Abstract
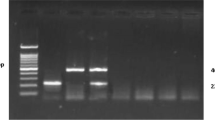
Theileria annulata (T. annulata) is a tick-borne apicomplexan parasite that affects bovine. It is endemic in many tropical and subtropics areas, including Odisha, India. The objective of this study is to identify T. annulata infection in the peripheral blood of cattle as a biological sample by conventional PCR (cPCR) and quantitative PCR (qPCR). The phylogenetic analysis was done using the T. annulata merozoite surface antigen (Tams 1) gene. Out of 552 samples of examined blood smears by microscopy, 454 (82.24%) animals were positive for Theileria species. Out of 454 samples, 96 samples were further examined by both cPCR and qPCR, 52 samples (54.16%) were found positive for T. annulata in both PCR methodologies. Phylogenetic analysis revealed that T. annulata Odisha isolate was closely related to T. annulata Uttarakhand, India isolate (KM061799) and Hyderabad, India isolate (MK034702) with Nucleotide sequence identity 95.36%, 95.25%, respectively. This is the first study to detect T. annulata by qPCR in Odisha and supported that both PCR techniques were equally effective for the detection of Tams 1 gene of T. annulata in cattle’s blood.




Similar content being viewed by others
References
Acharya AP, Panda SK, Prusty BK (2017) Diagnosis and confirmation of Theileria annulata infection in cattle in Odisha, India. J Entomol Zool Stud 5(4):1543–1546
Ananda KJ, D’Souza P, Puttalakshmamma GC (2009) Prevalence of Haemoprotozoan diseases in crossbred cattle in Bangalore north. Vet World 2:5–16
Azimi S, Mahravani H, Jeirani F, Shoshtari A (2011) Appling real time RT-PCR for bluetongue virus detection in Iran. Archi Razi 66:75–80
Bilgic HB, Karagenc T, Shiels B, Tait A, Eren H, Weir W (2010) Evaluation of cytochrome b as a sensitive target for PCR based detection of T. annulata carrier animals. Vet Parasitol 174:341–347
Bogema DR, Deutscher AT, Fell S, Collins D, Eamens GJ, Jenkins C (2015) Development and validation of a quantitative PCR assay using multiplexed hydrolysis probes for detection and quantification of Theileria orientalis isolates and differentiation of clinically relevant subtypes. J Clin Microbiol 53:941–950
Branco S, Orvalho J, Leitao A, Pereira I, Malta M, Mariano I, Carvalho T, Baptista R, Shiels BR, Peleteiro MC (2010) Fatal cases of Theileria annulata infection in calves in Portugal associated with neoplastic-like lymphoid cell proliferation. J Vet Sci 11:27–34
d’Oliveira C, Van Der Weide M, Habela P, Jacquiet P, Jongejan F (1995) Detection of Theileria annulata in blood samples of carrier cattle by PCR. J Clin Microbiol 13:2665–2669
Dandasena D, Bhandari V, Sreenivasamurthy GS, Murthy S, Roy S, Bhanot V, Singh S, Sharma P (2018) A Real-Time PCR based assay for determining parasite to host ratio and parasitaemia in the clinical samples of Bovine Theileriosis. Sci Rep 8(1):15441
Demessie Y, Derso S (2015) Tick Borne Hemoparasitic Diseases of Ruminants: A Review. Adv Bio Res 9:210–224
Gebrekidan H, Gasser RB, Stevenson MA, Jabbar A (2018) Multiplexed tandem PCR (MT-PCR) assay using the major piroplasm surface protein gene for the diagnosis of Theileria orientalis infection in cattle. J Clin Microbiol 56:1661–1671
Ghaemi P, Hoghooghi-Rad N, Shayan P, Eckert B (2012) Detection of Theileria orientalis in Iran by semi-nested PCR. Parasitol Res 110:527–531
Godara R, Sharma RL, Sharma CS (2009) Bovine Tropical Theileriosis in a neonate calf. Trop Anim Health Prod 42:551–553
Jain K (1993) Essentials of Veterinary Hematology. Blackwell Publishing, U.S.A
Kearns A, Guiver M, James V, King J (2001) Development and evaluation of a real-time quantitative PCR for the detection of human cytomegalovirus. J Virol Methods 95:121–131
Kundave VR, Patel AK, Patel PV, Hasnani JJ, Joshi CG (2014) Qualitative and quantitative assessment of Theileria annulata in cattle and buffaloes by Polymerase Chain Reaction. Trop Biomed 31(4):728–735
