Abstract
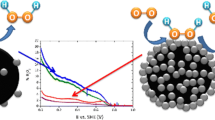
This study intends to provide some insight in the up-to-date elusive assessment of a correct choice of method for estimating the active surface area of Pt alloy nanoparticle catalysts. Taking PtNi3 nanoparticles as an example, we have compared three types of electrochemically active surface area (ECSA) data, CO-ECSA, Hupd-ECSA, and Hupd/CO-ECSA, which were evaluated from CO stripping and underpotentially deposited hydrogen stripping steps applied at different times along a reference catalyst activity test protocol. Considering a total of six different detailed voltammetric test protocols, we address Pt alloy particle size effects, analyze the effect of the time of application of CO and hydrogen stripping, and study their effect on the Pt mass and Pt surface-specific activities for the oxygen reduction reaction (ORR). In a discussion of the ratio of CO charge to hydrogen charge, it is shown that this quantity is more complex than previously thought and not associated with a specific surface structure. The Hupd/CO-ECSA data are found to be a reasonable balance for the estimate of surface area normalized, so-called specific catalytic ORR activities.







Similar content being viewed by others
References
H.A. Gasteiger, S.S. Kocha, B. Sompalli, F.T. Wagner, Activity benchmarks and requirements for Pt, Pt-alloy, and non-Pt oxygen reduction catalysts for PEMFC. Appl. Catal. B Environ. 56, 9–35 (2005)
S.K. Meher, G.R. Rao, Morphology-controlled promoting activity of nanostructured MnO2 for methanol and ethanol electrooxidation on Pt/C. J. Phys. Chem. C 117, 4888–4900 (2013)
N.M. Markovic, H.A. Gasteiger, P.N. Ross Jr., X. Jiang, I. Villegas, M.J. Weaver, Electro-oxidation mechanisms of methanol and formic acid on Pt-Ru alloy surfaces. Electrochim. Acta 40, 91–98 (1995)
H. Lee, S.E. Habas, G.A. Somorjai, P. Yang, Localized Pd overgrowth on cubic Pt nanocrystals for enhanced electrocatalytic oxidation of formic acid. J. Am. Chem. Soc. 130, 5406–5407 (2008)
C. Cui, L. Gan, M. Heggen, S. Rudi, P. Strasser, Compositional segregation in shaped Pt alloy nanoparticles and their structural behavior during electrocatalysis. Nat. Mater. 12, 765–771 (2013a)
D.F. van der Vliet, C. Wang, D. Li, A.P. Paulikas, J. Greeley, R.B. Rankin, D. Strmcnik, D. Tripkovic, N.M. Markovic, V.R. Stamenkovic, Unique electrochemical adsorption properties of Pt-skin surfaces. Angew. Chem. 124, 3193–3196 (2012)
K.J.J. Mayrhofer, D. Strmcnik, B.B. Blizanac, V. Stamenkovic, M. Arenz, N.M. Markovic, Measurement of oxygen reduction activities via the rotating disc electrode method: From Pt model surfaces to carbon-supported high surface area catalysts. Electrochim. Acta 53, 3181–3188 (2008)
M. Shao, J.H. Odell, S.-I. Choi, Y. Xia, Electrochemical surface area measurements of platinum- and palladium-based nanoparticles. Electrochem. Commun. 31, 46–48 (2013)
M. Fallavier, B. Hjörvarsson, M. Benmansour, J.P. Thomas, Determination of hydrogen surface coverage of Pt0.5Ni0.5 single crystals by NRA. Nucl Instrum Methods Phys Res Sect B Beam Interact Mater Atoms 64, 83–87 (1992)
M. Fallavier, M. Benmansour, B. Hjörvarsson, J.P. Thomas, M. Abon, H.A. Atli, J.C. Bertolini, Hydrogen on Pt0.5Ni0.5(110), alloying effects upon adsorption. Surf. Sci. 311, 24–32 (1994)
V. Stamenkovic, B.S. Mun, K.J.J. Mayrhofer, P.N. Ross, N.M. Markovic, J. Rossmeisl, J. Greeley, J.K. Nørskov, Changing the activity of electrocatalysts for oxygen reduction by tuning the surface electronic structure. Angew. Chem. Int. Ed. 45, 2897–2901 (2006)
