Abstract
Objective
There is a paucity of information on bone scanning for prostate cancer from low-resource countries. This study evaluated the role of bone scan in the primary staging of newly diagnosed prostate cancer in one such setting.
Methods
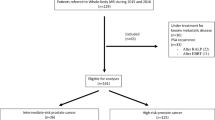
A retrospective analysis of 126 men with newly diagnosed prostate cancer undergoing an initial staging bone scan between January 2017 and December 2020 was carried out at a regional nuclear medicine center in Nigeria. Bone scan results were analyzed according to age, serum level of baseline prostate-specific antigen (PSA), and Gleason score. Equivocal scans and patients with no Gleason score or baseline PSA were excluded from the analysis. p < 0.05 was said to be significant statistically.
Results
Of 111 patients (aged 38–84 years, median 66 years), who met the inclusion criteria, 26 (23%) men had evidence of bony metastases as shown by a positive bone scan. Higher PSA levels and Gleason scores were associated with an increased risk of a positive bone scan, p < 0.001. No patient with a PSA level < 20 ng/mL and a Gleason score of < 7 had a positive bone scan.
Conclusion
The role of bone scanning in staging newly diagnosed prostate cancer patients in Nigeria is consistent with global reports. Our study confirms that a bone scan finding is well associated with the risk classification using PSA and Gleason score in our population.
Similar content being viewed by others
Data Availability
The study data is available from the records of the Department of Nuclear Medicine and the Urology Division of the Department of Surgery at the University College Hospital (UCH) Ibadan, Nigeria. Please contact author for data set requests.
References
Seraphin TP, Joko-Fru WY, Manraj SS, Chokunonga E, Somdyala NIM, Korir A, et al. Prostate cancer survival in sub-Saharan Africa by age, stage at diagnosis, and human development index: a population-based registry study. Cancer Causes Control. 2021. https://doi.org/10.1007/s10552-021-01453-X.
Powell IJ, Bock CH, Ruterbusch JJ, Sakr W. Evidence supports a faster growth rate and/or earlier transformation to clinically significant prostate cancer in black than in white American men, and influences racial progression and mortality disparity. J Urol. 2010;183:1792–7.
Taitt HE. Global trends and prostate cancer: a review of incidence, detection, and mortality as influenced by race, ethnicity, and geographic location. Am J Mens Health. 2018;12:1807–23.
Agalliu I, Adebiyi AO, Lounsbury DW, Popoola O, Jinadu K, Amodu O, et al. The feasibility of epidemiological research on prostate cancer in African men in Ibadan. Nigeria BMC Public Health. 2015;15:425.
Olapade-Olaopa EO, Obamuyide HA, Yisa GT. Management of advanced prostate cancer in Africa. Can J Urol. 2008;15:3890–8.
Ikuerowo SO, Omisanjo OA, Bioku MJ, Ajala MO, Nonyelim VP, Esho JO. Prevalence and characteristics of prostate cancer among participants of a communitybased screening in Nigeria using serum prostate specific antigen and digital rectal examination. Pan Afr Med J. 2013;15:129.
Bello JO, Buhari T, Mohammed TO, Olanipekun HB, Eguniwe AM, Fasiku OK, et al. Determinants of prostate specific antigen screening test uptake in an urban community in North-Central Nigeria. Afr Health Sci. 2019;19:1665–70.
Okolo CA, Akinosun OM, Shittu OB, Olapade-Olaopa EO, Okeke LI, Akang EEU, et al. Correlation of serum PSA and Gleason score in Nigerian men with prostate cancer. Afr J Urol. 2008;14:15–22.
Leng J, Ntekim AI, Ibraheem A, Anakwenze CP, Golden DW, Olopade OI. Infrastructural challenges lead to delay of curative radiotherapy in Nigeria. JCO Glob Oncol. 2020;6:269–76.
Cassell A, Yunusa B, Jalloh M, Ndoye M, Mbodji MM, Diallo A, et al. Management of advanced and metastatic prostate cancer: a need for a Sub-Saharan guideline. J Oncol. 2019;2019:1–9.
Folasire A, Ntekim A, Omikunle A, Ali-Gombe A. Association of Gleason risk groups with metastatic sites in prostate cancer. Afr J Biomed Res. 2015;18:189–96.
Qureshi A, Makhdomi K, Stones W. Prostate-specific antigen as a risk factor for skeletal metastasis in native ethnic African Men with prostate cancer: a case-control study. World J Nucl Med. 2017;16:32.
Sarkar S, Das S. A review of imaging methods for prostate cancer detection. Biomed Eng Comput Biol. 2016;7s1:1–15.
Ogbole GI, Adeyomoye AO, Badu-Peprah A, Mensah Y, Nzeh DA. Survey of magnetic resonance imaging availability in West Africa. Pan Afr Med J. 2018;30:240.
Orunmuyi AT, Lawal IO, Omofuma OO, Taiwo OJ, Sathekge MM. Underutilisation of nuclear medicine scans at a regional hospital in Nigeria: need for implementation research. Ecancermedicalscience. 2020;14:1093.
