to oral antibiotics and retinoids, biologic agents are a long-term treatment option that should be considered. TNF-alpha antagonists such as ADA and IFX currently have the most, albeit also limited, data. Any patient who presents with concomitant moderate to severe HS should be offered a biologic agent if there are no contraindications. Our study found that nearly one-half of the 34 patients who received treatment with biologics for DCS also had HS. The concomitant use of biologics and retinoids merits further investigation along with the use of biologics in conjunction with surgical treatments, which has demonstrated benefit in HS [56]. Furthermore, larger studies on biologic use in DCS, including IL-17, IL-23, or IL-12/23 inhibitors, are warranted. For patients with recalcitrant DCS symptoms, oral steroids such as prednisone may be considered as a rescue treatment and as a bridge to other long-term treatment options such as biologics, retinoids, or surgical excision. Finasteride is an adjunct treatment that also merits further investigation in DCS.
Surgical excisions may be needed when a patient’s disease is refractory to medical management. No recurrence was observed in any of the patients treated with excisions and all patients had improvement in DCS lesions. Notably, excisions with and without skin grafts in the study had low rates of complications, although the sample size was small. More studies are warranted to investigate the optimal surgical approach to DCS.
Although intralesional steroid injections were only reported in 2 patients, this commonly used intervention is probably simply underreported in the literature. PDT and laser treatments may be useful non-invasive interventions for patients who fail medical management. Combining medical and non-invasive procedural treatments may lead to longer periods of remission, as demonstrated by Su et al.’s study on isotretinoin combined with PDT. Further studies on laser treatments are also needed to determine the optimal candidates for therapy and treatment frequency.
Lastly, it is essential to note that chronic inflammatory skin diseases such as DCS can impart a substantial psychosocial burden. The location of the abscesses, nodules, and fistulas of DCS on the face and scalp may predispose patients to social isolation, anxiety, and depression. Eliciting and addressing potential concerns regarding relationships and emotional well-being and referring patients to mental health experts when appropriate is warranted.
Our study contributes to the literature by providing an updated, comprehensive systematic review of existing literature on DCS treatments to guide clinicians in their discussions with patients. Study limitations include an overall small number of studies and patients for each type of intervention. There is also risk of reporting bias since the majority of studies were case reports or series. In addition, there was heterogeneity between studies in timepoints for efficacy measurement as well as outcome measurement. Therefore, the results of this study should be interpreted with caution.
Overall, clinicians and patients should be aware that data regarding available DCS treatment options are limited and largely based on case reports and case series. Randomized controlled trials and large cohort studies are needed to compare the efficacy and optimal dosing of different treatment regimens and aid in the development of evidence-based guidelines for DCS treatment. Further studies are needed to characterize the subsets of patients who would be most likely to benefit from specific treatment regimens. Moreover, standardized treatment outcomes will allow for more rigorous comparisons across studies. A potential new era of treatment for DCS is on the horizon with the recent advances in biologic and small molecule inhibitor therapies, but further investigation is needed.
Data Availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
Vasanth V, Chandrashekar BS. Follicular occlusion tetrad. Indian Dermatol Online J. 2014;5(4):491–3. https://doi.org/10.4103/2229-5178.142517.
Malviya N, Garg A. Comorbidities and Systemic Associations. In: A Comprehensive Guide to Hidradenitis Suppurativa. Elsevier; 2022:69–76. https://doi.org/10.1016/B978-0-323-77724-7.00008-5
Brănişteanu DE, Molodoi A, Ciobanu D, et al. The importance of histopathologic aspects in the diagnosis of dissecting cellulitis of the scalp. Rom J Morphol Embryol. 2009;50(4):719–724.
Badaoui A, Reygagne P, Cavelier-Balloy B, et al. Dissecting cellulitis of the scalp: a retrospective study of 51 patients and review of literature. Br J Dermatol. 2016;174(2):421–3. https://doi.org/10.1111/bjd.13999.
Higgins JPT, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. https://doi.org/10.1136/bmj.d5928
Wells G, Shea B, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097
Gamissans M, Romaní J, López-Llunell C, Riera-Martí N, Sin M. Dissecting cellulitis of the scalp: A review on clinical characteristics and management options in a series of 14 patients. Dermatol Ther. 2022;35(8):e15626. https://doi.org/10.1111/dth.15626
Karpouzis A, Giatromanolaki A, Sivridis E, Kouskoukis C. Perifolliculitis capitis abscedens et suffodiens successfully controlled with topical isotretinoin. Eur J Dermatol. 2003;13(2):192–5.
