Abstract
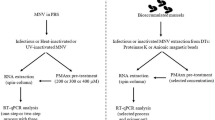
Norovirus is the leading cause of viral gastroenteritis globally. While person-to-person transmission is most commonly reported route of infection, human norovirus is frequently associated with foodborne transmission, including through consumption of contaminated bivalve molluscan shellfish. Reverse transcription (RT)-qPCR is most commonly used method for detecting human norovirus detection in foods, but does not inform on its infectivity, posing challenges for assessing intervention strategies aimed at risk elimination. In this study, RT-qPCR was used in conjunction with a derivative of the photoreactive DNA binding dye propidium monoazide (PMAxx™) (PMAxx-RT-qPCR) to evaluate the viral capsid integrity of norovirus genogroup I and II (GI and GII) in shellfish following high pressure processing (HPP). Norovirus GI.3 and GII.4 bioaccumulated oysters were subjected to HPP at pressures of 300 and 450 MPa at 15 °C, and 300, 450 and 600 MPa at 20 °C. Samples were analysed using both RT-qPCR and PMAxx-RT-qPCR. For each sample, norovirus concentration (genome copies/g digestive tissue) determined by RT-qPCR was divided by the PMAxx-RT-qPCR concentration, giving the relative non-intact (RNI) ratio. The RNI ratio values relate to the amount of non-intact (non-infectious) viruses compared to fully intact (possible infectious) viruses. Our findings revealed an increasing RNI ratio value, indicating decreasing virus integrity, with increasing pressure and decreasing pressure. At 300 MPa, for norovirus GI, the median [95% confidence interval, CI] RNI ratio values were 2.6 [1.9, 3.0] at 15 °C compared to 1.1 [0.9, 1.8] at 20 °C. At 450 MPa, the RNI ratio values were 5.5 [2.9, 7.0] at 15 °C compared to 1.3 [1.0, 1.6] at 20 °C. At 600 MPa, the RNI ratio value was 5.1 [2.9, 13.4] at 20 °C. For norovirus GII, RT-qPCR and PMAxx-RT-qPCR detections were significantly reduced at 450 and 600 MPa at both 15 °C and 20 °C, with the median [95% CI] RNI ratio value at 300 MPa being 1.1 [0.8, 1.6]. Following HPP treatment, the use of PMAxx-RT-qPCR enables the selective detection of intact and potential infectious norovirus, enhancing our understanding of the inactivation profiles and supporting the development of more effective risk assessment strategies.



Similar content being viewed by others
References
Arcangeli, G., Terregino, C., De Benedictis, P., Zecchin, B., Manfrin, A., Rossetti, E., Magnabosco, C., Mancin, M., & Brutti, A. (2012). Effect of high hydrostatic pressure on murine norovirus in Manila clams. Letters in Applied Microbiology, 54(4), 325–329. https://doi.org/10.1111/j.1472-765X.2012.03211.x
Bányai, K., Estes, M. K., Martella, V., & Parashar, U. D. (2018). Viral gastroenteritis. Lancet, 392, 175–186. https://doi.org/10.1016/S0140-6736(18)31128-0
Campos, C., Goblick, J., Lee, R., Wittamore, K., & Lees, D. N. (2017). Determining the zone of impact of norovirus contamination in shellfish production areas through microbiological monitoring and hydrographic analysis. Water Research, 124, 556–565. https://doi.org/10.1016/j.watres.2017.08.021
Canh, V. D., Kasuga, I., Furumai, H., & Katayama, H. (2019). Viability RT-qPCR combined with sodium deoxycholate pre-treatment for selective quantification of infectious viruses in drinking water samples. Food and Environmental Virology, 11(1), 40–51. https://doi.org/10.1007/s12560-019-09368-2
de Graaf, M., Villabruna, N., & Koopmans, M. P. (2017). Capturing norovirus transmission. Current Opinion in Virology, 22, 64–70. https://doi.org/10.1016/j.coviro.2016.11.008
Elizaquível, P., Aznar, R., & Sánchez, G. (2014). Recent developments in the use of viability dyes and quantitative PCR in the food microbiology field. Journal of Applied Microbiology, 116(1), 1–13. https://doi.org/10.1111/jam.12365
