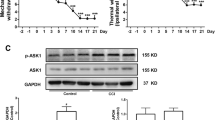
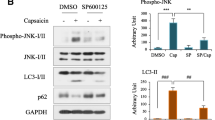
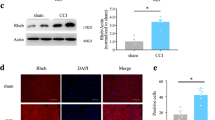
Abstract
Background
Neuropathic pain results from a lesion or disease of somatosensory system that causes pain, numbness, and weakness in human body. S-ketamine has anesthetic and analgesic potency and has been available for the treatment of chronic cancer pain, postoperative, and neuropathic pain. However, the protective mechanism of S-ketamine in neuropathic pain is not fully illustrated.
Objective
To investigate the protective mechanism of S-ketamine in neuropathic pain.
Results
Chronic constriction injury (CCI) caused the upregulation of Paw withdrawal threshold (PWT) and Thermal withdrawal latency (TWL), the induction in the expression of proinflammatory cytokines (IL-6, IL-1β, and TNF-α), and the accumulation of p62. S-ketamine administration significantly relieved the mechanical and thermal hyperalgesia and inflammatory reaction induced by CCI. In addition, S-ketamine increased the expressions of LC3II/LC3I and Beclin1, while decreased p62 expression in a concentration-dependent manner, indicating that S-ketamine promoted autophagy in the rat’s spinal cord after CCI treatment. Furthermore, CCI-caused upregulation in the expressions of p-PI3K, p-Akt and p-mTOR were reversed by S-ketamine administration.
Conclusion
S-ketamine could induce autophagy to alleviate neuropathic pain by inhibition of the PI3K/Akt/mTOR pathway. These findings facilitate understating on the molecular mechanism underlying the protective effects of S-ketamine on neuropathic pain.





Similar content being viewed by others
References
Andrade C (2017) Ketamine for depression, 3: does chirality matter? J Clin Psychiatry 78:e674–e677
Bennett GJ, Xie YK (1988) A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33:87–107
Berliocchi L et al (2015) Spinal autophagy is differently modulated in distinct mouse models of neuropathic pain. Mol Pain 11:3
Chang Y et al (2017) Ketamine, a Clinically Used Anesthetic, Inhibits Vascular Smooth Muscle Cell Proliferation via PP2A-Activated PI3K/Akt/ERK Inhibition. Int J Mol Sci 18:2545
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53:55–63
Colloca L et al (2017) Neuropathic pain. Nat Rev Dis Primers 3:17002
Falk E et al (2020) A rapid positive influence of S-ketamine on the anxiety of patients in palliative care: a retrospective pilot study. BMC Palliat Care 19:1
Fan JC, Song JJ, Wang Y, Chen Y, Hong DX (2017) Neuron-protective effect of subanesthestic-dosage ketamine on mice of Parkinson’s disease. Asian Pac J Trop Med 10:1007–1010
Gottlieb RA, Andres AM, Sin J, Taylor DP (2015) Untangling autophagy measurements: all fluxed up. Circ Res 116:504–514
Guo JS et al (2015) Increased autophagic activity in dorsal root ganglion attenuates neuropathic pain following peripheral nerve injury. Neurosci Lett 599:158–163
Guo X, Cheng L, Yang S, Che H (2020) Pro-inflammatory immunological effects of adipose tissue and risk of food allergy in obesity: focus on immunological mechanisms. Allergol Immunopathol 48:306–312
Heras-Sandoval D, Pérez-Rojas JM, Hernández-Damián J, Pedraza-Chaverri J (2014) The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal 26:2694–2701
Kim SY et al (2021) Coptisine induces autophagic cell death through down-regulation of PI3K/Akt/mTOR signaling pathway and up-regulation of ROS-mediated mitochondrial dysfunction in hepatocellular carcinoma Hep3B cells. Arch Biochem Biophys 697:108688
Komatsu M et al (2010) The selective autophagy substrate p62 activates the stress responsive transcription factor Nrf2 through inactivation of Keap1. Nat Cell Biol 12:213–223
Li J et al (2021) Combination of autophagy and NFE2L2/NRF2 activation as a treatment approach for neuropathic pain. Autophagy. https://doi.org/10.1080/15548627.2021.1900498
Liu WJ et al (2016) p62 links the autophagy pathway and the ubiqutin-proteasome system upon ubiquitinated protein degradation. Cell Mol Biol Lett 21:29
