Abstract
Background
Bone grafting is a widely used technique for repairing and regenerating defective bones. However, the use of bone graft substitutes is limited due to various constraints, including contamination and cost. To address these limitations, innovative antimicrobial bone replacement biomaterials have been developed.
Materials and Methods
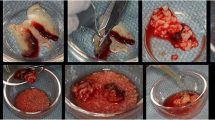
In this study, a novel bone graft combination was prepared using eggshell (ES), chitosan (Ch), and iron nanoparticles (FeNp). The in vitro properties of the graft were evaluated for cell proliferation, cell differentiation, and antibacterial properties. Acridine orange staining and von Kossa staining were used to quantify cell proliferation and mineralization. A clinically randomized design was employed, with participants allocated to the test group receiving the eggshell-derived FeNp bone graft and the control group receiving a commercially available bone graft. The study included 10 patients undergoing third molar tooth extraction, and bone repair post-grafting at the site was assessed at various time points (start, 30, 60, and 90 days). Clinical examinations measured wound probing and healing depth. Radiographs were used to assess changes in bone density, development characteristics, and height. Results: The results demonstrated that the test group, compared to the control group, exhibited antibacterial properties and higher proliferation rates (p < 0.05). The clinical trial revealed increased mean radiographic vertical bone level and bone density from baseline to 90 days, with statistically significant differences observed between the eggshell-derived bone graft and commercially available bone grafts (p < 0.05).
Conclusion
In conclusion, the eggshell-derived FeNp bone graft substitute demonstrated improved socket healing compared to commercially available bone grafts. Due to its origin from eggshell and negligible risk of disease transmission, the eggshell-derived bone graft provides a safe alternative to allografts. Moreover, the eggshell-FeNp graft substitute offers advantages such as low cost and high quality. However, further research is necessary to evaluate the efficacy of these grafts in larger patient populations.










Similar content being viewed by others
References
Kim SH, Kim W, Cho JH et al (2008) Comparison of bone formation in rabbits using hydroxyapatite and b-tricalcium phosphate scaffolds fabricated from egg shells. Adv Mater Res 47–50:999–1002
Park J, Bae S, Suh J et al (2008) Evaluation of bone healing with eggshell-derived bone graft substitutes in rat calvaria: a pilot study. J Biomed Mater Res A 87:203–214
Cai Y-D, Wang S-M, Chou AHK et al (2012) Zinc-coated car bonate apatite derived from avian eggshell for potential use as bone substitute. Part I: preparation and properties. Implant Dent 21:230–235
Kattimani VS, Chakravarthi PS, Kanumuru NR et al (2014) Eggshell derived hydroxyapatite as bone graft substitute in the healing of maxillary cystic bone defects: a preliminary report. J Int Oral Health 6:15–19
Kattimani V, Lingamaneni KP, Chakravarthi PS et al (2016) Eggshell-derived hydroxyapatite: a new era in bone regeneration. J CraniofacSurg 27:112–117
Kattimani VS, Lingamaneni KP (2019) Natural bioceramics: our experience with changing perspectives in the reconstruction of maxillofacial skeleton. J Korean Assoc Oral MaxillofacSurg 45:34–42
Kattimani V, Lingamaneni KP, Ek G, et al (2019) Eggshell derived nanohydroxyapatite(nHA) with platelet-rich fibrin (PRF) as a barrier membrane – a new technique for socket preservation. J Korean Assoc Oral MaxillofacSurg 2019.Epub ahead of print 2019, http://www.jkaoms.org/journal/view.html?uid=2929&vmd=Full
Munhoz EA, Ferreira Junior O, Yaedu RYF et al (2006) Radiographic assessment of impacted mandibular third molar sockets filled with composite xenogenic bone graft. DentoMaxillo Facial Radiol 35:371–375
Munhoz EA, Bodanezi A, Ferreira Junior O et al (2011) Bone crestal height and bone density after third-molar extraction and grafting: a long-term follow-up study. Clin Oral Invest 15:123–126