Estes, M. K., Ettayebi, K., Tenge, V. R., Murakami, K., Karandikar, U., Lin, S. C., Ayyar, B. V., Cortes-Penfield, N. W., Haga, K., Neill, F. H., Opekun, A. R., Broughman, J. R., Zeng, X. L., Blutt, S. E., Crawford, S. E., Ramani, S., Graham, D. Y., & Atmar, R. L. (2019). Human norovirus cultivation in nontransformed stem cell-derived human intestinal enteroid cultures: Success and challenges. Viruses, 11(7), 638. https://doi.org/10.3390/v11070638
Ettayebi, K., Crawford, S. E., Murakami, K., Broughman, J. R., Karandikar, U., Tenge, V. R., Neill, F. H., Blutt, S. E., Zeng, X. L., Qu, L., Kou, B., Opekun, A. R., Burrin, D., Graham, D. Y., Ramani, S., Atmar, R. L., & Estes, M. K. (2016). Replication of human noroviruses in stem cell-derived human enteroids. Science, 353(6306), 1387–1393. https://doi.org/10.1126/science.aaf5211
Fittipaldi, M., Rodriguez, N. J., Codony, F., Adrados, B., Peñuela, G. A., & Morató, J. (2010). Discrimination of infectious bacteriophage T4 virus by propidium monoazide real-time PCR. Journal of Virological Methods, 168(1–2), 228–232. https://doi.org/10.1016/j.jviromet.2010.06.011
Govaris, A., & Pexara, A. (2021). Inactivation of foodborne viruses by high-pressure processing (HPP). Foods, 10(2), 215. https://doi.org/10.3390/foods10020215
Gyawali, P., Fletcher, G. C., McCoubrey, D.-J., & Hewitt, J. (2019). Norovirus in shellfish: An overview of post-harvest treatments and their challenges. Food Control, 99, 171–179. https://doi.org/10.1016/j.foodcont.2018.12.049
Gyawali, P., & Hewitt, J. (2018). Detection of infectious noroviruses from wastewater and seawater using PEMAX™ treatment combined with RT-qPCR. Water, 10(7), 841. https://doi.org/10.3390/w10070841
Hardstaff, J. L., Clough, H. E., Lutje, V., McIntyre, K. M., Harris, J. P., Garner, P., & O’Brien, S. J. (2018). Foodborne and food-handler norovirus outbreaks: A systematic review. Foodborne Pathogens and Disease, 15(10), 589–597. https://doi.org/10.1089/fpd.2018.2452
Hewitt, J., Rivera-Aban, M., & Greening, G. E. (2009). Evaluation of murine norovirus as a surrogate for human norovirus and hepatitis A virus in heat inactivation studies. Journal of Applied Microbiology, 107(1), 65–71.
Imamura, S., Kanezashi, H., Goshima, T., Suto, A., Ueki, Y., Sugawara, N., Ito, H., Zou, B., Uema, M., Noda, M., & Akimoto, K. (2017). Effect of high-pressure processing on human noroviruses in laboratory-contaminated oysters by bio-accumulation. Foodborne Pathogens and Disease, 14(9), 518–523. https://doi.org/10.1089/fpd.2017.2294
ISO 15216-1:2017. (2017). Microbiology of the food chain—Horizontal method for determination of hepatitis A virus and norovirus using real-time RT-PCR—Part 1: Method for quantification. https://www.iso.org/standard/65681.html
Jeon, E. B., Choi, M., Kim, J. Y., Ha, K. S., Kwon, J. Y., Jeong, S. H., Lee, H., Jung, Y. J., Ha, J., & Park, S. (2020). Characterizing the effects of thermal treatment on human norovirus GII.4 viability using propidium monoazide combined with RT-qPCR and quality assessments in mussels. Food Control, 109, 106954. https://doi.org/10.1016/j.foodcont.2019.106954
Kageyama, T., Kojima, S., Shinohara, M., Uchida, K., Fukushi, S., Hoshino, F. B., Takeda, N., & Katayama, K. (2003). Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. Journal of Clinical Microbiology, 41(4), 1548–1557. https://doi.org/10.1128/JCM.41.4.1548-1557.2003
Karim, M. R., Fout, G. S., Johnson, C. H., White, K. M., & Parshionikar, S. U. (2015). Propidium monoazide reverse transcriptase PCR and RT-qPCR for detecting infectious enterovirus and norovirus. Journal of Virological Methods, 219, 51–61. https://doi.org/10.1016/j.jviromet.2015.02.020
Kazama, S., Miura, T., Masago, Y., Konta, K., Tohma, K., Manaka, T., Liu, X., Nakayama, T., Tanno, M., Saito, H., Oshitani, T., & Omura, T. (2017). Environmental surveillance of norovirus genogroups I and II for sensitive detection of epidemic variants. Applied and Environmental Microbiology, 83, e03406–e03416. https://doi.org/10.1128/AEM.03406-16
Kingsley, D. H. (2014). High pressure processing of bivalve shellfish and HPP’s use as a virus intervention. Foods, 3(2), 336–350. https://doi.org/10.3390/foods3020336