Liu X, Zhu M, Ju Y, Li A, Sun X (2019a) Autophagy dysfunction in neuropathic pain. Neuropeptides 75:41–48
Liu YD, Wang ZB, Han G, Jin L, Zhao P (2019b) Hyperbaric oxygen relieves neuropathic pain through AKT/TSC2/mTOR pathway activity to induce autophagy. J Pain Res 12:443–451
Meng XL et al (2020) Increased EZH2 levels in anterior cingulate cortex microglia aggravate neuropathic pain by inhibiting autophagy following brachial plexus avulsion in rats. Neurosci Bull 36:793–805
Mion G, Villevieille T (2013) Ketamine pharmacology: an update (pharmacodynamics and molecular aspects, recent findings). CNS Neurosci Ther 19:370–380
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147:728–741
Moscat J, Diaz-Meco MT (2009) p62 at the crossroads of autophagy, apoptosis, and cancer. Cell 137:1001–1004
Nielsen RV, Fomsgaard JS, Nikolajsen L, Dahl JB, Mathiesen O (2019) Intraoperative S-ketamine for the reduction of opioid consumption and pain one year after spine surgery: a randomized clinical trial of opioid-dependent patients. Eur J Pain (lond, Engl) 23:455–460
O’Connor AB, Dworkin RH (2009) Treatment of neuropathic pain: an overview of recent guidelines. Am J Med 122:S22-32
Rosenberger DC, Blechschmidt V, Timmerman H, Wolff A, Treede RD (2020) Challenges of neuropathic pain: focus on diabetic neuropathy. J Neural Transm (vienna, Austria: 1996) 127:589–624
Silva RL, Lopes AH, Guimarães RM, Cunha TM (2017) CXCL1/CXCR2 signaling in pathological pain: Role in peripheral and central sensitization. Neurobiol Dis 105:109–116
Sommer C, Leinders M, Üçeyler N (2018) Inflammation in the pathophysiology of neuropathic pain. Pain 159:595–602
Tang C, Livingston MJ, Liu Z, Dong Z (2020) Autophagy in kidney homeostasis and disease. Nat Rev Nephrol 16:489–508
Trimmel H et al (2018) S(+)-ketamine: current trends in emergency and intensive care medicine. Wien Klin Wochenschr 130:356–366
Wang X, Lin C, Lan L, Liu J (2021) Perioperative intravenous S-ketamine for acute postoperative pain in adults: a systematic review and meta-analysis. J Clin Anesth 68:110071
Wu J et al (2018) Licochalcone A suppresses hexokinase 2-mediated tumor glycolysis in gastric cancer via downregulation of the Akt signaling pathway. Oncol Rep 39:1181–1190
Xia H, Green DR, Zou W (2021) Autophagy in tumour immunity and therapy. Nat Rev Cancer 21:281–297
Xiang H et al (2020) Targeting autophagy-related protein kinases for potential therapeutic purpose. Acta Pharm Sin B 10:569–581
Xu HD, Qin ZH (2019) Beclin 1, Bcl-2 and autophagy. Adv Exp Med Biol 1206:109–126
Xu W et al (2021) S-ketamine alleviates carbon tetrachloride-induced hepatic injury and oxidative stress by targeting the Nrf2/HO-1 signaling pathway. Can J Physiol Pharmacol 99:1308–1315
Zhao S, Shao L, Wang Y, Meng Q, Yu J (2020) Ketamine exhibits anti-gastric cancer activity via induction of apoptosis and attenuation of PI3K/Akt/mTOR. Arch Med Sci 16:1140–1149
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
JH and XZ designed the experiments and LX carried them out. OL analyzed and interpreted the data, QL prepared the manuscript with contributions from all co-authors.
Corresponding author
Ethics declarations
Conflict of interest
Author Jia Han declares that he/she has no conflict of interest; author Xianjie Zhang declares that he/she has no conflict of interest; author Leqiang Xia declares that he/she has no conflict of interest; author Ou Liao declares that he/she has no conflict of interest; author Qiulan Li declares that he/she has no conflict of interest.
Ethical approval
Ethical approval was obtained from the Ethics Committee of People's Hospital of Deyang City.
Statement of informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Han, J., Zhang, X., Xia, L. et al. S-ketamine promotes autophagy and alleviates neuropathic pain by inhibiting PI3K/Akt/mTOR signaling pathway. Mol. Cell. Toxicol. 19, 81–88 (2023). https://doi.org/10.1007/s13273-022-00243-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-022-00243-z