Donohoe KJ, Cohen EJ, Giammarile F, Grady E, Greenspan BS, Henkin RE, et al. Appropriate use criteria for bone scintigraphy in prostate and breast cancer. J Nucl Med. 2017;58:14N-17N.
Suh CH, Shinagare AB, Westenfield AM, Ramaiya NH, Van den Abbeele AD, Kim KW. Yield of bone scintigraphy for the detection of metastatic disease in treatment-naive prostate cancer: a systematic review and meta-analysis. Clin Radiol. 2018;73:158–67.
Merdan S, Womble PR, Miller DC, Barnett C, Ye Z, Linsell SM, et al. Toward better use of bone scans among men with early-stage prostate cancer. Urology. 2014;84:793.
Al-Ghazo MA, Ghalayini IF, Al-Azab RS, Al-Azab RS, Bani-Hani I, Barham A, et al. Do all patients with newly diagnosed prostate cancer need staging radionuclide bone scan? A retrospective study. Int Braz J Urol. 2010;36:685–91.
Amoako Y, Hammond EB, Assasie-Gyimah A, Laryea DO, Ankrah A, Amoah G. Prostate-specific antigen and risk of bone metastases in west Africans with prostate cancer. World J Nucl Med. 2019;18:148.
Tanaka N, Fujimoto K, Shinkai T, Nakai Y, Kuwada M, Anai S, et al. Bone Scan can be spared in asymptomatic prostate cancer patients with PSA of ≤ 20 ng/ml and Gleason score of ≤ 6 at the initial stage of diagnosis. Jpn J Clin Oncol. 2011;41:1209–13.
Sevcenco S, Grubmüller B, Sonneck-Koenne C, Ahmadi Y, Knoll P, Floth A, et al. Bone scintigraphy in staging of newly diagnosed prostate cancer in regard of different risk groups. Asia Ocean J Nucl Med Biol. 2019;7:149.
Lin Y, Mao Q, Chen B, Wang L, Liu B, Zheng X, et al. When to perform bone scintigraphy in patients with newly diagnosed prostate cancer? a retrospective study. BMC Urol. 2017;17:41.
Badmus TA, Adesunkanmi ARK, Yusuf BM, Oseni GO, Eziyi AK, Bakare TIB, et al. Burden of prostate cancer in southwestern nigeria. Urology. 2010;76:412–6.
Ekeke O, Amusan O, Eke N. Management of prostate cancer in port harcourt, Nigeria: changing patterns. J West Afr Coll Surg. 2012;2:58–77.
Taiwo OJ, Orunmuyi AT. Maximal covering location problem for nuclear medicine clinics allocation in Nigeria. Spat Inf Res. 2021;29:919–935. https://doi.org/10.1007/s41324-021-00405-6.
Zacho HHD, Barsi T, Mortensen JJC, Mogensen MK, Bertelsen H, Jospehsen N, et al. Prospective multicenter study of bone scintigraphy in consecutive patients with newly diagnosed prostate cancer. Clin Nucl Med. 2014;39:26–31.
Heidenreich A, Aus G, Bolla M, Joniau S, Matveev VB, Schmid HP, Zattoni F, et al. EAU guidelines on prostate cancer. Eur Urol. 2008;53:68–80.
Author information
Authors and Affiliations
Contributions
The study was designed by Akintunde Taiwo Orunmuyi. Material preparation and data collection were performed by Akintunde T Orunmuyi, Augustine O Takure, Sikiru A Adebayo, and E.Oluwabunmi Olapade-Olaopa. The data analysis was performed by Olayinka Stephen Ilesanmi and Akintunde Taiwo Orunmuyi. The first draft of the manuscript was written by Akintunde Taiwo Orunmuyi (Introduction, Methods, Results, Discussion), Olayinka Stephen Ilesanmi (Methods, Results, Discussion), and E.Oluwabunmi Olapade-Olaopa (Introduction and Discussion). All authors commented on previous versions of the manuscript and all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Akintunde Orunmuyi, Sikiru Adebayo, Olayinka Ilesanmi, Augustine Takure, and E.Oluwabunmi Olapade-Olaopa declare that they have no conflict of interest.
Ethics Approval and Consent to Participate
The University of Ibadan/University College Hospital (UI/UCH) ethics committee approved this study (Ethics Number UI/EC/20/0198) and waived the need for patients’ consent due to the retrospective design of this study. All bone scans were performed and utilized as part of the routine clinical management of prostate cancer by the referring clinician; no patient was re-injected or re-imaged for the purpose of this study. The authors certify that the study was performed in accordance with the ethical standards as laid down in the Helsinki declaration as revised in 2013 and its later amendments.
Consent for Publication
Not applicable as the manuscript does not contain any data that can be identified to any individual person.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Orunmuyi, A.T., Adebayo, S.A., Ilesanmi, O.S. et al. A Bone Scan Is Valuable for Primary Staging of Newly Diagnosed Prostate Cancer in a Low-Resource Setting (Nigeria). Nucl Med Mol Imaging 56, 96–101 (2022). https://doi.org/10.1007/s13139-021-00735-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-021-00735-1