Kingsley, D. H., Hoover, D. G., Papafragkou, E., & Richards, G. P. (2002). Inactivation of hepatitis A virus and a calicivirus by high hydrostatic pressure. Journal of Food Protection, 65(10), 1605–1609. https://doi.org/10.4315/0362-028x-65.10.1605
Langlet, J., Kaas, L., Croucher, D., & Hewitt, J. (2018). Effect of the shellfish proteinase K digestion method on norovirus capsid integrity. Food and Environmental Virology, 10, 151–158. https://doi.org/10.1007/s12560-018-9336-6
Leifels, M., Cheng, D., Sozzi, E., Shoults, D. C., Wuertz, S., Mongkolsuk, S., & Sirikanchana, K. (2020). Capsid integrity quantitative PCR to determine virus infectivity in environmental and food applications—a systematic review. Water Research X, 11, 100080. https://doi.org/10.1016/j.wroa.2020.100080
Leifels, M., Jurzik, L., Wilhelm, M., & Hamza, I. A. (2015). Use of ethidium monoazide and propidium monoazide to determine viral infectivity upon inactivation by heat, UV- exposure and chlorine. International Journal of Hygiene and Environmental Health, 218(8), 686–693. https://doi.org/10.1016/j.ijheh.2015.02.003
Leon, J. S., Kingsley, D. H., Montes, J. S., Richards, G. P., Lyon, G. M., Abdulhafid, G. M., Seitz, S. R., Fernandez, M. L., Teunis, P. F., Flick, G. J., & Moe, C. L. (2011). Randomized, double-blinded clinical trial for human norovirus inactivation in oysters by high hydrostatic pressure processing. Applied and Environmental Microbiology, 77(15), 5476–5482. https://doi.org/10.1128/AEM.02801-10
Li, D., Tang, Q. J., Wang, J. F., Wang, Y. M., Zhao, Q., & Xue, C. H. (2009). Effects of high-pressure processing on murine norovirus-1 in oysters (Crassostrea gigas) in situ. Food Control, 20(11), 992–996. https://doi.org/10.1016/j.foodcont.2008.11.012
Li, X., Chen, C., & Kingsley, D. (2013). The influence of temperature, pH, and water immersion on the high hydrostatic pressure inactivation of GI.1 and GII.4 human noroviruses. International Journal of Food Microbiology, 167(2), 138–143. https://doi.org/10.1016/j.ijfoodmicro.2013.08.020
Li, X., & Chen, H. (2015). Evaluation of the porcine gastric mucin binding assay for high-pressure-inactivation studies using murine norovirus and Tulane virus. Applied and Environmental Microbiology, 81(2), 515–521. https://doi.org/10.1128/AEM.02971-14
Li, X., Huang, R., & Chen, H. (2017). Evaluation of assays to quantify infectious human norovirus for heat and high-pressure inactivation studies using Tulane virus. Food and Environmental Virology, 9(3), 314–325. https://doi.org/10.1007/s12560-017-9288-2
Lou, F., Neetoo, H., Chen, H., & Li, J. (2011). Inactivation of a human norovirus surrogate by high-pressure processing: Effectiveness, mechanism, and potential application in the fresh produce industry. Applied and Environmental Microbiology, 77(5), 1862–1871. https://doi.org/10.1128/AEM.01918-10
Lou, F., Ye, M., Ma, Y., Li, X., DiCaprio, E., Chen, H., Krakowka, S., Hughes, J., Kingsley, D., & Li, J. (2015). A gnotobiotic pig model for determining human norovirus inactivation by high-pressure processing. Applied and Environmental Microbiology, 81(19), 6679–6687. https://doi.org/10.1128/AEM.01566-15
Lowther, J. A., Gustar, N. E., Powell, A. L., Hartnell, R. E., & Lees, D. N. (2012). Two-year systematic study to assess norovirus contamination in oysters from commercial harvesting areas in the United Kingdom. Applied and Environmental Microbiology, 78(16), 5812–5817. https://doi.org/10.1128/AEM.01046-12
Masago, Y., Konta, Y., Kaza, S., Inaba, M., Imagawa, T., Tohma, K., Saito, M., Suzuki, A., Oshitani, H., & Omura, T. (2016). Comparative evaluation of real-time PCR methods for human noroviruses in wastewater and human stool. PLoS ONE, 11, e0160825. https://doi.org/10.1371/journal.pone.0160825
Parshionikar, S., Laseke, I., & Fout, S. G. (2010). Use of propidium monoazide in reverse transcriptase PCR to distinguish between infectious and noninfectious enteric virus in water samples. Applied and Environmental Microbiology, 76(13), 4318–4326.
