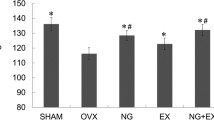
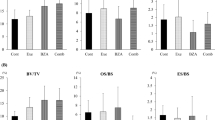
Abstract
Osteoporosis increases bone fragility and fractures. Preptin hormone is regulated by moderate exercise training and increases bone formation. Therefore, this study was conducted to see how estradiol administration and moderate exercise training affected osteoporotic changes in ovariectomized (OVX) rats. To achieve this aim, 36 healthy adult female Wistar albino rats were randomized into Sham, OVX, ovariectomized estradiol-treated (OVX + E) (OVX + E rats were treated using subcutaneous estradiol benzoate 2.5 μg/kg body weight/day), ovariectomized practicing moderate exercise training, ovariectomized estradiol-treated and practiced a moderate exercise training, and ovariectomized alendronate-treated (OVX + Alen) (OVX + Alen rats were treated orally with alendronate 3 mg/kg body weight/week) groups. Alendronate was used as a standard anti-osteoporotic drug. Moderate exercise training, including therapy with estradiol and alendronate for OVX rats began on the fourth week and lasted for six weeks. Results showed that OVX rats had estrogen and preptin deficiency in serum. These deficiencies were associated with a significant increase in bone resorption biomarkers (urinary deoxypyridinoline and hydroxyproline), and bone formation biomarkers (serum osteocalcin and bone-specific alkaline phosphatase). Also, serum pro-inflammatory cytokines (tumor necrosis factor alpha and interleukin-6) were increased, while bone osteopontin (OPN) expression was decreased. Subsequently, the osteoporotic alterations were verified based on histopathological changes. From the results, estradiol therapy and moderate exercise training significantly improved these findings to the same extent as that of the standard alendronate treatment. Therefore, through their anti-inflammatory properties, increasing bone OPN expression, and regulating serum preptin; estradiol therapy and moderate exercise training can reduce osteoporotic alterations in OVX rats. Thus, combined estradiol therapy and moderate exercise training could be a promising potential therapeutic protocol to reduce postmenopausal osteoporosis. Also, targeting serum preptin and bone osteopontin regulation could have a critical role in the treatment of postmenopausal osteoporosis.








Similar content being viewed by others
References
Abdel-Sater K, Mansour H (2012) Effects of leptin on metabolic bone turnover in ovariectomy rats. Acta Endocrinol 1841–0987:8
Abdel-Sater KA, Mansour H (2013) Bone biomarkers of ovariectomised rats after leptin therapy. Bratisl Lek Listy 114:303–307
Abu Taleb OM, Wissa MY, Abou El Nour RK, Awad HA, Moussa NM (2020) Potential effectiveness of exenatide in experimentally-induced osteoporosis. Egypt Rheumatol 42:57–62
Abuohashish HM, Ahmed MM, Al-Rejaie SS, Eltahir KEH (2015) The antidepressant bupropion exerts alleviating properties in an ovariectomized osteoporotic rat model. Acta Pharmacol Sin 36:209–220
Ahn N, Kim K (2016) Effects of 12-week exercise training on osteocalcin, high-sensitivity C-reactive protein concentrations, and insulin resistance in elderly females with osteoporosis. J Phys Ther Sci 28:2227–2231
Aydin S, Celik O, Gurates B et al (2013) Concentrations of preptin, salusins and hepcidins in plasma and milk of lactating women with or without gestational diabetes mellitus. Peptides 49:123–130
Baloğlu M, Özkorkmaz EG (2019) Biochemical and immunohistochemical investigations on bone formation and remodelling in ovariectomised rats with tamoxifen citrate administration. Folia Morphol 78:789–797
Bancroft J, Stevens A (2013) Theories and practice of histological techniques.) New York, London and Madrid: Churchil Livingstone
Bebars GM, Sallam SA, Gaber SS, Abdelaziz AH (2019) Assessment of Preptin peptide level in the sera of rachitic children and in breast milk of their mothers. Ital J Pediatr 45:34
Bergendal A, Kieler H, Sundström A, Hirschberg AL & Kocoska-Maras L (2016) Risk of venous thromboembolism associated with local and systemic use of hormone therapy in peri- and postmenopausal women and in relation to type and route of administration. Menopause (New York, N.Y.) 23, 593–9.
