Abstract
Background/objective
Early detection and prompt treatment of GDM will go a long way in reducing the burden of T2DM. This study was undertaken to evaluate whether abnormal lipid profile including fasting as well as postprandial triglycerides, fasting total cholesterol, LDL cholesterol, and HDL cholesterol at 12 weeks of gestation (first trimester) may be used as simple predictive risk markers for developing GDM at a later stage.
Methods
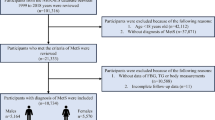
It was a prospective case-control study carried out in 45 subjects with GDM and selected equal number of age and BMI matched non-GDM subjects. A mixed meal consisting of two plain parathas and 20-g butter was given to all the subjects to evaluate 4-h triglyceride levels. Lipid levels were measured in all subjects at 11–13 weeks of gestation. At 24–28 weeks, all subjects were screened for GDM using a standardized oral glucose tolerance test.
Results
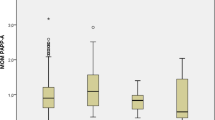
The study found significantly higher levels of fasting cholesterol, LDLc, and PPTg levels in subjects in their first trimester in those with GDM compared to those without GDM. Logistic regression analysis revealed higher odds of GDM with increasing 4-h PPTg levels {OR=1.01(1.001-1.02), p=0.02} and fasting cholesterol [OR=1.02 (1.001-1.03), p=0.001]. The ROC curve generated for the combined model of total cholesterol and PPTg has a higher area under the curve as compared to other parameters.
Conclusion
A combination of elevated fasting cholesterol and PPTg levels at an early stage of gestation can significantly predict future GDM.


Similar content being viewed by others
Data and resource sharing availability
Data will be available the reasonable request.
References
Metzger BE. International Association of Diabetes and Pregnancy Study Groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33(3):676–82.
Seshiah V, Balaji V, Balaji MS, Paneerselvam A, Kapur A. Pregnancy and diabetes scenario around the world: India. Int J Gynecol Obstet. 2009;104(SUPPL.).
Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27(5):1047–53.
Lee KW, Ching SM, Ramachandran V, Yee A, Hoo FK, Chia YC, Wan Sulaiman WA, Suppiah S, Mohamed MH, Veettil SK. Prevalence and risk factors of gestational diabetes mellitus in Asia: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2018;18(1):1–20.
Alia S, Pugnaloni S, Borroni F, Mazzanti L, Giannubilo SR, Ciavattini A, Vignini A. Impact of gestational diabetes mellitus in maternal and fetal health: An update. Diabetes Updates. 2019;5(3):1–6.
Lao TT, Ho LF, Chan BCP, Leung WC. Maternal age and prevalence of gestational diabetes mellitus [6]. Diabetes Care. 2006;29(4):948–9.
Yen IW, Lee CN, Lin MW, Fan KC, Wei JN, Chen KY, Chen SC, Tai YY, Kuo CH, Lin CH, Hsu CY, Chuang LM, Lin SY, Li HY. Overweight and obesity are associated with clustering of metabolic risk factors in early pregnancy and the risk of GDM. PLoS One. 2019;14(12):1–9.
Chan LY-S, Wong SF, Ho LC. Diabetic family history is an isolated risk factor for gestational diabetes after 30 years of age. Acta Obstet Gynecol Scand. 2002;81(2):115–7.
Carnethon MR, Palaniappan LP, Burchfiel CM, Brancati FL, Fortmann SP. Incidence of Type 2 Diabetes in Black and White Adults. Diabetes Care. 2002;25(8):1358–64.
Williams MA, Qiu C, Muy-Rivera M, Vadachkoria S, Song T, Luthy DA. Plasma adiponectin concentrations in early pregnancy and subsequent risk of gestational diabetes mellitus. J Clin Endocrinol Metab. 2004;89(5):2306–11.
Soheilykhah S, Mojibian M, Rahimi-Saghand S, Rashidi M, Hadinedoushan H. Maternal serum leptin concentration in gestational diabetes. Taiwan J Obstet Gynecol. 2011;50(2):149–53.
Veltman-Verhulst SM, van Haeften TW, Eijkemans MJC, de Valk HW, Fauser BCJM, Goverde AJ. Sex hormone-binding globulin concentrations before conception as a predictor for gestational diabetes in women with polycystic ovary syndrome. Human Reproduction (Oxford, England). 2010;25(12):3123–8.
Koukkou E, Watts GF, Lowy C. Serum lipid, lipoprotein and apolipoprotein changes in gestational diabetes mellitus: a cross-sectional and prospective study. J Clin Pathol. 1996;49(8):634–7.
Aslam M, Aggarwal S, Sharma KK, Galav V. Postprandial hypertriglyceridemia predicts development of insulin resistance glucose intolerance and type 2 diabetes. 2016;25(11(1)):1–15.
M. S, S.M. N, M. M, C.-M. M, N. S, K. N. First-trimester prediction of gestational diabetes mellitus: examining the potential of combining maternal characteristics and laboratory measures. Diabetes. 2010;59(12):3017–3022.
Kumru P, Arisoy R, Erdogdu E, Demirci O, Kavrut M, Ardıc C, Aslaner N, Ozkoral A, Ertekin A. Prediction of gestational diabetes mellitus at first trimester in low-risk pregnancies. Taiwan J Obstet Gynecol. 2016;55(6):815–20.
Wang J, Li Z, Lin L. Maternal lipid profiles in women with and without gestational diabetes mellitus. Medicine (United States). 2019;98(16).
Hollingsworth DR, Grundy SM. Dorothy r. hollingsworth and scott m. grundy. 1982;31(December):1092–1097.