Navarro-Triviño FJ, Almazán-Fernández FM, Ródenas-Herranz T, Ruiz-Villaverde R. Dissecting cellulitis of the scalp successfully treated with topical resorcinol 15. Dermatol Ther. 2020;33(3):e13406. https://doi.org/10.1111/dth.13406
Scheinfeld N. Dissecting cellulitis (Perifolliculitis Capitis Abscedens et Suffodiens): a comprehensive review focusing on new treatments and findings of the last decade with commentary comparing the therapies and causes of dissecting cellulitis to hidradenitis suppurativa. Dermatol Online J. 2014;20(5):22692.
Abdennader S, Vignon-Pennamen MD, Hatchuel J, Reygagne P. Alopecic and aseptic nodules of the scalp (pseudocyst of the scalp): a prospective clinicopathological study of 15 cases. Dermatology. 2011;222(1):31–5. https://doi.org/10.1159/000321475.
Melo DF, Jorge Machado C, Bordignon NL, da Silva LL, Ramos PM. Lymecycline as a treatment option for dissecting cellulitis and folliculitis decalvans. Dermatol Ther. 2020;33(6):e14051. https://doi.org/10.1111/dth.14051
Chu S, Michelle L, Ekelem C, Sung CT, Rojek N, Mesinkovska NA. Oral isotretinoin for the treatment of dermatologic conditions other than acne: a systematic review and discussion of future directions. Arch Dermatol Res. 2021;313(6):391–430. https://doi.org/10.1007/s00403-020-02152-4.
Melo DF, Trüeb RM, Dutra H, Lima MMDA, Machado CJ, Dias MFRG. Low-dose isotretinoin as a therapeutic option for dissecting cellulitis. Dermatol Ther. 2020;33(6):e14273. https://doi.org/10.1111/dth.14273
Lee CN, Chen W, Hsu CK, Weng TT, Lee JYY, Yang CC. Dissecting folliculitis (dissecting cellulitis) of the scalp: a 66-patient case series and proposal of classification. J Dtsch Dermatol Ges. 2018;16(10):1219–26. https://doi.org/10.1111/ddg.13649.
Prasad SC, Bygum A. Successful treatment with alitretinoin of dissecting cellulitis of the scalp in keratitis-ichthyosis-deafness syndrome. Acta Derm Venereol. 2013;93(4):473–4. https://doi.org/10.2340/00015555-1499.
Vossen ARJV, Ardon CB, Zee HH, Lubberts E, Prens EP. The anti-inflammatory potency of biologics targeting tumour necrosis factor-α, interleukin ( IL )-17A, IL -12/23 and CD 20 in hidradenitis suppurativa: an ex vivo study. Br J Dermatol. 2019;181(2):314–23. https://doi.org/10.1111/bjd.17641.
Frechet L, Puzenat E, Charollais R, et al. Dissecting cellulitis of the scalp treated by tumour necrosis factor inhibitors: a case series. Eur J Dermatol. 2021;31(1):81–5. https://doi.org/10.1684/ejd.2021.3969.
Awad A, Sinclair R. Treatment of dissecting cellulitis of the scalp with Tildrakizumab. Australas J Dermatol. 2022;63(3):404–6. https://doi.org/10.1111/ajd.13860.
Babalola F, Chima M, Jobarteh R, Gottlieb A. Refractory Dissecting cellulitis of the scalp treated with risankizumab. J Drugs Dermatol. 2022;21(3):313–4. https://doi.org/10.36849/JDD.6699.
Muzumdar S, Parikh S, Strober B. Treatment of refractory dissecting cellulitis of the scalp with guselkumab: Case report. J Dermatol Dermatol Surg. 2020;24(1):52–3. https://doi.org/10.4103/jdds.jdds-50-19.
De Bedout V, Harper H, Miteva M, Lev-Tov H. Treatment dissecting cellulitis of the scalp with secukinumab. J Drugs Dermatol. 2021;20(7):776–7. https://doi.org/10.36849/JDD.5615.
Adrian RM, Arndt KA. Perifolliculitis capitis: successful control with alternate-day corticosteroids. Ann Plast Surg. 1980;4(2):166–9.
Segurado-Miravalles G, Camacho-Martínez FM, Arias-Santiago S, et al. Epidemiology, clinical presentation and therapeutic approach in a multicentre series of dissecting cellulitis of the scalp. J Eur Acad Dermatol Venereol. 2017;31(4):e199–200. https://doi.org/10.1111/jdv.13948.
Gupta M, Mahajan VK, Mehta KS, Chauhan PS. Zinc therapy in dermatology: A review. Dermatol Res Pract. 2014. https://doi.org/10.1155/2014/709152
Berne B, Venge P, Ohman S. Perifolliculitis capitis abscedens et suffodiens (Hoffman). Complete healing associated with oral zinc therapy. Arch Dermatol. 1985;121(8):1028–1030.