Byun JS, Lee SS (2010) Effect of soybeans and sword beans on bone metabolism in a rat model of osteoporosis. Ann Nutr Metab 56:106–112
Chen Y-J, Huang C-H, Lee I-C, Lee Y-T, Chen M-H, Young T-H (2008) Effects of cyclic mechanical stretching on the mRNA expression of tendon/ligament-related and osteoblast-specific genes in human mesenchymal stem cells. Connect Tissue Res 49:7–14
Chen S-Y, Yu H-T, Kao J-P et al (2014) An NMR metabolomic study on the effect of alendronate in ovariectomized mice. PLoS One 9:e106559
Cornish J, Callon KE, Bava U et al (2007) Preptin, another peptide product of the pancreatic beta-cell, is osteogenic in vitro and in vivo. American journal of physiology. Endocrinol Metab 292:E117–E122
Da Paz L, De Falco V, Teng N, Dos Reis L, Pereira R, Jorgetti V (2001) Effect of 17ß-estradiol or alendronate on the bone densitometry, bone histomorphometry and bone metabolism of ovariectomized rats. Braz J Med Biol Res 34:1015–1022
Daly RM, Dalla Via J, Duckham RL, Fraser SF, Helge EW (2019) Exercise for the prevention of osteoporosis in postmenopausal women: an evidence-based guide to the optimal prescription. Braz J Phys Ther 23:170–180
Ducy P, Amling M, Takeda S et al (2000) Leptin inhibits bone formation through a hypothalamic relay: a central control of bone mass. Cell 100:197–207
El-Haroun H, Soliman M, El-Gawad A (2020) Comparative study on the possible protective effect of lepidium sativum versus teriparatide in induced osteoporosis in adult male Guinea pigs. Egypt J Histol 43:931–947
Felsenberg D, Boonen S (2005) The bone quality framework: determinants of bone strength and their interrelationships, and implications for osteoporosis management. Clin Ther 27:1–11
Feng R, Wang L, Li Z et al (2019) A systematic comparison of exercise training protocols on animal models of cardiovascular capacity. Life Sci 217:128–140
Fodor D, Bondor C, Albu A, Simon S-P, Craciun A, Muntean L (2013) The value of osteopontin in the assessment of bone mineral density status in postmenopausal women. J Investig Med 61:15–21
Gallo D, Zannoni GF, Apollonio P, et al. (2005) Characterization of the pharmacologic profile of a standardized soy extract in the ovariectomized rat model of menopause: effects on bone, uterus, and lipid profile. Menopause (New York, N.Y.) 12, 589–600.
Halasy-Nagy J, Rodan G, Reszka A (2001) Inhibition of bone resorption by alendronate and risedronate does not require osteoclast apoptosis. Bone 29:553–559
Heather B, Karen J (2002) Estrogen replacement reverses ovariectomy-induced vaginal hyperalgesia in the rat. Maturitas 41:157–165
Heilmeier U, Youm J, Torabi S, Link TM (2016) Osteoporosis imaging in the geriatric patient. Curr Radiol Rep 4:18
Honjo S, Mizunuma H, Soda M, Igarashi M (1989) Effect of estrogen replacement therapy on the serum osteocalcin level in the postmenopausal and castrated women. Nihon Sanka Fujinka Gakkai Zasshi 41:1571–1576
Ibm II (2017) Statistics for Windows, Version 25.0. Armonk, NY: IBM Corporation.