Khan SH. Serum triglycerides and cholesterol status in patients with. 2017;(January).
K. R. B, S. V, R. P. S. A study of lipid parameters among GDM and non GDM pregnant women: a hospital based study. International Journal of Reproduction, Contraception, Obstet Gynecol. 2017;6(12):5488.
Quinlivan JA, Danielle L. Cholesterol abnormalities are common in women with prior gestational diabetes. J Diabetes Metab. 2013;04(04):4–7.
Madhu SV, Kant S, Srivastava S, Kant R, Sharma SB, Bhadoria DP. Postprandial lipaemia in patients with impaired fasting glucose, impaired glucose tolerance and diabetes mellitus. Diabetes Res Clin Pract. 2008;80(3):380–5.
Mishra BK, Velmurugan M, Gambhir JK, Madhu SV. Postprandial lipemia and its relation to TCF7L2 gene polymorphisms in normoglycemic first-degree relatives of type 2 diabetes patients. Int J Diabetes Dev Ctries. 2019;39:268–72.
Mishra BK, Banerjee BD, Agrawal V, Madhu SV. Association of PPARγ gene expression with postprandial hypertriglyceridaemia and risk of type 2 diabetes mellitus. Endocrine. 2020;68:549–56.
Enquobahrie DA, Williams MA, Qiu C, Luthy DA. Early pregnancy lipid concentrations and the risk of gestational diabetes mellitus. Diabetes Res Clin Pract. 2005;70(2):134–42.
Lai M, Fang F, Ma Y, Yang J, Huang J, Li N, Kang M, Xu X, Zhang J, Wang Y, Peng Y. Elevated midtrimester triglycerides as a biomarker for postpartum hyperglycemia in gestational diabetes. J Diabetes Res. 2020;2020:1–7.
Hernandez TL, Van Pelt RE, Anderson MA, Daniels LJ, West NA, Donahoo WT, et al. A higher-complex carbohydrate diet in gestational diabetes mellitus achieves glucose targets and lowers postprandial lipids: a randomized crossover study. Diabetes Care. 2014;37(5):1254–62.
Axelsen M, Smith U, Eriksson JW, Taskinen MR, Jansson PA. Postprandial hypertriglyceridemia and insulin resistance in normoglycemic first-degree relatives of patients with type 2 diabetes. Ann Intern Med. 1999;131(1):27–31.
Kumar V, Madhu SV, Singh G, Gambhir JK. Post-prandial hypertriglyceridemia in patients with type 2 diabetes mellitus with and without macrovascular disease. J Assoc Physicians India. 2010;58:603–7.
Madhu SV, Mittal V, Krishna Ram B, Srivastava DK. Postprandial lipid abnormalities in type 2 diabetes mellitus. J Assoc Physicians India. 2005;53:1043–6.
Li G, Kong L, Zhang L, Fan L, Su Y, Rose JC, Zhang W. Early pregnancy maternal lipid profiles and the risk of gestational diabetes mellitus stratified for body mass index. Reprod Sci. 2015;22(6):712–7.
Emet T, Ustüner I, Güven SG, Balık G, Ural UM, Tekin YB, et al. Plasma lipids and lipoproteins during pregnancy and related pregnancy outcomes. Arch Gynecol Obstet. 2013;288(1):49–55.
Rizzo M, Berneis K, Altinova AE, Toruner FB, Akturk M, Ayvaz G, Rini GB, Spinas GA, Arslan M. Atherogenic lipoprotein phenotype and LDL size and subclasses in women with gestational diabetes. Diabet Med. 2008;25(12):1406–11.
Sánchez-Vera I, Bonet B, Viana M, Quintanar A, Martín MD, Blanco P, Donnay S, Albi M. Changes in plasma lipids and increased low-density lipoprotein susceptibility to oxidation in pregnancies complicated by gestational diabetes: consequences of obesity. Metab Clin Exp. 2007;56(11):1527–33.
Chatuphonprasert W, Jarukamjorn K, Ellinger I. Physiology and pathophysiology of steroid biosynthesis, transport and metabolism in the human placenta. Front Pharmacol. 2018;9(SEP):1–29.
Medina-Gomez G, Gray SL, Yetukuri L, Shimomura K, Virtue S, Campbell M, Curtis RK, Jimenez-Linan M, Blount M, Yeo GSH, Lopez M, Seppänen-Laakso T, Ashcroft FM, Orešič M, Vidal-Puig A. PPAR gamma 2 prevents lipotoxicity by controlling adipose tissue expandability and peripheral lipid metabolism. PLoS Genet. 2007;3(4):e64.
Oh YS, Bae GD, Baek DJ, Park EY, Jun HS. Fatty acid-induced lipotoxicity in pancreatic beta-cells during development of type 2 diabetes. Front Endocrinol. 2018;9(JUL):1–10.
Herrera E, Lasunción MA, Gomez-Coronado D, Aranda P, López-Luna P, Maier I. Role of lipoprotein lipase activity on lipoprotein metabolism and the fate of circulating triglycerides in pregnancy. Am J Obstet Gynecol. 1988;158(6 Pt 2):1575–83.
Funding
This work was supported by University College of Medical Sciences IMRG grant-2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Ethical clearance was obtained from postgraduate review committee of University College of Medical Sciences and written consent was obtained from all the study participants.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Madhu, S.V., Bhardwaj, S., Mishra, B.K. et al. Total cholesterol and postprandial triglyceride levels as early markers of GDM in Asian Indian women. Int J Diabetes Dev Ctries 42, 630–635 (2022). https://doi.org/10.1007/s13410-022-01046-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-022-01046-7