Kobayashi H, Aiba S, Tagami H. Successful treatment of dissecting cellulitis and acne conglobata with oral zinc. Br J Dermatol. 1999;141(6):1137–8. https://doi.org/10.1046/j.1365-2133.1999.03223.x.
Kurokawa I. Perifolliculitis capitis abscedens et suffodiens successfully treated with Saireito. J Dermatol. 2019;46(8):e299–301. https://doi.org/10.1111/1346-8138.14832.
Baneu NS, Bloancă VA, Szilagyi D, et al. Surgical management of dissecting cellulitis of the scalp using free latissimus dorsi flap and meshed split–thickness skin graft: A case report. Medicine. 2021;100(4):e24092. https://doi.org/10.1097/MD.0000000000024092
Dellon AL, Orlando JC. Perifolliculitis capitis: surgical treatment for the severe case. Ann Plast Surg. 1982;9(3):254–9. https://doi.org/10.1097/00000637-198209000-00012.
de Melo WC, Avci P, de Oliveira MN, et al. Photodynamic inactivation of biofilm: taking a lightly colored approach to stubborn infection. Expert Rev Anti Infect Ther. 2013;11(7):669–93. https://doi.org/10.1586/14787210.2013.811861.
Wu HE, Liu YB, Cui L, Xu GJ, Sun XD. A prospective study of the effect of fire micro-needling plus 5% topical ALA-PDT for the treatment of dissecting cellulitis of the scalp. Photodiagnosis Photodyn Ther. 2021;35:102396. https://doi.org/10.1016/j.pdpdt.2021.102396
Su Y, Xu Q, Zhang C, Zhang C. Photodynamic therapy pre-treated by fire needle combined with isotretinoin in the treatment of refractory perifolliculitis capitis abscedens et suffodiens: Case report. Photodiagnosis Photodyn Ther. 2021;33:102103. https://doi.org/10.1016/j.pdpdt.2020.102103
He J, Yang Y, Jin L, et al. 20% ALA-PDT pretreated by fire needle in patients with dissecting cellulitis of the scalp: An interim analysis. Photodiagnosis Photodyn Ther. 2021;36:102544. https://doi.org/10.1016/j.pdpdt.2021.102544
Scheinfeld NS. A case of dissecting cellulitis and a review of the literature. Dermatol Online J. 2003;9(1):8.
Krasner BD, Hamzavi FH, Murakawa GJ, Hamzavi IH. Dissecting cellulitis treated with the long-pulsed Nd:YAG laser. Dermatol Surg. 2006;32(8):1039–44. https://doi.org/10.1111/j.1524-4725.2006.32227.x.
Chui CT, Berger TG, Price VH, Zachary CB. Recalcitrant scarring follicular disorders treated by laser-assisted hair removal: a preliminary report. Dermatol Surg. 1999;25(1):34–7. https://doi.org/10.1046/j.1524-4725.1999.08100.x.
Xu M, Yan J. 2940-nm multifractional erbium:yttrium-aluminum-garnet laser for the treatment of dissecting cellulitis of the scalp: Case reports and literature review. J Dermatol. 2022;49(7):e230–1. https://doi.org/10.1111/1346-8138.16344.
Boyd AS, Binhlam JQ. Use of an 800-nm pulsed-diode laser in the treatment of recalcitrant dissecting cellulitis of the scalp. Arch Dermatol. 2002;138(10):1291–3. https://doi.org/10.1001/archderm.138.10.1291.
Glass LF, Berman B, Laub D. Treatment of perifolliculitis capitis abscedens et suffodiens with the carbon dioxide laser. J Dermatol Surg Oncol. 1989;15(6):673–6. https://doi.org/10.1111/j.1524-4725.1989.tb03608.x.
Chinnaiyan P, Tena LB, Brenner MJ, Welsh JS. Modern external beam radiation therapy for refractory dissecting cellulitis of the scalp. Br J Dermatol. 2005;152(4):777–9. https://doi.org/10.1111/j.1365-2133.2005.06536.x.
Paul S, Bach D, LeBoeuf NR, Devlin PM, Lipworth AD. Successful use of brachytherapy for a severe hidradenitis suppurativa variant. Dermatol Ther. 2016;29(6):455–8. https://doi.org/10.1111/dth.12393.
McMullan FH, Zeligman I. Perifolliculitis capitis abscedens et suffodiens; its successful treatment with x-ray epilation. AMA Arch Derm. 1956;73(3):256–63. https://doi.org/10.1001/archderm.1956.01550030058005.