Kanis JA, Gertz B, Singer F, Ortolani S (1995) Rationale for the use of alendronate in osteoporosis. Osteoporos Int 5:1–13
Kanjana K, Haygarth KS, Wu W, Bartels DM (2013) Laboratory studies in search of the critical hydrogen concentration. Radiat Phys Chem 82:25–34
Kim BT, Mosekilde L, Duan Y et al (2003) The structural and hormonal basis of sex differences in peak appendicular bone strength in rats. J Bone Mineral Res 18:150–155
Kitaura H, Marahleh A, Ohori F et al (2020) Osteocyte-related cytokines regulate osteoclast formation and bone resorption. Int J Mol Sci 21:5169
Kolios L, Hoerster AK, Sehmisch S et al (2010) Do estrogen and alendronate improve metaphyseal fracture healing when applied as osteoporosis prophylaxis? Calcif Tissue Int 86:23–32
Lasota A, Danowska-Klonowska D (1995) (2004) Experimental osteoporosis–different methods of ovariectomy in female white rats. Rocz Akad Med Bialymst 49(Suppl 1):129–131
Lee K, Jessop H, Suswillo R, Zaman G, Lanyon L (2003) Bone adaptation requires oestrogen receptor-α. Nature 424:389–389
Li N, Zheng YB, Han J et al (2013) Lower circulating preptin levels in male patients with osteoporosis are correlated with bone mineral density and bone formation. BMC Musculoskelet Disord 14:49
Mierzwicka A, Kuliczkowska-Plaksej J, Kolačkov K, Bolanowski M (2018) Preptin in women with polycystic ovary syndrome. Gynecol Endocrinol 34:470–475
Miller FN, Wiegman DL (1977) Anesthesia-induced alteration of small vessel response to norepinephrine. Eur J Pharmacol 44:331–337
Mohammad Rahimi GR, Bijeh N, Rashidlamir A (2020) Effects of exercise training on serum preptin, undercarboxylated osteocalcin and high molecular weight adiponectin in adults with metabolic syndrome. Exp Physiol 105:449–459
Müller ST, Keiler AM, Kräker K, Zierau O, Bernhardt R (2018) Influence of estrogen on individual exercise motivation and bone protection in ovariectomized rats. Lab Anim 52:479–489
Mustafa RA, NaA A, Hijazi HH, Header EA, Azzeh FS (2018) Biological effect of calcium and vitamin D dietary supplements against osteoporosis in ovariectomized rats. Prog Nutr 20:86–93
Nazari Soltan Aahmad S, Nourollahi S, Kazerouni F et al (2018) Investigation of the relation between bone mass density and serum preptin levels in pre- and postmenopausal women. J Bone Miner Metab 36:710–715
Notomi T, Okimoto N, Okazaki Y, Nakamura T, Suzuki M (2003) Tower climbing exercise started 3 months after ovariectomy recovers bone strength of the femur and lumbar vertebrae in aged osteopenic rats. J Bone Mineral Res 18:140–149
Novelli E, Diniz Y, Galhardi C et al (2007) Anthropometrical parameters and markers of obesity in rats. Lab Anim 41:111–119
Osman AS, Aly Labib DA, Omar AI (2018) Do acid suppressive drugs (pantoprazole and ranitidine) attenuate the protective effect of alendronate in estrogen-deficient osteoporotic rats? Egypt Rheumatol 40:99–106
Osterhoff G, Morgan EF, Shefelbine SJ, Karim L, Mcnamara LM, Augat P (2016) Bone mechanical properties and changes with osteoporosis. Injury 47:S11–S20
Ozkan Y, Timurkan ES, Aydin S et al (2013) Acylated and desacylated ghrelin, preptin, leptin, and nesfatin-1 Peptide changes related to the body mass index. Int J Endocrinol 2013:236085–236085
Özşahin ETT, Çam B, Dere F et al (2017) The effect of alendronate sodium on trabecular bone structure in an osteoporotic rat model. Turkish J Phys Med Rehabil 63:165
Popović T, Šrbić R, Matavulj M, Obradović Z & Sibinčić S (2016) Experimental model of osteoporosis on 14 weeks old ovariectomised rats: biochemical, histological and biomechanical study. Biologia Serbica 38.