V.R. Stamenkovic, B. Fowler, B.S. Mun, G. Wang, P.N. Ross, C.A. Lucas, N.M. Markovic, Improved oxygen reduction activity on Pt3Ni(111) via increased surface site availability. Science 315, 493–497 (2007)
L. Gan, M. Heggen, S. Rudi, P. Strasser, Core-shell compositional fine structures of dealloyed PtxNi1-x nanoparticles and their impact on oxygen reduction catalysis. Nano Lett 12, 5423–5430 (2012)
S. Rudi, X. Tuaev, P. Strasser, Electrocatalytic oxygen reduction on dealloyed Pt1-xNix alloy nanoparticle electrocatalysts. Electrocatalysis 3, 265–273 (2012)
L. Gan, P. Strasser, in Electrocatalysis for fuel cells: A non and low platinum approach, ed. by M. Shao (Springer, London, 2013), pp. 533–560
L. Gan, M. Heggen, R. O'Malley, B. Theobald, P. Strasser, Understanding and controlling nanoporosity formation for improving the stability of bimetallic fuel cell catalysts. Nano Lett. 13, 1131–1138 (2013)
L. Gan, C. Cui, S. Rudi, P. Strasser, Core-shell and nanoporous particle architectures and their effect on the activity and stability of Pt ORR electrocatalysts. Top. Catal. in press (2013).
M. Oezaslan, P. Strasser, Activity of dealloyed PtCo3 and PtCu3 nanoparticle electrocatalyst for oxygen reduction reaction in polymer electrolyte membrane fuel cell. J. Power Sources 196, 5240–5249 (2011)
M. Oezaslan, F. Hasché, P. Strasser, In situ observation of bimetallic alloy nanoparticle formation and growth using high-temperature XRD. Chem. Mater. 23, 2159–2165 (2011)
F. Hasché, M. Oezaslan, P. Strasser, Activity, stability, and degradation mechanisms of dealloyed PtCu3 and PtCo3 nanoparticle fuel cell catalysts. ChemCatChem 3, 1805–1813 (2011)
P. Strasser, Dealloyed core-shell fuel cell electrocatalysts. Rev. Chem. Eng. 25, 255–295 (2009)
C. Cui, M. Ahmadi, F. Behafarid, L. Gan, M. Neumann, M. Heggen, B.R. Cuenya, P. Strasser, Shape-selected bimetallic nanoparticle electrocatalysts: Evolution of their atomic-scale structure, chemical composition, and electrochemical reactivity under various chemical environments. Faraday Disc 162, 91–112 (2013b)
M. Ahmadi, F. Behafarid, C.H. Cui, P. Strasser, B.R. Cuenya, Long-range segregation phenomena in shape-selected bimetallic nanoparticles: Chemical state effects. ACS Nano 7, 9195–9204 (2013)
Subbaraman, Enhancing hydrogen evolution acivity in water splitting by tailoring Li-Ni(OH)2-Pt interfaces. Science 334, 1256–1260 (2012)
R. Subbaraman, D. Tripkovic, K.C. Chang, D. Strmcnik, A.P. Paulikas, P. Hirunsit, M. Chan, J. Greeley, V. Stamenkovic, N.M. Markovic, Trends in activity for the water electrolyser reactions on 3d M(Ni,Co,Fe,Mn) hydr(oxy)oxide catalysts. Nat. Mater. 11, 550–557 (2012)
Acknowledgments
We thank S. Selve and the ZELMI department for the TEM images. We thank A. Wittebrock for her great help with the electrochemical measurements. This work was supported by US DOE EERE award DE-EE0000458 via subcontract through General Motors. The project also received funds from the Federal Ministry of Education and Research under the project reference number 16 N11929. Responsibility for the contents of this publication lies with the author.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rudi, S., Cui, C., Gan, L. et al. Comparative Study of the Electrocatalytically Active Surface Areas (ECSAs) of Pt Alloy Nanoparticles Evaluated by Hupd and CO-stripping voltammetry. Electrocatalysis 5, 408–418 (2014). https://doi.org/10.1007/s12678-014-0205-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-014-0205-2