MOYER DG, WILLIAMS RM. Perifolliculitis capitis abscedens et suffodiens. A report of six cases. Arch Dermatol. 1962;85:378–384. https://doi.org/10.1001/archderm.1962.01590030076010
Wasserman E. Perifolliculitis capitis abscedens et suffodiens with rheumatoid arthritis; report of a case. AMA Arch Derm Syphilol. 1951;64(6):787–9. https://doi.org/10.1001/archderm.1951.01570120122015.
Aboul-Fettouh N, Silapunt S, Mays S, Nguyen T. Intracavitary Foam Sclerotherapy in Dissecting Cellulitis of the Scalp. Dermatol Surg. 2021;47(9):1317–9. https://doi.org/10.1097/DSS.0000000000003035.
Asemota E, Chang YC, Goldberg LJ. Innovative management of recalcitrant dissecting cellulitis with compression therapy. JAMA Dermatol. 2016;152(11):1280–1. https://doi.org/10.1001/jamadermatol.2016.2743.
Díaz-Pérez LM, Escobar-Ramírez K, Sánchez-Dueñas LE. A new familial presentation of dissecting cellulitis: The genetic implications on scarring alopecias. JAAD Case Rep. 2020;6(8):705–7. https://doi.org/10.1016/j.jdcr.2020.05.027.
Shaffer N, Billick RC, Srolovitz H. Perifolliculitis capitis abscedens et suffodiens. Resolution with combination therapy. Arch Dermatol. 1992;128(10):1329–31.
Ross EK, Tan E, Shapiro J. Update on primary cicatricial alopecias. J Am Acad Dermatol. 2005;53(1):1–37; quiz 38–40. https://doi.org/10.1016/j.jaad.2004.06.015
Federico A, Rossi A, Caro G, et al. Are dissecting cellulitis and hidradenitis suppurativa different diseases? Clin Dermatol. 2021;39(3):496–9. https://doi.org/10.1016/j.clindermatol.2021.01.002.
Garg A, Kirby JS, Lavian J, Lin G, Strunk A. Sex- and Age-Adjusted Population Analysis of Prevalence Estimates for Hidradenitis Suppurativa in the United States. JAMA Dermatol. 2017;153(8):760–4. https://doi.org/10.1001/jamadermatol.2017.0201.
Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: A publication from the United States and Canadian Hidradenitis Suppurativa Foundations: Part II: Topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81(1):91–101. https://doi.org/10.1016/j.jaad.2019.02.068.
Dunn LK, Gaar LR, Yentzer BA, O’Neill JL, Feldman SR. Acitretin in dermatology: a review. J Drugs Dermatol. 2011;10(7):772–82.
Bechara FG, Podda M, Prens EP, et al. Efficacy and safety of adalimumab in conjunction with surgery in moderate to severe hidradenitis suppurativa: the SHARPS Randomized Clinical Trial. JAMA Surg. 2021;156(11):1001–9. https://doi.org/10.1001/jamasurg.2021.3655.
Jolliffe DS, Sarkany I. Perifolliculitis capitis abscedens et suffoidiens (dissecting cellulitis of the scalp). Clin Exp Dermatol. 1977;2(3):291–3. https://doi.org/10.1111/j.1365-2230.1977.tb02574.x.
Brook I. Recovery of anaerobic bacteria from a case of dissecting cellulitis. Int J Dermatol. 2006;45(2):168–9. https://doi.org/10.1111/j.1365-4632.2005.02594.x.
Cárdenas ML, López F, Moreno LH. Report of a patient with acne conglobata and perifolliculitis capitis abscedens et suffodiens. Colomb Med. 2011;42(2):224–7. https://doi.org/10.25100/cm.v42i2.775.
Koshelev MV, Garrison PA, Wright TS. Concurrent hidradenitis suppurativa, inflammatory acne, dissecting cellulitis of the scalp, and pyoderma gangrenosum in a 16-year-old boy. Pediatr Dermatol. 2014;31(1):e20-21. https://doi.org/10.1111/pde.12196.
Varshney N, Al Hammadi A, Sam H, Watters AK. Perifolliculitis capitis abscedens et suffodiens in an 18-year-old Aboriginal Canadian patient: case report and review of the literature. J Cutan Med Surg. 2007;11(1):35–9. https://doi.org/10.2310/7750.2007.00004.
CORMIE RL. Perifolliculitis capitis abscedens et suffodiens. Scott Med J. 1962;7:488–493. https://doi.org/10.1177/003693306200701105
GASNER WG. Perifolliculitis capitis abscedens et suffodiens; report of a case and response to therapy. N Y State J Med. 1957;57(5):947.
Greenblatt DT, Sheth N, Teixeira F. Dissecting cellulitis of the scalp responding to oral quinolones. Clin Exp Dermatol. 2008;33(1):99–100. https://doi.org/10.1111/j.1365-2230.2007.02598.x.