Pytlik M, Kaczmarczyk-Sedlak I, Sliwiński L, Janiec W, Rymkiewicz I (2004) Effect of concurrent administration of alendronate sodium and retinol on development of changes in histomorphometric parameters of bones induced by ovariectomy in rats. Pol J Pharmacol 56:571–579
Röhling M, Herder C, Stemper T & Müssig K (2016) Influence of acute and chronic exercise on glucose uptake. J Diabet Res
Salazar M, Hernandes L, Ramos AL et al (2015) Effect of alendronate sodium on tooth movement in ovariectomized rats. Arch Oral Biol 60:776–781
Sharaf H, Shaffie N, Morsy F, Badawi M, Abbas N (2015) Role of some phytoestrogens in recovering bone loss: histological results from experimental ovariectomized rat models. J Arab Soc Med Res 10:65–75
Silva J, Fernandes T, Soci UP, Monteiro AW, Phillips MI, Em DEO (2012) Swimming training in rats increases cardiac MicroRNA-126 expression and angiogenesis. Med Sci Sports Exerc 44:1453–62
Singh A, Gill G, Kaur H, Amhmed M, Jakhu H (2018) Role of osteopontin in bone remodeling and orthodontic tooth movement: a review. Prog Orthod 19:1–8
Stachenfeld NS (2014) Hormonal changes during menopause and the impact on fluid regulation. Reproduct Sci (Thousand Oaks, Calif.) 21: 555–561
Stavropoulou A, Christopoulou GE, Anastassopoulos G et al (2005) Alteration in serum leptin correlates with alterations in serum N-telopeptide of collagen type I and serum osteocalcin during the progression of osteoporosis in ovariectomized rats. Clin Chem Lab Med 43:1359–1365
Sun X, Fengbo L, Xinlong M et al (2015) The effects of combined treatment with naringin and treadmill exercise on osteoporosis in ovariectomized rats. Sci Rep 5:1–9
Surve VV, Andersson N, Lehto-Axtelius D, Håkanson R (2001) Comparison of osteopenia after gastrectomy, ovariectomy and prednisolone treatment in the young female rat. Acta Orthop Scand 72:525–532
Suzuki N, Yoshimura Y, Deyama Y, Suzuki K, Kitagawa Y (2008) Mechanical stress directly suppresses osteoclast differentiation in RAW264. 7 cells. Int J Mol Med 21:291–296
Taherdoost H (2016) Sampling methods in research methodology; how to choose a sampling technique for research. How to Choose a Sampling Technique for Research
Tanaka S, Narusawa K, Onishi H et al (2011) Lower osteocalcin and osteopontin contents of the femoral head in hip fracture patients than osteoarthritis patients. Osteoporos Int 22:587–597
Tomeleri CM, Ribeiro AS, Souza MF et al (2016) Resistance training improves inflammatory level, lipid and glycemic profiles in obese older women: A randomized controlled trial. Exp Gerontol 84:80–87
Turkozan N, Ulusoy A, Balcioglu H, Bal F, Ozel S (2009) The effects of ovariectomy and naproxen treatment on the strength of femoral midshaft and molar alveolar region in rats. Int J Morphol 27:659–666
Weitzmann MN, Pacifici R (2006) Estrogen deficiency and bone loss: an inflammatory tale. J Clin Investig 116:1186–1194
Xiao C, Li W, Lu T, Wang J, Han J (2019) Preptin promotes proliferation and osteogenesis of MC3T3-E1 cells by upregulating β-catenin expression. IUBMB Life 71:854–862
Xiong H, Wei L, Hu Y, Zhang C, Peng B (2010) Effect of alendronate on alveolar bone resorption and angiogenesis in rats with experimental periapical lesions. Int Endod J 43:485–491
Yarrow JF, Mccoy SC, Ferreira JA et al (2012) A rehabilitation exercise program induces severe bone mineral deficits in estrogen deficient rats following extended disuse. Menopause (New York, NY) 19:1267
Yoon K-H, Cho D-C, Yu S-H, Kim K-T, Jeon Y, Sung J-K (2012) The change of bone metabolism in ovariectomized rats : analyses of microct scan and biochemical markers of bone turnover. J Korean Neurosurg Soc 51:323–327
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
All authors contributed in research design and interpretation of the studies. KAA, HOM, and NAM conducted experiments and performed data analysis. KAA, RRAA, RSR, and NAM contributed to review and writing of the manuscript. All individuals contributed in this research are included in the list of authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdelfattah Abulfadle, K., Refaat Abdelkader Atia, R., Osama Mohammed, H. et al. The potential anti-osteoporotic effect of exercise—induced increased preptin level in ovariectomized rats. Anat Sci Int 98, 22–35 (2023). https://doi.org/10.1007/s12565-022-00666-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-022-00666-7