Rachmadi, A. T., Kitajima, M., Pepper, I. A., & Gerba, C. P. (2016). Enteric and indicator virus removal by surface flow wetlands. Science of the Total Environment, 542, 976–982. https://doi.org/10.1016/j.scitotenv.2015.11.001
Randazzo, W., Khezri, M., Ollivier, J., Le Guyader, F. S., Rodríguez-Díaz, J., Aznar, R., & Sánchez, G. (2018). Optimization of PMAxx pretreatment to distinguish between human norovirus with intact and altered capsids in shellfish and sewage samples. International Journal of Food Microbiology, 266, 1–7. https://doi.org/10.1016/j.ijfoodmicro.2017.11.011
Randazzo, W., López-Gálvez, F., Allende, A., Aznar, R., & Sánchez, G. (2016). Evaluation of viability PCR performance for assessing norovirus infectivity in fresh-cut vegetables and irrigation water. International Journal of Food Microbiology, 229, 1–6. https://doi.org/10.1016/j.ijfoodmicro.2016.04.010
Richards, G. P. (2012). Critical review of norovirus surrogates in food safety research: Rationale for considering volunteer studies. Food and Environmental Virology, 4(1), 6–13. https://doi.org/10.1007/s12560-011-9072-7
Roy, P. K., Jeon, E. B., Kim, J. Y., & Park, S. Y. (2023). Application of high-pressure processing (or high hydrostatic pressure) for the inactivation of human norovirus in Korean traditionally preserved raw crab. Viruses, 15(7), 1599. https://doi.org/10.3390/v15071599
Sánchez, G., Aznar, R., Martínez, A., & Rodrigo, D. (2011). Inactivation of human and murine norovirus by high-pressure processing. Foodborne Pathogens and Disease, 8(2), 249–253. https://doi.org/10.1089/fpd.2010.0667
Sarmento, S. K., Guerra, C. R., Malta, F. C., Coutinho, R., Miagostovich, M. P., & Fumian, T. M. (2020). Human norovirus detection in bivalve shellfish in Brazil and evaluation of viral infectivity using PMA treatment. Marine Pollution Bulletin, 157, 111315. https://doi.org/10.1016/j.marpolbul.2020.111315
Takahashi, M., Okakura, Y., Takahashi, H., Yamane, H., Akashige, S., Kuda, T., & Kimura, B. (2019). Evaluation of inactivation of murine norovirus in inoculated shell oysters by high hydrostatic pressure treatment. Journal of Food Protection, 82(12), 2169–2173. https://doi.org/10.4315/0362-028X.JFP-19-186
Tong, L., Ding, G., Yang, M., Su, L., Wang, S., Wang, Y., Zheng, L., Zhou, D., & Zhao, F. (2023). High-hydrostatic-pressure inactivation of GI.5 and GII.4 human norovirus and effects on the physical, chemical, and taste characteristics of oyster (Crassostrea virginica). LWT Food Science and Technology. https://doi.org/10.1016/j.lwt.2023.114554
Wales, S. Q., Pandiscia, A., Kulka, M., Sanchez, G., & Randazzo, W. (2024). Challenges for estimating human norovirus infectivity by viability RT-qPCR as compared to replication in human intestinal enteroids. International Journal of Food Microbiology, 411, 110507. https://doi.org/10.1016/j.ijfoodmicro.2023.110507
Wolf, S., Hewitt, J., & Greening, G. E. (2010). Viral multiplex quantitative PCR assays for tracking sources of fecal contamination. Applied and Environmental Microbiology, 76(5), 1388–1394. https://doi.org/10.1128/AEM.02249-09
Ye, M., Li, X., Kingsley, D. H., Jiang, X., & Chen, H. (2014). Inactivation of human norovirus in contaminated oysters and clams by high hydrostatic pressure. Applied and Environmental Microbiology, 80(7), 2248–2253. https://doi.org/10.1128/AEM.04260-13
Acknowledgements
The murine norovirus (MNV-1) was kindly provided by Professor H. Virgin (Washington School of Medicine, MO, US).
Funding
Funding was provided by Ministry of Business, Innovation and Employment: Strategic Science Investment Fund (Seafood Safety Research Platform, Grant No. CAWX1801).
Author information
Authors and Affiliations
Contributions
ATR planned the study, performed the experiments, analysed the results, and wrote the manuscript. PG planned the study and performed the experiments. GS performed the HPP. AJ performed the molecular assays. GF was involved in supervising the HPP experiment. JH was granted funding, conceived the original idea, wrote the manuscript and supervised the project. All authors critically reviewed the article and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rachmadi, A.T., Gyawali, P., Summers, G. et al. PMAxx-RT-qPCR to Determine Human Norovirus Inactivation Following High-Pressure Processing of Oysters. Food Environ Virol (2024). https://doi.org/10.1007/s12560-024-09585-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12560-024-09585-4