Onderdijk AJ, Boer J. Successful treatment of dissecting cellulitis with ciprofloxacin. Clin Exp Dermatol. 2009;34(7):e507. https://doi.org/10.1111/j.1365-2230.2009.03595.x
Ramesh V. Dissecting cellulitis of the scalp in 2 girls. Dermatologica. 1990;180(1):48–50. https://doi.org/10.1159/000247985.
Mittal RR, Gill SS, Jot T. Follicular occlusion triad. Indian J Dermatol Venereol Leprol. 1993;59(2):88–9.
Salim A, David J, Holder J. Dissecting cellulitis of the scalp with associated spondylarthropathy: case report and review. J Eur Acad Dermatol Venereol. 2003;17(6):689–91. https://doi.org/10.1046/j.1468-3083.2003.00731.x.
Scerri L, Williams HC, Allen BR. Dissecting cellulitis of the scalp: response to isotretinoin. Br J Dermatol. 1996;134(6):1105–8.
Bjellerup M, Wallengren J. Familial perifolliculitis capitis abscedens et suffodiens in two brothers successfully treated with isotretinoin. J Am Acad Dermatol. 1990;23(4 Pt 1):752–3. https://doi.org/10.1016/s0190-9622(08)81076-6.
Benvenuto ME, Rebora A. Fluctuant nodules and alopecia of the scalp. Perifolliculitis capitis abscedens et suffodiens. Arch Dermatol. 1992;128(8):1115–1117, 1118–1119. https://doi.org/10.1001/archderm.128.8.1115b
El Sayed F, Ammoury A, Dhaybi R, Aftimos G, Bazex J. Perifolliculitis capitis abscedens et suffodiens: an unusual case triggered by trauma. J Eur Acad Dermatol Venereol. 2006;20(9):1143–4. https://doi.org/10.1111/j.1468-3083.2006.01639.x.
Healsmith MF, Graham-Brown RAC. Treatment of perifolliculitis capitis abscedens et suffodiens with isotretinoin. J Dermatol Treat. 1992;3(1):27–8.
Khaled A, Zeglaoui F, Zoghlami A, Fazaa B, Kamoun MR. Dissecting cellulitis of the scalp: response to isotretinoin. J Eur Acad Dermatol Venereol. 2007;21(10):1430–1. https://doi.org/10.1111/j.1468-3083.2007.02239.x.
Koca R, Altinyazar HC, Ozen OI, Tekin NS. Dissecting cellulitis in a white male: response to isotretinoin. Int J Dermatol. 2002;41(8):509–13. https://doi.org/10.1046/j.1365-4362.2002.01552_3.x.
Libow LF, Friar DA. Arthropathy associated with cystic acne, hidradenitis suppurativa, and perifolliculitis capitis abscedens et suffodiens: treatment with isotretinoin. Cutis. 1999;64(2):87–90.
Marquis K, Christensen LC, Rajpara A. Dissecting Cellulitis of the Scalp with Excellent Response to Isotretinoin. Pediatr Dermatol. 2017;34(4):e210–1. https://doi.org/10.1111/pde.13143.
Melo DF, Lemes LR, Alves AL, Frattini SC, Obadia DL, Tortelly VD. Low-dose oral isotretinoin in a young female with dissecting cellulitis. Pediatr Dermatol. 2021;38(6):1573–4. https://doi.org/10.1111/pde.14829.
Mihić LL, Tomas D, Situm M, et al. Perifolliculitis capitis abscedens et suffodiens in a caucasian: diagnostic and therapeutic challenge. Acta Dermatovenerol Croat. 2011;19(2):98–102.
Mundi JP, Marmon S, Fischer M, Kamino H, Patel R, Shapiro J. Dissecting cellulitis of the scalp. Dermatol Online J. 2012;18(12):8.
Omulecki A, Dabkowski J, Zak-Prelich M. Perifolliculitis capitis abscedens et suffodiens–a case report. Med Sci Monit. 2000;6(3):602–4.
Ortiz-Prieto A, Pulpillo-Ruiz A, Conejo-Mir J. Dissecting cellulitis of the scalp. Actas Dermosifiliogr. 2017;108(8):778. https://doi.org/10.1016/j.ad.2016.04.025.
Scavo S, Lazzaro Danzuso G, La Rocca R, Magro G, Neri S. Isotretinoin in Perifolliculitis capitis abscedens et suffodiens: A case report and review of the literature. Clin Drug Invest. 2002;22(11):791–4. https://doi.org/10.2165/00044011-200222110-00007.