Mohammad Al-Saeed AT, Omer LT, Abdo J, Habibi G, Salih DA, Seitzer U, Ahmed J (2010) Epidemiological studies on tropical theileriosis (Theileria annulata infection of cattle) in Kurdistan Region, Iraq. Parasitol Res 106(2):403–407
Nazifi S, Razavi SM, Esmailnejad Z, Gheisari H (2009) Study on acute phase proteins (haptoglobin, serum amyloid A, fibrinogen, and ceruloplasmin) changes and their diagnostic values in bovine tropical theileriosis. Parasitol Res 105:41–46
Niedbalski W, Ksy A, Procajo Z, Szweda W, Mikulska SE, Platt SA, Guo-Wen L, Zhi-Gang Z, Wen-Yan Y, Lian-Yu Y (2010) Rapid detection and quantification of foot-and-mouth disease virus by a real-time reverse transcription PCR. B Vet I Pulawy 54
Pereira MR, Rocha-Silva F, Graciele-Melo C, Lafuente CR, Magalhães T, Caligiorne RB (2014) Comparison between conventional and real-time PCR assays for diagnosis of visceral leishmaniasis. Biomed Res Int 63:93–100
Perera PK, Gasser RB, Firestone SM, Smith L, Roeber F, Jabbar A (2015) A semiquantitative multiplexed tandem PCR for detection and differentiation of four Theileria orientalis genotypes in cattle. J Clin Microbiol 53:79–87
Ros-García A, Nicolás A, García-Pérez AL, Juste RA, Hurtado A (2012) Development and evaluation of a real-time PCR assay for the quantitative detection of Theileria annulata in cattle. Parasit Vectors 5:171
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Santos M, Soares R, Costa P (2013) Revisiting the Tams1-encoding gene as a species-specific target for the molecular detection of Theileria annulata in bovine blood samples. Ticks Tick Borne Dis 4:72–77
Sengupta PP, Bansal GC, Ray D (1993) Ocular lesions in experimental theileriosis. J Vet Parasitol 7:127–129
Sitotaw T, Regassa F, Zeru F, Kahsay AG (2014) Epidemiological significance of major hemoparasites of ruminants in and around Debre-Zeit, Central Ethiopia. J Parasitol Vector Bio 6:16–22
Sivakumar T, Kothalawala H, Abeyratne SA, Vimalakumar SC, Meewewa AS, Hadirampela DT, Puvirajan T, Sukumar S, Kuleswarakumar K, Chandrasiri AD, Igarashi I, Yokoyama N (2012) A PCR-based survey of selected Babesia and Theileria parasites in cattle in Sri Lanka.Vet Parasitol 190(1–2):263–267
Sudan V, Sharma RL, Yadav R, Borah MK (2012) Turning sickness in a cross bred cow naturally infected with Theileria annulata. J Parasit Dis 36:226–229
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA 6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30(12):2725–2729
Zaeemi M, Haddadzadeh H, Khazraiinia P, Kazemi B, Bandehpour M (2011) Identification of different Theileria species (Theileria lestoquardi, Theileria ovis, and Theileria annulata) in naturally infected sheep using nested PCR–RFLP. Parasitol Res 108:837–843
Zarlenga DS, Higgins J (2001) PCR as adiagnostic and quantitative technique in veterinary parasitology. Vet Parasitol 101:215–230
Author information
Authors and Affiliations
Contributions
SKS and IN conceived and designed the study; AS and GRG contributed to the writing and revision of the manuscript; MS performed laboratory testing; AS and CM contributed to the genome sequencing and phylogenetic analysis. SS and MD read and revised the manuscript. All authors’ read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors of this study stated that they have no conflicts of interest.
Ethical standards
There is no precise law for blood sample collection in India for clinical diagnosis and so no consent was obligatory. In this study 4 ml blood collected from the jugular vein under aseptic conditions from cattle after taking the permission of the farm owners.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Selim, A.M., Das, M., Senapati, S.K. et al. Molecular detection of Theileria annulata infection in cattle by conventional PCR and quantitative real time PCR in India. J Parasit Dis 45, 72–77 (2021). https://doi.org/10.1007/s12639-020-01278-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-020-01278-6