Misch CE, Dietsh F (1993) Bone-grafting materials in implant dentistry. Implant Dent 2(3):158–167
Kaher’s KL, Nagar JC. Ecografts–contemporary biologically derived bone grafts-a review
Sanosh KP, Chu MC, Balakrishnan A, Kim TN, Cho SJ (2009) Utilization of biowaste eggshells to synthesize nanocrystalline hydroxyapatite powders. Mater Lett 63(24–25):2100–2102
Salama R, Khashaba M, El Rouby D (2019) Histomorphometric evaluation of a nano-sized eggshell-containing supplement as a natural alloplast: an animal study. Saudi Dent J 31(3):375–381
Neunzehn J, Szuwart T, Wiesmann HP (2015) Eggshells as natural calcium carbonate source in combination with hyaluronan as beneficial additives for bone graft materials, an in vitro study. Head Face Med 11(1):12
Periayah MH, Halim AS, Saad AZ (2016) Chitosan: a promising marine polysaccharide for biomedical research. Pharmacogn Rev 10(19):39
Sahoo D, Sahoo S, Mohanty P, Sasmal S, Nayak PL (2009) Chitosan: a new versatile bio-polymer for various applications. Des Monomers Polym 12(5):377–404
Vinsova J, Vavrikova E (2011) Chitosan derivatives with antimicrobial, antitumour and antioxidant activities-a review. Curr Pharm Des 17(32):3596–3607
Boisselier E, Astruc D (2009) Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. ChemSoc Rev 38:1759–1782
Zeng XB, Hu H, Xie LQ, Lan F, Jiang W, Wu Y et al (2012) Magnetic responsivehydroxyapatite composite scaffolds construction for bone defect reparation. Int J Nanomed 7:3365–3378
Gergely G, Wéber F, Lukács I, Tóth AL, Horváth ZE, Mihály J, Balázsi C (2010) Preparation and characterization of hydroxyapatite from eggshell. Ceram Int 36(2):803–806
Nagahama H, Maeda H, Kashiki T, Jayakumar R, Furuike T, Tamura H (2009) Preparation and characterization of novel chitosan/gelatin membranes using chitosan hydrogel. Carbohyd Polym 76(2):255–260
Li J, Zhang F, Zhang N, Geng X, Meng C, Wang X, Yang Y (2019) Osteogenic capacity and cytotherapeutic potential of periodontal ligament cells for periodontal regeneration in vitro and in vivo. PeerJ 8(7):e6589
Kouhestani F, Dehabadi F, Shahriari MH, Motamedian SR (2018) Allogenic versus synthetic granules for bone tissue engineering: an in vitro study. Prog Biomater 7(2):133–141
Ji QX, Deng J, Xing XM, Yuan CQ, Yu XB, Xu QC, Yue J (2010) Biocompatibility of a chitosan-based injectable thermosensitive hydrogel and its effects on dog periodontal tissue regeneration. CarbohydrPolym 82:1153–1160
Potu BK, Bhat KM, Rao MS, Nampurath GK, Chamallamudi MR, Nayak SR, Muttigi MS (2009) Petroleum ether extract of Cissusquadrangularis (Linn.) enhances bone marrow mesenchymal stem cell proliferation and facilitates osteoblastogenesis. Clinics 64(10):993–998
Mahdy SA, Raheed QJ, Kalaichelvan PT (2012) Antimicrobial activity of zero-valent iron nanoparticles. Int J Modern Eng Res 2(1):578–581
Mohapatra M, Anand S (2010) Synthesis and application of nano-structured iron oxide/hydroxides—a review. Int J Eng Technol 2(8):127–146
Tran N, Mir A, Mallik D, Sinha A, Nayar S, Webster TJ (2010) Bactericidal effect of iron oxide nanoparticles on staphylococcus aureus. Int J Nanomed 5(1):277–283
Lee C, Kim JY, Lee WI, Nelson KL, Yoon J, Sedlak DL (2008) Bactericidal effect of zero-valent iron nanoparticles on Escherichia coli. Environ Sci Technol 42(13):4927–4933. https://doi.org/10.1021/es800408u
Acknowledgments
The authors would like to acknowledge Dr. Shivalingappa B Javali, MSc, Phd, Associate Professor in Statistics, Department of Community Medicine, USM-KLE International Medical program, Belagavi, Shivalingappa.javali@gmail.com. The authors would like to acknowledge all staff of Dr. Prabhakar Kore Basic Science Research Center, KLE Academy of Higher Education and Research, Belgaum, for their assistance and cooperation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nelogi, S., Roy, R. & Ambi, M. Assessing the Therapeutic Effects of Novel Antibacterial Eggshell-Iron Nanoparticle-Derived Bone Grafts and Allografts in Implanted Sockets: A Preliminary Prospective Clinical Study. J. Maxillofac. Oral Surg. (2024). https://doi.org/10.1007/s12663-024-02183-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12663-024-02183-8