Schewach-Millet M, Ziv R, Shapira D. Perifolliculitis capitis abscedens et suffodiens treated with isotretinoin (13-cis-retinoic acid). J Am Acad Dermatol. 1986;15(6):1291–2. https://doi.org/10.1016/s0190-9622(86)80042-1.
Stites PC, Boyd AS. Dissecting cellulitis in a white male: a case report and review of the literature. Cutis. 2001;67(1):37–40.
Taylor AE. Dissecting cellulitis of the scalp: response to isotretinoin. Lancet. 1987;2(8552):225. https://doi.org/10.1016/s0140-6736(87)90818-x.
Tchernev G. Folliculitis et perifolliculitis capitis abscedens et suffodiens controlled with a combination therapy: Systemic antibiosis (Metronidazole Plus Clindamycin), dermatosurgical approach, and high-dose isotretinoin. Indian J Dermatol. 2011;56(3):318–20. https://doi.org/10.4103/0019-5154.82492.
Sand FL, Thomsen SF. Off-label use of TNF-alpha inhibitors in a dermatological university department: retrospective evaluation of 118 patients. Dermatol Ther. 2015;28(3):158–65. https://doi.org/10.1111/dth.12222.
Navarini AA, Trüeb RM. 3 cases of dissecting cellulitis of the scalp treated with adalimumab: control of inflammation within residual structural disease. Arch Dermatol. 2010;146(5):517–20. https://doi.org/10.1001/archdermatol.2010.16.
Alsantali A, Almalki B, Alharbi A. Recalcitrant dissecting cellulitis of the scalp treated successfully with adalimumab with hair regrowth: A case report. Clin Cosmet Invest Dermatol. 2021;14((Alsantali A., santli26@hotmail.com) Department of Dermatology, Hair Disorder Unit, King Fahad Armed Forces Hospital, Jeddah, Saudi Arabia):455–458. https://doi.org/10.2147/CCID.S301451
Kurokawa I. Perifolliculitis capitis abscedens et suffodiens with hidradenitis suppurativa and nodulocystic acne treated with adalimumab. J Dermatol. 2021;48(8):e374–5. https://doi.org/10.1111/1346-8138.15945.
Martin-García RF, Rullán JM. Refractory dissecting cellulitis of the scalp successfully controlled with adalimumab. P R Health Sci J. 2015;34(2):102–4.
Masnec IS, Franceschi N. Perifolliculitis capitis abscedens et suffodiens treated with anti-tumor necrosis factor-alpha – Possible new treatment option. Acta Dermatovenerol Croat. 2018;26(3):255–9.
Maxon E, Modlin K, Durso TA, Miletta NR. A case report of tumor necrosis factor alpha inhibitors in an active duty service member with dissecting cellulitis of the scalp resistant to treatment. Mil Med. 2020;185(7–8):e1309–11. https://doi.org/10.1093/milmed/usz394.
Minakawa S, Matsuzaki Y, Rokunohe D, et al. Hidradenitis suppurativa with perifolliculitis capitis abscedens et suffodiens successfully treated with a human anti-tumour necrosis factor monoclonal antibody. Clin Exp Dermatol. 2021;46(8):1586–8. https://doi.org/10.1111/ced.14789.
Sukhatme SV, Lenzy YM, Gottlieb AB. Refractory dissecting cellulitis of the scalp treated with adalimumab. J Drugs Dermatol. 2008;7(10):981–3.
Takahashi T, Yamasaki K, Terui H, et al. Perifolliculitis capitis abscedens et suffodiens treatment with tumor necrosis factor inhibitors: A case report and review of published cases. J Dermatol. 2019;46(9):802–7. https://doi.org/10.1111/1346-8138.14998.
Sanchez-Diaz M, Martinez-Lopez A, Salvador-Rodriguez L, Montero-Vilchez T, Arias-Santiago S, Molina-Leyva A. The role of biologic treatment in special scenarios in hidradenitis suppurativa: Facial and nape phenotype, dissecting cellulitis of the scalp, and lymphedema. Dermatol Ther. 2021;34(2):e14829. https://doi.org/10.1111/dth.14829
Brandt HRC, Malheiros APR, Teixeira MG, Machado MCR. Perifolliculitis capitis abscedens et suffodiens successfully controlled with infliximab. Br J Dermatol. 2008;159(2):506–7. https://doi.org/10.1111/j.1365-2133.2008.08674.x.
Syed TA, Asad ZUA, Salem G, Garg K, Rubin E, Agudelo N. Dissecting cellulitis of the scalp: A rare dermatological manifestation of Crohn’s disease. ACG Case Rep J. 2018;5(3). https://doi.org/10.14309/crj.2018.8
Wollina U, Gemmeke A, Koch A. Dissecting cellulitis of the scalp responding to intravenous tumor necrosis factor-alpha antagonist. J Clin Aesthetic Dermatol. 2012;5(4):36–9.
