Abstract
Background
The discovery of miRNAs as promising new biomarkers in the field of cardiovascular disease has caused great expectations. Stability in the bloodstream, specific regulation, and high sensitivity and specificity suggest that the feasibility of miRNAs as cardiovascular biomarkers can even displace protein-based biomarkers. The objective of our study was to determine the plasma expression profile of miRNAs, which are reported to have important correlations with cardiovascular diseases, in patients with type 2 diabetes mellitus in relation to cardiovascular complications.
Methods
We isolated plasma miRNAs from 6 patients with type 2 diabetes mellitus without cardiovascular disease (control group) and 9 patients with type 2 diabetes mellitus and cardiovascular disease (target group). Following reverse transcription and subsequent real-time PCR analysis of the same amount of starting miRNAs, the following miRNAs were successfully analyzed: miR-16-5p, miR-155-3p, miR-155-5p, miR-210, miR-221-3p, and miR-424-5p.
Results
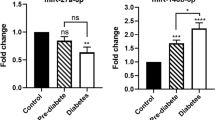
The relative expression analysis showed a statistically significant increase in the expression of all these miRNAs in the target group. The highest level of increase was established for miR-424-5p with an average relative quantification value of 10.5, followed by miR-155-5p with an average value of 7.5.
Conclusion
Cardiovascular risk assessment, supported by emerging circulating biomarkers, such as miRNAs, is important for stratifying high-risk individuals, optimizing treatment strategies, and enhancing our understanding of basic biology. Our study showed the highest increase in expression levels of miR-424-5p in target group and emphasized its role as a biomarker for cardiovascular damage in patients with type 2 diabetes mellitus.


Similar content being viewed by others
Availability of data and material
All data and material are available at the Molecular Medicine Centre, Medical University Sofia.
References
Cho NHSJ, Karuranga S, Huang Y, da Rocha Fernandes JD, Ohlrogge AW, Malanda B. IDF Diabetes Atlas: global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract. 2018;138:271–81.
Bulugahapitiya U, Siyambalapitiya S, Sithole J, Idris I. Is diabetes a coronary risk equivalent? Systematic review and meta-analysis. Diabet Med. 2009;26:142–8.
Dupont C, Armant DR, Brenner CA. Epigenetics: definition, mechanisms and clinical perspective. Semin Reprod Med. 2009;27:351–7.
Cannell IG, Kong YW, Bushell M. How do microRNAs regulate gene expression? Biochem Soc Trans. 2008;36:1224–31.
Ding Y, Sun X, Shan PF. MicroRNAs and cardiovascular disease in diabetes mellitus. Biomed Res Int. 2017;2017:1–8. https://doi.org/10.1155/2017/4080364.
Thomas MR, Lip GY. Novel risk markers and risk assessments for cardiovascular disease. Circ Res. 2017;120:133–49.
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci U S A. 2008;105:10513–8.
Jones Buie JN, Goodwin AJ, Cook JA, Halushka PV, Fan H. The role of miRNAs in cardiovascular disease risk factors. Atherosclerosis. 2016;254:271–81.
Arroyo JD, Chevillet JR, Kroh EM, Ruf IK, Pritchard CC, Gibson DF, et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc Natl Acad Sci U S A. 2011;108:5003–8.
Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9:654–9.
He Y, Ding Y, Liang B, et al. A systematic study of dysregulated microRNA in type 2 diabetes mellitus. In: Cho WC, editor. International journal of molecular sciences, 2017; 18(3): p. 456.
Belgardt BF, Ahmed K, Spranger M, Latreille M, Denzler R, Kondratiuk N, et al. The microRNA-200 family regulates pancreatic b-cell survival in type 2 diabetes. Nat Med. 2015;21:619e27.
Lin X, Guan H, Huang Z, Liu J, Li H, Wei G, et al. Downregulation of Bcl-2 expression by miR-34a mediates palmitate-induced Min6 cells apoptosis. J Diabetes Res. 2014;1729.
Zhang W, Xie HY, Ding SM, Xing CY, Chen A, Lai MC, et al. CADM1 regulates the G1/S transition and represses tumorigenicity through the Rb-E2F pathway in hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2016;15:289e96.
Zhu Y, You W, Wang H, Li Y, Qiao N, Shi Y, et al. MicroRNA-24/MODY generegulatory pathway mediates pancreatic b-cell dysfunction. Diabetes. 2013;62:3194e206.
Yang WM, Jeong HJ, Park SY, Lee W. Saturated fatty acid-induced miR-195 impairs insulin signaling and glycogen metabolism in HepG2 cells. FEBS Lett. 2014b;588:3939e46.
Nathan G, Kredo-Russo S, Geiger T, Lenz A, Kaspi H, Hornstein E, et al. MiR-375 promotes redifferentiation of adult human b-cells expanded in vitro. PLoS One. 2015;10:1729.
Shae A, Azarpira N, Karimi MH, Soleimani M, Dehghan S. Differentiation of human-induced pluripotent stem cells into insulin-producing clusters by microRNA-7. Exp Clin Transplant. 2015;16:121e8.
Gilbert ER, Liu D. Epigenetics: the missing link to understanding b-cell dysfunction in the pathogenesis of type 2 diabetes. Epigenetics. 2012;7:841e52.
Plaisance V, Waeber G, Regazzi R, Abderrahmani A. Role of microRNAs in islet b-cell compensation and failure during diabetes. J Diabetes Res. 2014;2014:618652.
Williams MD, Mitchell GM. MicroRNAs in insulin resistance and obesity. Exp Diabetes Res. 2012;2012:484696.
Nielsen S, Akerstrom T, Rinnov A, Yfanti C, Scheele C, Pedersen BK, et al. The miRNA plasma signature in response to acute aerobic exercise and endurance training. PLoS One. 2014;9:e87308.