Powers MC, Mehta D, Ozog D. Cutting Out the Tracts: Staged Excisions for Dissecting Cellulitis of the Scalp. Dermatol Surg. 2017;43(5):738–40. https://doi.org/10.1097/DSS.0000000000001005.
Williams CN, Cohen M, Ronan SG, Lewandowski CA. Dissecting cellulitis of the scalp. Plast Reconstr Surg. 1986;77(3):378–82. https://doi.org/10.1097/00006534-198603000-00005.
Arneja JS, Vashi CN, Gursel E, Lelli JL. Management of fulminant dissecting cellulitis of the scalp in the pediatric population: Case report and literature review. Can J Plast Surg. 2007;15(4):211–4. https://doi.org/10.1177/229255030701500406.
Bachynsky T, Antonyshyn OM, Ross JB. Dissecting folliculitis of the scalp. A case report of combined treatment using tissue expansion, radical excision, and isotretinoin. J Dermatol Surg Oncol. 1992;18(10):877–880. https://doi.org/10.1111/j.1524-4725.1992.tb02920.x
Bellew SG, Nemerofsky R, Schwartz RA, Granick MS. Successful treatment of recalcitrant dissecting cellulitis of the scalp with complete scalp excision and split-thickness skin graft. Dermatol Surg. 2003;29(10):1068–70. https://doi.org/10.1046/j.1524-4725.2003.29305.x.
Housewright CD, Rensvold E, Tidwell J, Lynch D, Butler DF. Excisional surgery (scalpectomy) for dissecting cellulitis of the scalp. Dermatol Surg. 2011;37(8):1189–91. https://doi.org/10.1111/j.1524-4725.2011.02049.x.
Maintz L, Betz RC, Allam JP, et al. Keratitis-ichthyosis-deafness syndrome in association with follicular occlusion triad. Eur J Dermatol. 2005;15(5):347–52.
Moschella SL, Klein MH, Miller RJ. Perifolliculitis capitis abscedens et suffodiens. Report of a successful therapeutic scalping. Arch Dermatol. 1967;96(2):195–197.
Ramasastry SS, Granick MS, Boyd JB, Futrell JW. Severe perifolliculitis capitis with osteomyelitis. Ann Plast Surg. 1987;18(3):241–4. https://doi.org/10.1097/00000637-198703000-00011.
Nijhawan RI. Staged full-thickness excisions and porcine xenograft placement for extensive dissecting cellulitis of the scalp. Dermatol Surg. 2019;45(10):1324–7. https://doi.org/10.1097/DSS.0000000000001732.
Unal S, Unal S, Kuyucu S, Kuyucu N. Dissecting cellulitis of the scalp: A case report. Int Pediatr. 2004;19(2):103–5.
Feng Y, Zhang Y, Guo H, et al. Treatment of dissecting cellulitis of the scalp with 10% ALA-PDT. Lasers Surg Med. 2019;51(4):332–8. https://doi.org/10.1002/lsm.23016.
Zhang L, Wang P, Shi L, et al. Topical 5-aminolevulinic acid photodynamic therapy improved refractory acne conglobata and perifolliculitis capitis abscedens et suffodiens rather than hidradenitis suppurativa. J Innovative Opt Health Sci. 2016;9(1). https://doi.org/10.1142/S1793545816400022
Liu H, Ding A, Aloke G, et al. Surgical interventions combined with photodynamic therapy in the treatment of Perifolliculitis Capitis Abscendens et Suffodiens: Report of 5 cases. Photodiagnosis Photodyn Ther. 2020;30:101660. https://doi.org/10.1016/j.pdpdt.2020.101660
Feng H, Zhu C, Jin H. The efficacy and safety of 5-Aminolevulinic acid photodynamic therapy (ALA-PDT) as an adjunct therapy for symptoms in patients with dissecting cellulitis of the scalp: A retrospective study. Photodiagnosis Photodyn Ther. 2021;34:102322. https://doi.org/10.1016/j.pdpdt.2021.102322
Cui X, Zhu J, Yao X, Zhu W, Xu P, Wu X. Photodynamic therapy combined with dermatosurgical approach for Perifolliculitis Capitis Abscedens et Suffodiens. Photodiagnosis Photodyn Ther. 2020;30:101767. https://doi.org/10.1016/j.pdpdt.2020.101767
Liu Y, Ma Y, Xiang LH. Successful treatment of recalcitrant dissecting cellulitis of the scalp with ALA-PDT: case report and literature review. Photodiagnosis Photodyn Ther. 2013;10(4):410–3. https://doi.org/10.1016/j.pdpdt.2013.03.008.
Yan J, Zhang G, Liao C, Wang X, Shi L. ALA-iPDT for follicular occlusion tetrad concomitant with pachyonychia congenital type II and ankylosing spondylitis. Photodiagnosis Photodyn Ther. 2022;39:102891. https://doi.org/10.1016/j.pdpdt.2022.102891
Zhan Y, Chen X, Zhou Y, Zou P, Xiao R. Dissecting cellulitis of the scalp successfully treated with ALA-PDT: Case report. Photodiagnosis Photodyn Ther. 2018;24:182–4. https://doi.org/10.1016/j.pdpdt.2018.10.001.
Garelli V, Didona D, Paolino G, Didona B, Calvieri S, Rossi A. Dissecting cellulitis: responding to topical steroid and oral clindamycin. G Ital Dermatol Venereol. 2017;152(3):324–5. https://doi.org/10.23736/S0392-0488.16.05296-2.
Georgala S, Korfitis C, Ioannidou D, Alestas T, Kylafis G, Georgala C. Dissecting cellulitis of the scalp treated with rifampicin and isotretinoin: case reports. Cutis. 2008;82(3):195–8.
Ankad BS, Anusha HL, Nikam BP. Trichoscopic assessment of treatment response in dissecting cellulitis of scalp treated with combination therapy: A case report. Dermatol Ther. 2022;35(9):e15718. https://doi.org/10.1111/dth.15718
Bolz S, Jappe U, Hartschuh W. Successful treatment of perifolliculitis capitis abscedens et suffodiens with combined isotretinoin and dapsone. J Dtsch Dermatol Ges. 2008;6(1):44–7. https://doi.org/10.1111/j.1610-0387.2007.06399.x.
Donovan J. Dissecting cellulitis (DSC) after interferon beta-1a treatment and scalp trauma. JAAD Case Rep. 2015;1(5):303–4. https://doi.org/10.1016/j.jdcr.2015.07.003.
Goldsmith PC, Dowd PM. Successful therapy of the follicular occlusion triad in a young woman with high dose oral antiandrogens and minocycline. J R SOC MED. 1993;86(12):729–30.
Jacobs F, Metzler G, Kubiak J, Röcken M, Schaller M. New approach in combined therapy of perifolliculitis capitis abscedens et suffodiens. Acta Derm Venereol. 2011;91(6):726–7. https://doi.org/10.2340/00015555-1146.
Ljubojevic S, Pasic A, Lipozencic J, Skerlev M. Perifolliculitis capitis abscedens et suffodiens. J Eur Acad Dermatol Venereol. 2005;19(6):719–21. https://doi.org/10.1111/j.1468-3083.2005.01215.x.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Rahul Masson: drafting of the manuscript, checking data. Charlotte Y. Jeong: data acquisition, checking data, critical revision of the manuscript. Elaine Ma: data acquisition, checking data, critical revision of the manuscript. Natalie M. Fragoso: critical revision of the manuscript. Ashley B. Crew: critical revision of the manuscript. Vivian Y. Shi: critical revision of the manuscript. Jennifer L. Hsiao: concept and design, critical revision of the manuscript, supervision and administration.
Corresponding author
Ethics declarations
Conflict of Interest
Natalie M. Fragoso is an investigator for Acelyrin. Vivian Y. Shi is on the board of directors for the Hidradenitis Suppurativa Foundation (HSF), is an advisor for the National Eczema Association, is a stock shareholder of Learn Health, and has served as an advisory board member, investigator, speaker, and/or received research funding from Sanofi Genzyme, Regeneron, AbbVie, Genentech, Eli Lilly, Novartis, SUN Pharma, LEO Pharma, Pfizer, Incyte, Boehringer Ingelheim, Alumis Aristea Therapeutics, Menlo Therapeutics, Dermira, Burt’s Bees, Galderma, Kiniksa, UCB, Target-PharmaSolutions, Altus Lab/cQuell, MYOR, Polyfins Technology, GpSkin, and Skin Actives Scientific. Jennifer L. Hsiao is on the board of directors for the Hidradenitis Suppurativa Foundation, has served as a consultant for Aclaris, Boehringer Ingelheim, Novartis, and UCB, and has served as a consultant and speaker for AbbVie. Rahul Masson, Charlotte Y. Jeong, Elaine Ma, and Ashley B. Crew report no conflicts of interest.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Masson, R., Jeong, C.Y., Ma, E. et al. Treatments for Dissecting Cellulitis of the Scalp: A Systematic Review and Treatment Algorithm. Dermatol Ther (Heidelb) 13, 2487–2526 (2023). https://doi.org/10.1007/s13555-023-01018-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-023-01018-7