Cakmak HA, Barman HA, Ikitimur B, Coskunpinar E, Oltulu YM, Can G, et al. The assessment of relationship between dysregulated microRNAs and left ventricular mass and mass index in systolic heart failure. J Am Coll Cardiol. 2013;62:C17–C.
Ali SS, Kala C, Abid M, Ahmad N, Sharma US, Khan NA. Pathological microRNAs in acute cardiovascular diseases and microRNA therapeutics. J Acute Dis. 2016;5:9–1.
Du F, Yu F, Wang Y, Hui Y, Carnevale K, Fu M, et al. MicroRNA-155 deficiency results in decreased macrophage inflammation and attenuated atherogenesis in apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2014;34:759–67.
Jamaluddin MS, Weakley SM, Zhang L, Kougias P, Lin PH, Yao Q, et al. miRNAs: roles and clinical applications in vascular disease. Expert Rev Mol Diagn. 2011;11:79–89.
Contu R, Latronico MV, Condorelli G. Circulating microRNAs as potential biomarkers of coronary artery disease: a promise to be fulfilled? Circ Res. 2010;107:573–4.
Silva GJJ, Bye A, El Azzouzi H, Wisloff U. MicroRNAs as important regulators of exercise adaptation. Prog Cardiovasc Dis. 2017;60:130–51.
Coskunpinar E, Cakmak HA, Kalkan AK, Tiryakioglu NO, Erturk M, Ongen Z. Circulating miR-221-3p as a novel marker for early prediction of acute myocardial infarction. Gene. 2016;591:90–6.
Zhou Y, Richards AM, Wang P. MicroRNA-221 is cardioprotective and anti-fibrotic in a rat model of myocardial infarction. Molecular Therapy Nucleic Acids. 2019;17:185–97.
Fasanaro P, D’Alessandra Y, Di Stefano V, Melchionna R, Romani S, Pompilio G, et al. MicroRNA-210 modulates endothelial cell response to hypoxia and inhibits the receptor tyrosine kinase ligand Ephrin-A3. J Biol Chem. 2008;283:15878–83.
Pulkkinen K, Malm T, Turunen M, Koistinaho J, Yla-Herttuala S. Hypoxia induces microRNA miR-210 in vitro and in vivo ephrin-A3 and neuronal pentraxin 1 are potentially regulated by miR-210. FEBS Lett. 2008;582:2397–401.
Bostjancic E, Zidar N, Glavac D. MicroRNA microarray expression profiling in human myocardial infarction. Dis Markers. 2009;27:255–68.
Greco S, Fasanaro P, Castelvecchio S, D'Alessandra Y, Arcelli D, Di Donato M, et al. MicroRNA dysregulation in diabetic ischemic heart failure patients. Diabetes. 2012;61:1633–41.
Sayed AS, Xia K, Li F, Deng X, Salma U, Li T, et al. The diagnostic value of circulating microRNAs for middle-aged (40-60-year-old) coronary artery disease patients. Clinics. 2015;70:257–63.
Guan Y, Song X, Sun W, Wang Y, Liu B. Effect of Hypoxia-Induced MicroRNA-210 Expression on cardiovascular disease and the underlying mechanism. Oxidative Med Cell Longev. 2019;2019:4727283.
Osipova J, Fischer DC, Dangwal S, Volkmann I, Widera C, Schwarz K, et al. Diabetes-associated microRNAs in pediatric patients with type 1 diabetes mellitus: a cross-sectional cohort study. J Clin Endocrinol Metab. 2014;99(9):E1661–5.
Amr KS, Abdelmawgoud H, Ali ZY, Shehata S, Raslan HM. Potential value of circulating microRNA-126 and microRNA-210 as biomarkers for type 2 diabetes with coronary artery disease. Br J Biomed Sci. 2018;75(2):82–7.
Ghosh G, Subramanian IV, Adhikari N, Zhang X, Joshi HP, Basi D, et al. Hypoxia-induced microRNA-424 expression in human endothelial cells regulates HIF-alpha isoforms and promotes angiogenesis. J Clin Invest. 2010;120:4141–54.
Kakimoto Y, Tanaka M, Hayashi H, Yokoyama K, Osawa M. Overexpression of miR-221 in sudden death with cardiac hypertrophy patients. Heliyon. 2018;4(6):e00639.
Funding
The study was supported by the Grant of Bulgarian National Scientific Foundation with no. КП-06-Н 33/10, 2019.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Methodology: Ivelina Mihaleva, Silva Kyurkchiyan, Rumyana Dodova, Ivanka Dimova. Formal analysis and investigation: Ivelina Mihaleva, Pavlina Gateva, Rumen Nikolov, Tsvetanka Markova, Ivanka Dimova. Writing—original draft preparation: Ivelina Mihaleva, Ivanka Dimova. Writing—review and editing: Pavlina Gateva, Rumen Nikolov, Tsvetanka Markova. Funding acquisition: Ivanka Dimova. Resources: Ivanka Dimova. Supervision: Ivanka Dimova
Corresponding author
Ethics declarations
Ethics approval
The collection of patients’ samples was approved by the institutional ethical committee (Medical University Sofia) with the approval no. 1209/2018. The research does not include animals.
Consent to participate
All participants in the study signed the informed consent before collection of the samples.
Consent for publication
It is included in the text of the informed consent signed by the patient.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mihaleva, I., Kyurkchiyan, S., Dodova, R. et al. MiRNA expression analysis emphasized the role of miR-424 in diabetic cardiovascular complications. Int J Diabetes Dev Ctries 41, 579–585 (2021). https://doi.org/10.1007/s13410-021-00934-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-021-00934-8